Revolutionizing Oxime Ether Production: A Metal-Free Visible Light Approach for Commercial Scale
Revolutionizing Oxime Ether Production: A Metal-Free Visible Light Approach for Commercial Scale
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the realm of fine chemical and pharmaceutical intermediate production. Patent CN114835660A introduces a groundbreaking preparation method for oxime ethers that leverages visible light-mediated, metal-free catalysis to construct critical C-O bonds. This technology represents a significant departure from traditional thermal processes, utilizing carbon tetrabromide as an inexpensive organic photocatalyst to drive cross-dehydrogenative coupling reactions under mild conditions. For R&D directors and process chemists, this innovation offers a compelling alternative to legacy methods that often rely on toxic transition metals or harsh reaction environments. The ability to synthesize complex oxime ether scaffolds with high efficiency and minimal environmental impact addresses the growing demand for sustainable manufacturing practices in the global supply chain.
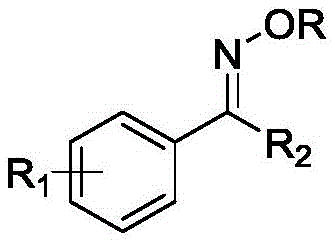
This novel approach not only simplifies the operational workflow but also significantly enhances the purity profile of the final product, a critical factor for downstream applications in drug discovery and agrochemical development. By eliminating the need for heavy metal catalysts such as copper iodide, which are prone to leaving residual traces that require costly removal steps, this method streamlines the purification process. The patent details a robust protocol that achieves impressive yields, demonstrating the viability of photo-organic catalysis for large-scale industrial applications. As companies strive to reduce their carbon footprint and adhere to stricter regulatory standards regarding metal impurities, adopting such metal-free technologies becomes a strategic imperative for maintaining competitiveness in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxime ethers has relied on methodologies that present significant challenges in terms of safety, cost, and environmental compliance. Traditional routes often involve the use of strong bases and alkylating agents in high-pressure autoclaves, requiring precise temperature control between 10°C and 60°C to manage exothermic risks. Other established methods utilize transition metal catalysts, such as copper iodide, to facilitate dehydrogenative cross-coupling reactions. While effective, these metal-catalyzed systems introduce the persistent issue of metal residue contamination, necessitating additional purification steps like chromatography or specialized scavenging treatments to meet pharmaceutical grade specifications. Furthermore, the reliance on precious or toxic metals increases the raw material costs and complicates waste disposal protocols, creating a burden on both the economic and ecological aspects of production.
The Novel Approach
In stark contrast, the methodology described in patent CN114835660A utilizes a visible light-mediated system that operates at ambient temperature and pressure, fundamentally altering the risk profile of the synthesis. By employing carbon tetrabromide as a photocatalyst, the reaction harnesses the energy of blue LED light to generate reactive bromine radicals in situ, which then abstract hydrogen atoms from cyclic ether solvents to initiate the coupling process. This metal-free strategy completely bypasses the need for transition metals, thereby eliminating the risk of heavy metal contamination in the final API or intermediate. The process is characterized by its operational simplicity, requiring only standard glassware and LED light sources rather than specialized high-pressure equipment. This shift not only reduces capital expenditure on reactor infrastructure but also significantly lowers the barrier to entry for scaling up production, making it an attractive option for contract development and manufacturing organizations seeking efficient solutions.
Mechanistic Insights into Visible Light-Mediated C-O Bond Formation
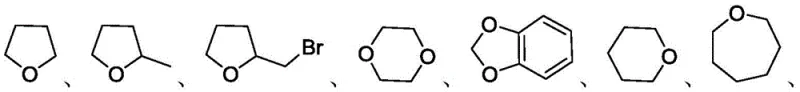
The core of this innovative synthesis lies in the unique photochemical properties of carbon tetrabromide, which undergoes homolytic cleavage of the C-Br bond upon irradiation with visible light. This process generates highly reactive bromine radicals that serve as the primary hydrogen atom transfer agents within the reaction system. These radicals selectively abstract hydrogen atoms from the alpha-position of the cyclic ether alkylating agents, such as tetrahydrofuran or tetrahydropyran, generating carbon-centered radicals. These transient species then undergo single electron transfer processes and electrophilic substitution reactions with the oxime substrates, ultimately forging the desired C-O bond to yield the oxime ether product. Understanding this radical mechanism is crucial for optimizing reaction parameters, as the efficiency of radical generation and propagation directly influences the overall conversion rate and selectivity of the transformation.
