Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and fine chemical manufacturing, particularly regarding efficiency, safety, and scalability. A significant breakthrough in this domain is detailed in patent CN113004181B, which discloses a novel method for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. Thioesters are pivotal structural motifs in organic chemistry, serving as stable and versatile acyl donors for the synthesis of aldehydes, ketones, esters, and amides, and playing crucial roles in biological processes such as the Krebs cycle and native chemical ligation. However, traditional synthetic routes often rely on hazardous or operationally challenging reagents. This new methodology addresses these critical pain points by utilizing inexpensive and readily available benzyl chloride compounds and sulfonyl chlorides as starting materials, thereby offering a robust pathway for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation reaction of thiols with carboxylic acids or their derivatives. While chemically feasible, this conventional approach presents severe logistical and safety challenges for industrial-scale operations. The primary drawback is the reliance on thiol compounds as the sulfur source, which are notorious for their extremely unpleasant, pervasive odors that can contaminate entire production facilities and pose significant health risks to personnel. Furthermore, thiols are potent catalyst poisons; they can irreversibly bind to transition metal centers, deactivating the catalyst and leading to inconsistent reaction rates, lower yields, and increased waste. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes also exist but often suffer from limited substrate scope or harsh reaction conditions that are incompatible with sensitive functional groups commonly found in complex drug molecules.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally shifts the paradigm by replacing problematic thiols with sulfonyl chlorides. This substitution is transformative for process chemistry, as sulfonyl chlorides are odorless, stable, and commercially abundant commodities. The reaction employs a sophisticated catalytic system comprising palladium acetate and a bulky bidentate phosphine ligand, specifically 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), which facilitates the activation of the benzyl chloride substrate. Crucially, the process utilizes tungsten hexacarbonyl [W(CO)₆] not merely as a passive carbonyl source but also as an intrinsic reducing agent, streamlining the reagent stoichiometry. This approach ensures high reaction efficiency and broad functional group tolerance, allowing for the synthesis of diverse thioester derivatives under relatively mild conditions compared to traditional acylation protocols.
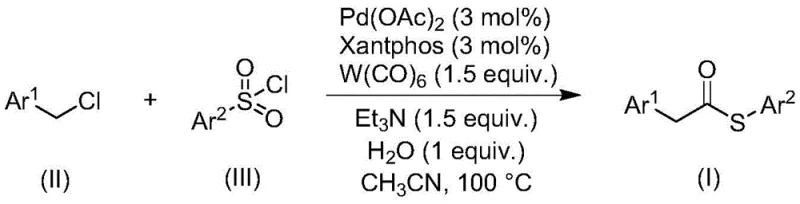
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The mechanistic elegance of this transformation lies in the synergistic interplay between the palladium catalyst and the tungsten carbonyl species. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ. The presence of the Xantphos ligand is critical here; its large bite angle stabilizes the palladium center and promotes the subsequent migratory insertion of carbon monoxide. Unlike traditional carbonylations that require high-pressure CO gas cylinders—a significant safety hazard in many jurisdictions—this method generates the necessary CO moiety from the decomposition of tungsten hexacarbonyl under the reaction conditions. This "CO-surrogate" strategy drastically enhances the safety profile of the process, making it viable for standard glass-lined reactors without specialized high-pressure equipment. Following CO insertion, the sulfonyl chloride interacts with the acyl-palladium intermediate, likely undergoing a desulfitive process to install the sulfur atom, ultimately yielding the thioester product after reductive elimination.
From an impurity control perspective, this mechanism offers distinct advantages. The use of water as a co-reagent, alongside triethylamine as a base, helps to scavenge acidic byproducts (such as HCl and SO₂) generated during the desulfitive step, preventing acid-catalyzed degradation of the product or starting materials. The specific choice of acetonitrile as the preferred organic solvent ensures that all polar and non-polar components remain in solution, facilitating homogeneous catalysis and minimizing the formation of insoluble palladium black, which can lead to product contamination. The result is a clean reaction profile where the primary impurities are easily separable via standard silica gel chromatography, ensuring that the final thioester compounds meet the stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize side reactions. The protocol involves charging a reactor with the specific molar ratios of catalyst, ligand, and reagents as defined in the patent examples, typically maintaining a benzyl chloride to sulfonyl chloride ratio of roughly 1:1.2 to drive the reaction to completion. The mixture is then heated to 100°C in acetonitrile for approximately 24 hours. This thermal energy is sufficient to activate the tungsten hexacarbonyl and sustain the catalytic cycle without requiring excessive pressure. Post-reaction processing is notably straightforward, involving simple filtration to remove inorganic salts and tungsten residues, followed by purification. For a detailed, step-by-step standard operating procedure including exact quantities and workup specifics, please refer to the guide below.
