Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Introduction to Novel Triazole Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine motifs which are critical for metabolic stability and bioavailability. A significant breakthrough in this domain is documented in Chinese Patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the growing demand for reliable pharmaceutical intermediate suppliers by offering a route that utilizes the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source. The strategic importance of this scaffold cannot be overstated, as evidenced by its presence in high-value therapeutic agents such as Sitagliptin and various Factor IXa inhibitors shown in the structural overview below.
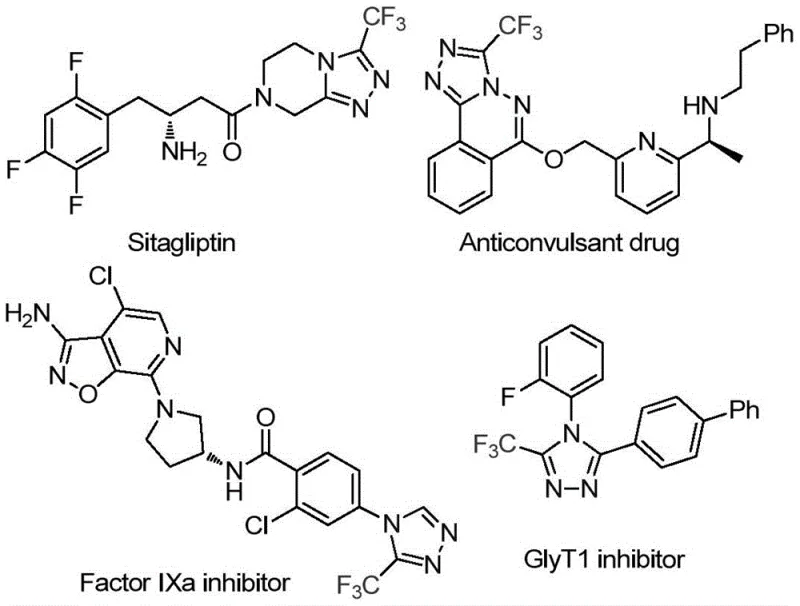
This patent represents a paradigm shift in how we approach the commercial scale-up of complex pharmaceutical intermediates. By leveraging a tandem cyclization reaction promoted by molecular iodine, the process achieves high reaction efficiency without the stringent requirement for anhydrous or anaerobic conditions. For R&D directors and process chemists, this implies a drastic reduction in development time and risk, as the method tolerates ambient atmospheric conditions while maintaining high purity standards. The ability to synthesize these valuable heterocycles using readily available starting materials positions this technology as a cornerstone for cost reduction in API manufacturing, ensuring a stable supply chain for downstream drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazole rings bearing trifluoromethyl groups has been fraught with synthetic challenges that hinder large-scale adoption. Traditional pathways often rely on multi-step sequences involving hazardous reagents, expensive transition metal catalysts, or specialized C1 building blocks that are difficult to source in bulk quantities. Many existing protocols necessitate rigorous exclusion of moisture and oxygen, requiring specialized equipment such as gloveboxes or Schlenk lines, which significantly inflates capital expenditure and operational complexity. Furthermore, conventional methods frequently suffer from narrow substrate scope, where the presence of sensitive functional groups on the aromatic ring can lead to decomposition or low yields, thereby limiting the versatility of the synthesis for diverse drug discovery programs. These factors collectively contribute to extended lead times and elevated production costs, creating bottlenecks for procurement managers seeking efficient supply solutions.
The Novel Approach
In stark contrast, the methodology described in patent CN114920707B introduces a streamlined, atom-economical approach that elegantly bypasses these historical hurdles. The core innovation lies in the dual functionality of DMF, which acts simultaneously as the polar aprotic solvent and the donor of the carbon atom required for ring closure. This eliminates the need for external, costly carbon sources and simplifies the reaction mixture significantly. The use of molecular iodine as a promoter facilitates the activation of DMF under relatively mild thermal conditions (110-130°C), enabling the transformation to proceed smoothly in the presence of air. This operational simplicity is a game-changer for industrial applications, as it removes the need for inert gas purging and allows for the use of standard reactor vessels. The result is a robust process capable of accommodating a wide range of substituents on the hydrazide starting material, thereby enhancing the practicability of the method for generating diverse libraries of bioactive molecules.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the interaction between molecular iodine and DMF, which activates the solvent towards nucleophilic attack. Depending on the specific pathway taken, either the formyl group or the N-methyl group of DMF serves as the carbon synthon. When the formyl group participates, it undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization and elimination of dimethylamine to yield the target triazole. Alternatively, when the N-methyl group is utilized, DMF reacts with iodine to generate an amine salt species, which then engages in nucleophilic addition with the hydrazide. This pathway involves the elimination of N-methylformamide to generate an azadiene intermediate, which subsequently undergoes oxidative aromatization to furnish the final product. This mechanistic flexibility ensures high conversion rates across different substrate classes.
