Scalable Manufacturing of Benzacridine Derivatives for Advanced Oncology Drug Development
Scalable Manufacturing of Benzacridine Derivatives for Advanced Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that can effectively target oncogenic drivers with high specificity and low toxicity. Patent CN102351870B introduces a significant advancement in this domain by disclosing a comprehensive method for preparing benzacridine derivatives and their application as anticancer agents. These compounds are designed to interact strongly with the c-myc proto-oncogene DNA, a critical target in cancer therapy due to its role in cell proliferation and apoptosis regulation. The disclosed technology provides a robust synthetic pathway that transforms simple aromatic precursors into complex heterocyclic systems capable of intercalating with DNA. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for evaluating its potential as a reliable pharmaceutical intermediate supplier solution. The patent outlines a versatile chemistry that allows for substantial structural modification at multiple positions, enabling the fine-tuning of biological activity and physicochemical properties to meet stringent drug development criteria.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of acridine-based alkaloids has relied heavily on extraction from natural sources or convoluted multi-step syntheses that suffer from poor atom economy and harsh reaction conditions. Natural extraction is inherently limited by the low abundance of source materials and the difficulty in separating closely related analogs, leading to inconsistent batch quality and supply chain volatility. Furthermore, classical synthetic routes often involve the use of expensive transition metal catalysts or hazardous reagents that generate significant waste streams, complicating environmental compliance and increasing the overall cost of goods. Many existing methods struggle to introduce diverse functional groups at specific positions on the acridine ring without compromising the integrity of the core structure, limiting the ability of medicinal chemists to explore structure-activity relationships effectively. These bottlenecks often result in prolonged development timelines and inflated costs, making it challenging to bring promising anticancer candidates to clinical trials in a timely manner.
The Novel Approach
The methodology presented in the patent offers a streamlined and highly efficient alternative that addresses these historical challenges through a rational design of the synthetic route. By utilizing a convergent strategy that builds the benzacridine core from readily available starting materials like 1,2-dimethoxybenzene and succinic anhydride, the process ensures a consistent and scalable supply of key intermediates. The route employs well-established reactions such as Friedel-Crafts acylation and intramolecular cyclization, which are known for their reliability and ease of scale-up in industrial settings. A key innovation lies in the late-stage functionalization capabilities, where the introduction of various amine side chains allows for the rapid generation of a diverse library of derivatives without the need to restart the synthesis from the beginning. This modularity significantly accelerates the lead optimization process, providing a distinct advantage for cost reduction in pharmaceutical intermediate manufacturing.
This strategic approach not only simplifies the production workflow but also enhances the purity profile of the final active pharmaceutical ingredients by minimizing the formation of difficult-to-remove byproducts. The use of common solvents and reagents further reduces the dependency on specialized supply chains, mitigating risks associated with raw material shortages. Consequently, this novel approach represents a paradigm shift towards more sustainable and economically viable production of complex anticancer intermediates.
Mechanistic Insights into POCl3-Mediated Cyclization and Functionalization
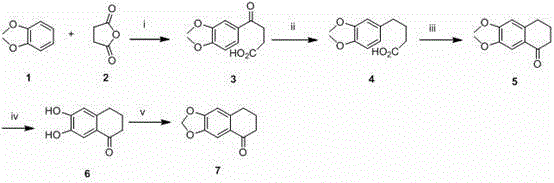
The core of this synthetic technology revolves around a sophisticated sequence of transformations that construct the tetracyclic benzacridine framework with high precision. The process initiates with a Friedel-Crafts acylation where aluminum chloride acts as a Lewis acid catalyst to facilitate the attachment of a succinyl chain to the veratrole ring, forming a keto-acid intermediate. This is followed by a reduction step using triethylsilane and trifluoroacetic acid, which selectively reduces the ketone to a methylene group while preserving other sensitive functionalities. Subsequent cyclization using polyphosphoric acid closes the second ring to form a tetralone derivative, establishing the rigid backbone required for DNA intercalation. The critical step in the formation of the acridine nitrogen heterocycle involves the condensation of the tetralone intermediate with an aniline derivative in the presence of phosphorus oxychloride under reflux conditions. This dehydration cyclization is highly efficient and drives the formation of the chloro-substituted benzacridine core, which serves as a pivotal handle for further diversification.
