Scalable Synthesis of Belinostat Intermediate II for Global API Production
Scalable Synthesis of Belinostat Intermediate II for Global API Production
The pharmaceutical industry's demand for Histone Deacetylase Inhibitors (HDACIs) continues to surge, driven by the critical role of compounds like Belinostat in oncology therapy. Patent CN102786448B introduces a transformative methodology for synthesizing the key intermediate, Formula II, which serves as the cornerstone for Belinostat production. This technical insight report analyzes the strategic advantages of this six-step synthetic route, which begins with m-carboxyl phenylsulfonic acid. By shifting away from hazardous traditional reagents like oleum and diazotization gases, this process offers a safer, more environmentally compliant pathway for reliable pharmaceutical intermediates supplier networks. The innovation lies not just in the chemistry, but in the operational feasibility, ensuring that high-purity Belinostat precursors can be manufactured with reduced potential safety hazards and enhanced supply chain continuity.
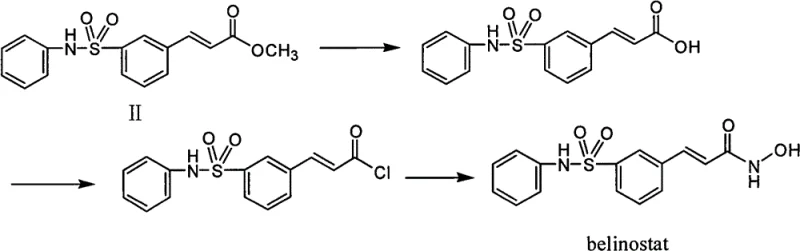
The transition from laboratory-scale discovery to commercial viability often hinges on the robustness of the intermediate synthesis. This patent details a route where every intermediate exists as a solid, facilitating easy detection and purification through recrystallization. For procurement and supply chain leaders, this translates to a drastic simplification of logistics and quality control protocols. The elimination of column chromatography, a time-consuming and solvent-intensive process, represents a significant leap forward in cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the use of common reagents such as phosphorus oxychloride and aniline ensures that raw material sourcing remains stable and unaffected by the volatility associated with specialized hazardous chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Belinostat and its precursors has been plagued by severe safety and environmental challenges. As illustrated in prior art such as WO0230879, traditional routes often rely heavily on sulfonation reactions using oleum (fuming sulfuric acid). Oleum is notoriously dangerous; it reacts violently with water, possesses strong corrosive properties, and poses significant explosion risks during storage and transport. The handling of such materials requires specialized infrastructure and rigorous safety protocols, driving up capital expenditure and operational costs. Additionally, other documented methods, such as those published in Synthetic Communications, utilize diazotization reactions and the introduction of sulfur dioxide gas. These processes introduce toxic gases into the manufacturing environment, creating substantial occupational health risks and requiring complex scrubbing systems to meet environmental regulations. The inherent instability and hazard profile of these conventional pathways make them less than ideal for modern, large-scale commercial scale-up of complex pharmaceutical intermediates.
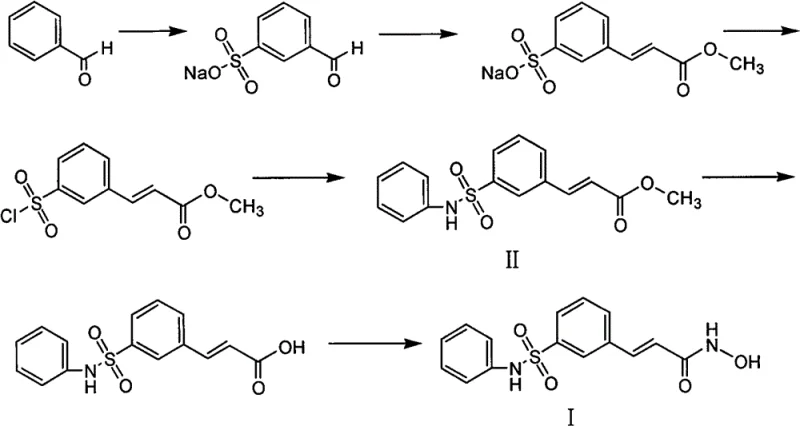
The Novel Approach
In stark contrast, the methodology disclosed in CN102786448B circumvents these pitfalls by adopting a bottom-up approach starting from m-carboxyl phenylsulfonic acid. This strategy effectively bypasses the need for direct sulfonation with oleum. The process initiates with a straightforward esterification to protect the carboxyl group, followed by a controlled chlorination to activate the sulfonyl group. The subsequent coupling with aniline is performed under mild conditions, avoiding the extreme temperatures and pressures often associated with older sulfonamide formation techniques. By converting the ester to an alcohol and then to an aldehyde before the final olefination, the route maintains high stereocontrol and purity. This novel approach not only mitigates the safety risks associated with explosive reagents but also streamlines the workflow, making it exceptionally suitable for reducing lead time for high-purity pharmaceutical intermediates in a GMP environment.
Mechanistic Insights into the Six-Step Synthetic Route
The core of this synthetic strategy relies on a precise sequence of functional group transformations that maximize yield while minimizing byproduct formation. The initial esterification of the sodium salt of m-carboxyl phenylsulfonic acid using methanol and hydrochloric acid is a thermodynamically favorable equilibrium that is easily driven to completion. Following this, the conversion to the sulfonyl chloride using phosphorus oxychloride (POCl3) is a critical activation step. POCl3 acts as a potent chlorinating agent, replacing the sulfonate oxygen with chlorine to create a highly electrophilic center. This activation is essential for the subsequent nucleophilic attack by aniline. The reaction conditions are optimized to use an excess of aniline, which serves a dual purpose as both the reactant and the acid scavenger, thereby simplifying the workup procedure and eliminating the need for additional base additives that could complicate downstream purification.