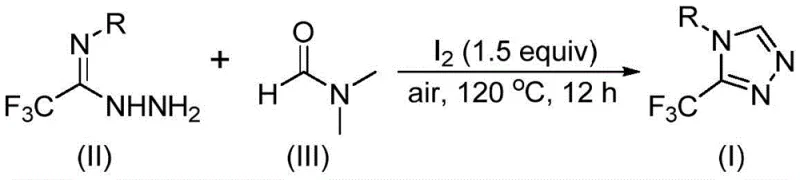
From an impurity control perspective, the mechanism offers distinct advantages. The use of iodine as a mild oxidant and promoter helps drive the reaction to completion, minimizing the accumulation of partially cyclized intermediates that often plague triazole syntheses. The elimination byproducts, such as dimethylamine or N-methylformamide, are volatile or easily separable during the workup phase, facilitating the isolation of high-purity products. The patent data indicates that the reaction is highly selective, with minimal formation of side products even when using substrates with electron-withdrawing groups like trifluoromethyl or halogens. This selectivity is vital for meeting the stringent purity specifications required for pharmaceutical intermediates, reducing the burden on downstream purification steps such as recrystallization or chromatography. The detailed understanding of this catalytic cycle allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and minimize waste generation.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and safety. The protocol is designed to be user-friendly, utilizing standard glassware and heating equipment without the need for specialized inert atmosphere setups. The key to success lies in the precise control of reaction temperature and the stoichiometric ratio of the iodine promoter relative to the hydrazide substrate. While the reaction is robust, optimizing the workup procedure—specifically the filtration and silica gel treatment steps—is essential for removing residual iodine and DMF byproducts to achieve the desired quality standards. The following guide outlines the standardized steps derived from the patent examples to assist technical teams in replicating this high-efficiency transformation.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel under air atmosphere.
- Heat the reaction mixture to 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Perform post-treatment via filtration, washing, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis strategy offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of value is the substantial cost savings achieved through reagent simplification. By utilizing DMF, a commodity chemical available in massive volumes globally, as both solvent and reactant, the process eliminates the need for purchasing expensive, specialized C1 donors or exotic catalysts. This consolidation of materials significantly reduces the raw material bill of goods (COGS) and simplifies inventory management, as fewer distinct SKUs need to be sourced and stored. Furthermore, the avoidance of precious metal catalysts removes the complex and costly requirement for heavy metal scavenging and validation, which is a major regulatory hurdle in API manufacturing. This streamlining translates directly into lower production costs and faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the use of inexpensive, bulk-available reagents. Molecular iodine is significantly cheaper than palladium, copper, or rhodium catalysts often used in heterocycle synthesis, and it does not leave behind toxic metal residues that require expensive removal processes. Additionally, the reaction operates under air, negating the energy and infrastructure costs associated with maintaining nitrogen or argon atmospheres. The high reaction efficiency reported in the patent examples suggests that less starting material is wasted, improving the overall mass balance and yield per batch. These factors combine to create a highly cost-effective manufacturing route that enhances profit margins for high-volume production runs without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on universally available starting materials. DMF and molecular iodine are produced by numerous chemical manufacturers worldwide, reducing the risk of supply disruptions caused by single-source dependencies. The trifluoroethyliminohydrazide precursors are also described as easily accessible or rapidly synthesizable from common aromatic amines, ensuring a steady flow of inputs. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert gas systems or moisture control. This reliability allows for more accurate forecasting and planning, ensuring that downstream API synthesis schedules are met consistently, which is critical for maintaining continuous drug supply to patients.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is straightforward due to the absence of sensitive reagents and the use of a high-boiling solvent that facilitates heat transfer. The operational simplicity reduces the training burden on plant operators and minimizes the risk of human error during batch execution. From an environmental standpoint, the atom economy is improved by incorporating the solvent into the product, reducing the volume of chemical waste generated per unit of product. While DMF itself requires careful handling and disposal, the elimination of heavy metal catalysts and the reduction in auxiliary reagents contribute to a greener chemical profile. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory compliance regarding waste discharge and environmental impact assessments.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the detailed experimental data and background information provided in the patent documentation. These answers address common concerns regarding reaction scope, operational safety, and product quality, providing a quick reference for technical decision-makers evaluating this synthesis route for their specific projects. Understanding these nuances is essential for effectively integrating this method into existing manufacturing workflows.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or methyl group), which drastically simplifies the reagent list and reduces raw material costs compared to traditional methods requiring separate C1 donors.
Q: Does this process require inert gas protection?
A: No, the patent explicitly states that the reaction proceeds efficiently under an air atmosphere, eliminating the need for expensive nitrogen or argon shielding and simplifying operational requirements for large-scale production.
Q: What is the substrate scope for the R group?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like methoxy and methylthio, as well as electron-withdrawing groups like fluoro, chloro, and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN114920707B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt this iodine-promoted cyclization for large-scale manufacturing ensures that our clients receive a consistent, reliable supply of material that accelerates their drug development timelines.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can benefit from a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this DMF-based method. We encourage you to contact us today to request specific COA data for our available triazole intermediates and to schedule a consultation regarding route feasibility assessments for your target molecules. Together, we can drive down costs and enhance the efficiency of your pharmaceutical supply chain.