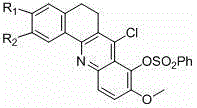
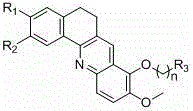
Following the construction of the core, the mechanism allows for precise control over the substitution pattern through nucleophilic aromatic substitution or quaternization reactions. The chlorine atom at the specific position on the acridine ring is highly susceptible to displacement by various amines, enabling the installation of solubilizing groups or pharmacophores that enhance cellular uptake. Additionally, the methoxy groups on the aromatic rings can be demethylated to phenols and subsequently alkylated with dibromoalkanes to introduce linker chains. This level of mechanistic control ensures that impurities arising from non-specific reactions are minimized, resulting in a cleaner crude product that requires less intensive purification. For quality assurance teams, this predictable reactivity profile translates to more robust analytical methods and tighter specifications for the final drug substance.
How to Synthesize Benzacridine Derivatives Efficiently
Executing this synthesis requires careful attention to reaction parameters such as temperature, stoichiometry, and solvent selection to maximize yield and purity. The initial acylation must be conducted at controlled low temperatures to prevent poly-acylation, followed by a carefully managed workup to recover the aluminum salts. The cyclization steps demand anhydrous conditions and rigorous exclusion of moisture to ensure the effectiveness of the dehydrating agents like polyphosphoric acid and phosphorus oxychloride. Detailed standardized operating procedures are essential to maintain consistency across different production batches, particularly during the final amine substitution where reaction kinetics can vary based on the steric bulk of the amine. The detailed standardized synthesis steps are outlined below to guide process development teams in replicating these results.
- Perform Friedel-Crafts acylation of 1,2-dimethoxybenzene with succinic anhydride using AlCl3 catalyst.
- Execute reduction and intramolecular cyclization using polyphosphoric acid to form the tetralone core.
- Conduct final cyclization with phosphorus oxychloride and subsequent amine substitution to finalize the benzacridine structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. The reliance on commodity chemicals rather than exotic reagents means that the cost of raw materials is inherently lower and less susceptible to market fluctuations. This stability is crucial for long-term supply agreements and helps in forecasting production budgets with greater accuracy. Furthermore, the elimination of complex chiral resolution steps or rare earth catalysts simplifies the manufacturing process, reducing the capital expenditure required for specialized equipment and waste treatment facilities. The high yields reported in the patent embodiments suggest that the process is materially efficient, minimizing the loss of valuable intermediates and maximizing the output per unit of input.
- Cost Reduction in Manufacturing: The synthetic pathway leverages inexpensive and abundant starting materials such as veratrole and succinic anhydride, which are produced on a massive industrial scale globally. By avoiding the use of precious metal catalysts like palladium in the main backbone construction (using iron powder for reduction instead), the process significantly lowers the cost of goods sold. The straightforward purification methods, primarily involving crystallization and standard column chromatography, reduce the consumption of expensive silica gel and solvents. This economic efficiency allows for competitive pricing strategies when sourcing these intermediates for preclinical and clinical supply.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, such as the chloro-benzacridine core, which can be rapidly converted into various final derivatives based on demand. This flexibility mitigates the risk of supply disruptions caused by changes in specific drug candidate requirements. Since the reagents are non-hazardous and widely traded, procurement teams can easily qualify multiple vendors for each raw material, ensuring a redundant and secure supply chain. The robustness of the chemistry also means that technology transfer between different manufacturing sites can be accomplished with minimal friction.
- Scalability and Environmental Compliance: The reactions described operate under conditions that are amenable to large-scale batch processing, with temperatures and pressures that do not require specialized high-pressure reactors. The waste streams generated are primarily acidic or organic in nature, which can be treated using standard effluent treatment protocols common in fine chemical plants. By designing a route that minimizes the number of isolation steps and uses recyclable solvents where possible, the process aligns with modern green chemistry principles. This alignment facilitates smoother regulatory approvals and reduces the environmental footprint associated with the production of anticancer medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzacridine derivatives. Understanding these aspects is critical for stakeholders evaluating the feasibility of incorporating this technology into their drug development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What is the primary biological target of these benzacridine derivatives?
A: According to patent CN102351870B, these derivatives exhibit strong inhibitory effects on the expression of the c-myc proto-oncogene DNA, making them potent candidates for anticancer therapy.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes widely available commodity chemicals such as 1,2-dimethoxybenzene (veratrole), succinic anhydride, and o-vanillin, ensuring a stable supply chain for large-scale production.
Q: How does this method improve upon traditional alkaloid extraction?
A: This synthetic route offers superior control over impurity profiles and stereochemistry compared to natural extraction, while allowing for structural diversification to optimize pharmacological properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzacridine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of benzacridine derivative meets the highest standards required for oncology research. Our commitment to quality extends beyond mere compliance; we actively engage in process optimization to enhance yields and reduce impurities, delivering value at every stage of the supply chain.
We invite you to collaborate with us to unlock the full potential of this anticancer technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us be your partner in advancing novel cancer therapies from the laboratory to the clinic.