From an impurity control perspective, the absence of transition metals simplifies the impurity profile significantly, removing the need for complex metal scavenging resins or extensive aqueous workups designed to chelate metal ions. The primary byproducts are typically derived from the catalyst itself or minor over-oxidation products, which are generally easier to separate via standard column chromatography or crystallization techniques. The use of 4A molecular sieves as a water scavenger further ensures that the reaction environment remains anhydrous, preventing the hydrolysis of sensitive intermediates and maximizing the yield. This level of control over the reaction milieu allows for the production of high-purity intermediates that are essential for the synthesis of potent pharmaceuticals and agrochemicals, where even trace impurities can affect biological activity or stability.
How to Synthesize Oxime Ethers Efficiently
The practical implementation of this synthesis route involves a straightforward procedure that balances reagent stoichiometry with precise light exposure to maximize efficiency. The process begins with the careful selection of substrates, where various ketoximes or aldoximes are paired with cyclic ether solvents that act as both the reaction medium and the alkylating source. To ensure optimal performance, the reaction mixture must be thoroughly degassed and maintained under an inert atmosphere, typically argon, to prevent quenching of the radical species by oxygen. The detailed standardized synthesis steps below outline the specific conditions required to achieve the high yields reported in the patent data, serving as a foundational guide for process chemists looking to replicate or adapt this technology.
- Prepare the reaction mixture by combining ketoxime or aldoxime substrates with cyclic ether alkylating agents and carbon tetrabromide catalyst in anhydrous n-butyl acetate.
- Add 4A molecular sieves as a water scavenger to maintain anhydrous conditions essential for high yield and prevent hydrolysis side reactions.
- Irradiate the mixture with a 3W to 30W blue LED light source under argon atmosphere for 4 to 6 hours to initiate the radical cross-dehydrogenative coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible light-mediated technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of expensive transition metal catalysts translates directly into reduced raw material costs, as carbon tetrabromide is a widely available and inexpensive organic reagent compared to specialized metal complexes. Furthermore, the simplified workup procedure, which avoids complex metal removal steps, reduces the consumption of auxiliary chemicals and solvents, leading to lower operational expenditures and waste treatment costs. The mild reaction conditions also imply lower energy consumption, as there is no need for heating or high-pressure maintenance, contributing to a more sustainable and cost-effective manufacturing footprint that aligns with modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive organic halides drastically lowers the bill of materials for each batch produced. By removing the necessity for post-reaction metal scavenging processes, manufacturers can save significantly on both reagent costs and processing time, resulting in a leaner production model. The use of common solvents like n-butyl acetate, which serves dual purposes as both solvent and reactant in some variations, further optimizes the material balance and reduces inventory complexity.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various oximes and cyclic ethers, are commodity chemicals with stable and robust global supply chains. Unlike specialized catalysts that may suffer from supply bottlenecks or long lead times, the reagents for this process are readily accessible from multiple vendors. This diversity in sourcing options mitigates the risk of production delays due to material shortages, ensuring a consistent and reliable flow of intermediates to downstream customers who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: The inherent safety of operating at room temperature and atmospheric pressure makes this process highly scalable without the need for expensive pressure-rated reactors. The metal-free nature of the reaction simplifies environmental compliance, as wastewater streams do not contain heavy metal contaminants that require specialized treatment facilities. This ease of scale-up and reduced environmental burden facilitates faster technology transfer from the laboratory to pilot and commercial plants, accelerating time-to-market for new products while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-mediated oxime ether synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or R&D pipelines.
Q: What are the advantages of using Carbon Tetrabromide over Copper Catalysts for Oxime Ether synthesis?
A: Using Carbon Tetrabromide eliminates the risk of heavy metal residue contamination, which is critical for pharmaceutical applications. It also operates under milder conditions (room temperature, visible light) compared to traditional thermal methods, reducing energy costs and safety hazards associated with high-pressure reactors.
Q: Can this visible-light mediated process be scaled for industrial production?
A: Yes, the patent data demonstrates successful scale-up reactions achieving yields of 85% on a larger scale. The use of inexpensive organic catalysts and standard LED light sources makes the process highly adaptable for large-scale manufacturing without requiring specialized high-pressure equipment.
Q: What is the substrate scope for this oxime ether preparation method?
A: The method exhibits broad substrate tolerance, accommodating various ketoximes and aldoximes with electron-donating or withdrawing groups. It effectively couples with cyclic ethers such as tetrahydrofuran, tetrahydropyran, and epoxy hexane, providing versatility for synthesizing diverse chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxime Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the visible light-mediated catalysis described in patent CN114835660A. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of metal residues and other critical impurities.
We invite potential partners to engage with our technical procurement team to discuss how this metal-free technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that drive efficiency and sustainability in your supply chain.