The latter half of the synthesis demonstrates sophisticated control over oxidation states. The reduction of the methyl ester to the primary alcohol using lithium chloride and potassium borohydride is a selective transformation that leaves the sulfonamide moiety intact. This is followed by a mild oxidation using Pyridinium Chlorochromate (PCC) adsorbed on silica gel. The use of supported PCC is a crucial mechanistic detail; it enhances oxidation efficiency and facilitates the removal of chromium residues, which are critical impurities in pharmaceutical manufacturing. Finally, the Horner-Wadsworth-Emmons (HWE) reaction with trimethyl phosphonoacetate installs the acrylic acid side chain with high (E)-selectivity. This stereoselectivity is vital for the biological activity of the final Belinostat molecule. The entire sequence is designed to ensure that each intermediate precipitates as a solid, allowing for rigorous purity checks via melting point and NMR before proceeding to the next step.
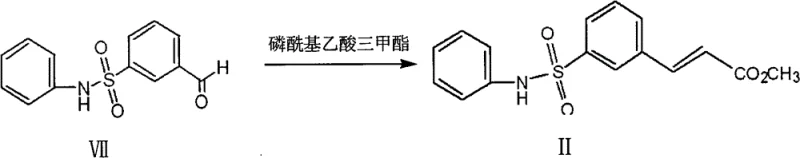
How to Synthesize Belinostat Intermediate II Efficiently
The synthesis of Belinostat Intermediate II via this patented route offers a standardized protocol that balances reaction kinetics with operational safety. The process begins with the protection of the carboxylic acid, followed by activation of the sulfonate, coupling, reduction, oxidation, and finally olefination. Each step has been optimized to use commercially available reagents and solvents like toluene and THF, ensuring that the process is not dependent on exotic or hard-to-source chemicals. The detailed标准化 synthesis steps见下方的指南 provide a roadmap for replicating this high-efficiency pathway in a pilot or production plant setting.
- Esterification of m-carboxyl phenylsulfonic acid using methanol and hydrochloric acid to form the methyl ester.
- Chlorination of the sulfonate salt using phosphorus oxychloride to generate the sulfonyl chloride.
- Coupling with aniline to form the sulfonamide bond, followed by reduction of the ester to an alcohol.
- Oxidation of the alcohol to an aldehyde using PCC on silica gel, followed by Horner-Wadsworth-Emmons olefination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the radical simplification of the supply chain risk profile. By eliminating the need for fuming sulfuric acid and sulfur dioxide gas, facilities can operate with lower insurance premiums and reduced regulatory burden. The reliance on solid intermediates that can be purified by recrystallization rather than chromatography significantly reduces solvent consumption and waste generation. This aligns perfectly with modern green chemistry initiatives and reduces the environmental footprint of the manufacturing process. Furthermore, the robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality material for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic implications of this route are profound. By avoiding expensive and hazardous reagents like oleum, the direct material costs are lowered. More importantly, the elimination of column chromatography—a process that is both solvent-intensive and labor-heavy—drastically reduces operating expenses. The ability to purify intermediates via simple filtration and recrystallization translates to faster cycle times and lower utility costs. Additionally, the use of aniline as both reactant and acid binder simplifies the bill of materials, further contributing to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals. Reagents such as methanol, phosphorus oxychloride, and aniline are produced on a massive global scale, ensuring that supply disruptions are unlikely. In contrast, specialized sulfonating agents or diazotization kits can have longer lead times and are subject to stricter transportation regulations. The solid nature of the intermediates also simplifies storage and logistics; solids are generally more stable and easier to transport than sensitive liquid reagents or gases, thereby enhancing the reliability of inventory management and distribution networks.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues, but this route is inherently designed for scale. The exothermic risks associated with oleum sulfonation are completely removed. The waste stream is significantly cleaner, devoid of heavy metal contaminants from chromatography or acidic gases from sulfonation. This makes the permitting process for new manufacturing lines smoother and ensures long-term compliance with increasingly stringent environmental regulations. The process is thus future-proofed against regulatory tightening, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Belinostat intermediates. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for R&D and procurement professionals evaluating this technology for potential integration into their supply chains.
Q: Why is this new synthesis route safer than conventional methods?
A: Conventional routes often utilize hazardous reagents like fuming sulfuric acid (oleum) or gaseous sulfur dioxide and diazotization conditions. This novel method avoids these dangerous chemicals by starting from stable sulfonic acid salts and using standard chlorination and coupling reagents, significantly reducing explosion risks and corrosion hazards.
Q: How does this process improve purification and yield?
A: The process is designed so that intermediates in every step are solids. This allows for purification via simple recrystallization rather than labor-intensive column chromatography. This not only improves operational convenience but also ensures high purity suitable for industrial scale-up.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method utilizes readily available and inexpensive reagents. The avoidance of hazardous gases and the use of solid intermediates that are easy to handle and detect make the process highly robust and scalable for ton-level manufacturing of Belinostat intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Belinostat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the commercial scale-up of complex pharmaceutical intermediates, leveraging advanced process chemistry to optimize yields and purity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Belinostat intermediate adheres to the highest international standards.
We invite you to collaborate with us to unlock the full potential of this innovative synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and reliability in your Belinostat supply chain.