- Combine palladium acetate, Xantphos ligand, tungsten hexacarbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 24 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology represents a strategic opportunity to optimize cost structures and mitigate supply risks. The shift away from thiols eliminates the need for specialized odor-control infrastructure and expensive personal protective equipment (PPE) associated with handling volatile sulfur compounds, leading to substantial operational cost savings. Furthermore, the reliance on commodity chemicals like benzyl chlorides and sulfonyl chlorides ensures a stable and competitive supply base, reducing vulnerability to price volatility often seen with niche specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted. By utilizing tungsten hexacarbonyl as a dual-purpose reagent (carbonyl source and reducing agent), the method eliminates the procurement and handling costs associated with separate reducing agents like zinc dust or silanes. Additionally, the avoidance of high-pressure CO gas removes the capital expenditure requirement for autoclaves and gas monitoring systems, allowing the reaction to proceed in standard atmospheric or low-pressure vessels. This simplification of the hardware footprint directly translates to lower depreciation costs and reduced maintenance overheads for the manufacturing facility.
- Enhanced Supply Chain Reliability: The robustness of the starting materials cannot be overstated. Benzyl chlorides and sulfonyl chlorides are produced on a massive global scale for various industries, ensuring consistent availability and short lead times. Unlike complex chiral thiols or sensitive acid chlorides that may have single-source suppliers, these precursors are available from multiple vendors worldwide. This redundancy secures the supply chain against disruptions, ensuring continuous production schedules for critical pharmaceutical intermediates and preventing costly downtime due to raw material shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is facilitated by its homogeneous nature and moderate temperature requirements. The absence of toxic thiol waste streams significantly simplifies effluent treatment, reducing the burden on wastewater treatment plants and lowering environmental compliance costs. The reaction produces benign byproducts that are easier to manage than the heavy metal sludge often associated with alternative reduction methods. This aligns perfectly with modern green chemistry principles, enhancing the corporate sustainability profile of the manufacturing entity while ensuring regulatory compliance in strict jurisdictions.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope and operational parameters. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for process development decisions.
Q: Why is sulfonyl chloride preferred over thiols in this synthesis?
A: Sulfonyl chlorides are used as the sulfur source to avoid the unpleasant odor and catalyst poisoning issues commonly associated with thiol compounds, significantly improving operational safety and catalyst longevity.
Q: What is the dual role of tungsten hexacarbonyl in this reaction?
A: Tungsten hexacarbonyl serves a critical dual function as both the carbonyl source (providing the CO group) and the reducing agent, eliminating the need for additional external reducing agents and simplifying the reagent profile.
Q: Does this method tolerate diverse functional groups on the aromatic rings?
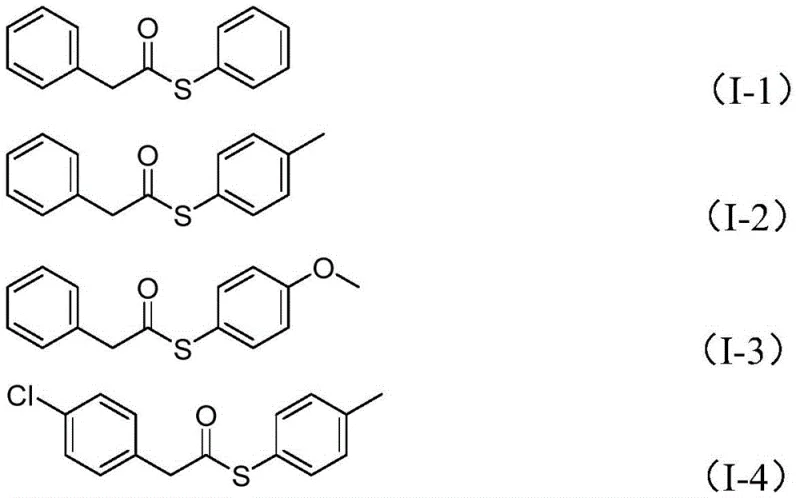
A: Yes, the method exhibits wide substrate functional group tolerance, successfully accommodating substituents such as methyl, methoxy, tert-butyl, halogens (Cl, F, Br), and alkoxycarbonyl groups on both the benzyl and sulfonyl components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of fine chemical manufacturing. Our team of expert process chemists has thoroughly analyzed the potential of this carbonylation technology and is fully equipped to translate these laboratory findings into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are designed to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee product quality.
We invite you to collaborate with us to leverage this advanced synthesis method for your project needs. Whether you require custom synthesis of specific thioester derivatives or optimization of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
