Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough for Commercial Scale-Up of Complex Heterocycles
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methods for constructing nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A significant advancement in this domain is detailed in Chinese Patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source in an iodine-promoted tandem cyclization. For R&D directors and process chemists, this approach offers a compelling alternative to traditional multi-step syntheses that often rely on hazardous reagents or complex catalytic systems. The ability to generate these valuable cores under aerobic conditions using commercially available starting materials positions this methodology as a cornerstone for the next generation of high-purity pharmaceutical intermediates.
The structural versatility of the 1,2,4-triazole ring is evident in its presence within numerous bioactive molecules, ranging from anticonvulsants to enzyme inhibitors like Factor IXa and GlyT1 inhibitors. The introduction of a trifluoromethyl group at the 3-position further amplifies the pharmacological potential of these scaffolds by modulating lipophilicity and electronic properties. As illustrated in the broader context of drug discovery, the demand for reliable access to these fluorinated heterocycles is escalating. The patented method addresses this demand by providing a direct route that bypasses the logistical hurdles associated with specialized C1 synthons, thereby streamlining the supply chain for complex heterocycles used in modern medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that impede large-scale manufacturing. Conventional routes often necessitate the use of expensive transition metal catalysts, such as palladium or copper complexes, which introduce significant cost burdens and complicate downstream purification due to stringent limits on residual metal content in final drug substances. Furthermore, many established protocols require rigorously anhydrous and anaerobic conditions to prevent catalyst deactivation or side reactions, demanding specialized equipment like gloveboxes or Schlenk lines that are difficult to justify in a production setting. The reliance on distinct, often unstable carbon sources for ring closure adds another layer of complexity, increasing the raw material inventory and safety risks associated with handling reactive intermediates. These factors collectively contribute to prolonged lead times and elevated manufacturing costs, creating a bottleneck for the commercial scale-up of complex polymer additives and pharmaceutical ingredients alike.
The Novel Approach
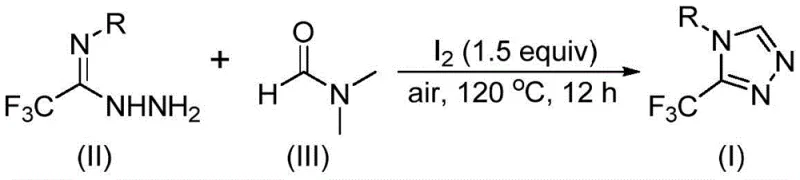
In stark contrast, the methodology described in CN114920707B leverages the intrinsic reactivity of DMF to drive the cyclization process, effectively turning a common waste stream component into a valuable reactant. By employing molecular iodine as a mild promoter, the reaction activates the DMF molecule to participate directly in the ring formation, eliminating the need for external carbon donors. This innovation drastically simplifies the reaction setup, allowing the transformation to proceed in an open vessel under air, which is a rare feat for such intricate heterocycle formations. The operational simplicity extends to the workup procedure, which involves standard filtration and chromatography, avoiding the quenching steps often required for sensitive organometallic reagents. This streamlined workflow not only enhances safety but also aligns perfectly with the principles of green chemistry, offering a sustainable pathway for cost reduction in API manufacturing.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and impurity control. The reaction proceeds through a fascinating dual-pathway mechanism depending on which fragment of the DMF molecule acts as the carbon source. In the pathway where the formyl group serves as the carbon donor, the process initiates with a condensation reaction between the formyl moiety of DMF and the hydrazide nitrogen of the trifluoroethyliminohydrazide substrate. This generates a hydrazone intermediate which subsequently undergoes intramolecular cyclization. The final step in this sequence involves the elimination of a dimethylamine molecule, driving the equilibrium towards the formation of the aromatic 3-trifluoromethyl-1,2,4-triazole product. This pathway highlights the electrophilic nature of the DMF carbonyl carbon when activated by the reaction conditions.
Alternatively, when the N-methyl group of DMF acts as the carbon source, the mechanism involves an initial activation of the solvent by molecular iodine. This interaction generates an amine salt species, which, upon elimination of hydrogen iodide, becomes susceptible to nucleophilic attack by the hydrazide. The resulting adduct eliminates a molecule of N-methylformamide to yield a reactive azadiene intermediate. This intermediate then undergoes a subsequent intramolecular nucleophilic addition followed by oxidative aromatization to furnish the target triazole. The ability of the system to toggle between these pathways ensures high conversion rates across a diverse range of substrates. From a quality control perspective, the mild oxidative conditions minimize the formation of over-oxidized byproducts, ensuring that the high-purity 1,2,4-triazole derivatives obtained meet the rigorous standards required for pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis is designed for practicality, utilizing standard laboratory glassware and readily available reagents to facilitate easy adoption in both research and production environments. The protocol begins by combining the trifluoroethyliminohydrazide precursor with molecular iodine in a substantial excess of DMF, which serves to dissolve the reactants and drive the equilibrium forward. The mixture is then heated to a moderate temperature range, typically between 110°C and 130°C, and maintained under an air atmosphere for a period of 10 to 15 hours. This thermal energy is sufficient to overcome the activation barrier for the tandem cyclization without degrading the sensitive trifluoromethyl group. Following the reaction period, the crude mixture is subjected to a straightforward workup involving filtration to remove inorganic salts, followed by silica gel chromatography to isolate the pure product. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reaction vessel with molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate, and excess DMF solvent under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for a duration of 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, drying, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the economic implications of this patented technology are profound. By replacing expensive, specialized reagents with commodity chemicals like DMF and iodine, the direct material costs associated with producing these intermediates are significantly reduced. The elimination of precious metal catalysts removes the need for costly scavenging resins and extensive analytical testing for metal residues, further lowering the overall cost of goods sold. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or environmental factors, leading to more consistent batch-to-batch yields and reduced waste generation. This reliability translates directly into a more resilient supply chain capable of meeting fluctuating market demands without the risk of production stoppages.
- Cost Reduction in Manufacturing: The substitution of high-cost transition metal catalysts with inexpensive molecular iodine results in substantial savings on raw material expenditures. Additionally, the dual function of DMF as both solvent and reactant reduces the total volume of chemicals required, minimizing waste disposal costs and simplifying the bill of materials. The avoidance of cryogenic conditions or high-pressure equipment further lowers capital expenditure and utility costs, making this route economically superior to traditional methods for large-scale production.
- Enhanced Supply Chain Reliability: Since the key starting materials, including DMF and iodine, are globally available commodity chemicals, the risk of supply disruption is minimized. The trifluoroethyliminohydrazide precursors can be synthesized from widely accessible aromatic amines and trifluoroacetic acid derivatives, ensuring a stable upstream supply. This accessibility allows manufacturers to source materials from multiple vendors, fostering competition and securing better pricing terms while guaranteeing continuity of supply for critical pharmaceutical projects.
- Scalability and Environmental Compliance: The operation under air atmosphere and the use of non-volatile, high-boiling solvents facilitate safe scale-up from gram to ton quantities without the need for specialized inert gas infrastructure. The simplified workup procedure reduces the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate sustainability metrics of the manufacturing entity, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or methyl group), which eliminates the need for separate, expensive C1 building blocks and simplifies the reaction setup significantly.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key innovations of patent CN114920707B is that the reaction proceeds efficiently under an air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly reducing operational complexity.
Q: What is the substrate scope for the R group in this synthesis?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with electron-donating groups like ethyl and methoxy, as well as electron-withdrawing groups like fluoro, chloro, and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications for all our intermediates, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel triazole derivatives or bulk supply of established intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and bring your innovative therapies to market faster and more cost-effectively.
