Advancing Pharmaceutical Intermediate Synthesis via Efficient Cobalt-Catalyzed C-H Functionalization
The landscape of organic synthetic chemistry is constantly evolving to meet the rigorous demands of the pharmaceutical industry, particularly in the construction of complex heterocyclic scaffolds. Patent CN109721590B introduces a significant breakthrough in the synthesis of C2 alpha acyloxy indoles, a structural motif prevalent in bioactive molecules exhibiting antiviral activities against HIV, HPV, and influenza. This technology leverages a cobalt-catalyzed direct C-H functionalization strategy, utilizing tert-butyl peroxyesters as both oxidants and acyloxy sources. For global procurement and R&D teams, this represents a pivotal shift towards more sustainable and cost-effective manufacturing routes for high-value pharmaceutical intermediates. The methodology addresses critical pain points in traditional synthesis, such as the reliance on expensive noble metal catalysts and harsh oxidizing conditions, offering a streamlined pathway that aligns with modern green chemistry principles while maintaining high yields and purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct C-H functionalization at the C2 alpha position of indoles has been fraught with significant chemical and economic challenges. Prior art methods, such as those utilizing TBAI (tetrabutylammonium iodide) with PhI(OAc)2 or palladium-catalyzed systems, suffer from severe substrate limitations, often restricted to 2,3-disubstituted indoles. Furthermore, these conventional routes frequently struggle with regioselectivity, leading to mixtures of C2 and C3 functionalized products that require difficult and yield-reducing separation processes. The reliance on hypervalent iodine reagents not only drives up raw material costs but also generates stoichiometric amounts of iodobenzene waste, complicating downstream purification and creating environmental disposal burdens. These factors collectively hinder the scalability and commercial viability of producing C2 alpha-functionalized indoles for large-scale API manufacturing.
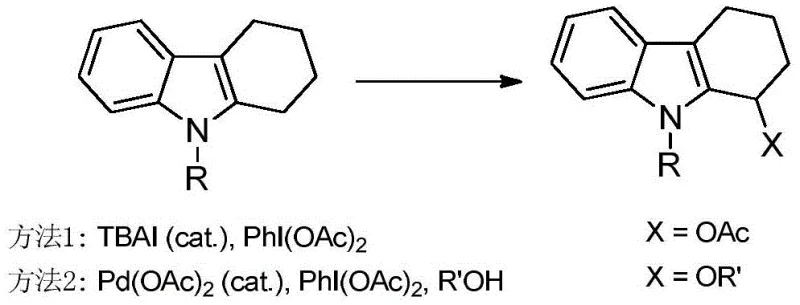
The Novel Approach
In stark contrast to these legacy technologies, the invention disclosed in CN109721590B presents a robust and versatile cobalt-catalyzed protocol that fundamentally reshapes the synthetic landscape. By employing inexpensive Co(II) salts such as Co(OAc)2 or Co(acac)2, the process achieves efficient acyloxylation of 2-substituted indoles under mild thermal conditions ranging from 50 to 120 degrees Celsius. The use of tert-butyl peroxyesters serves a dual purpose, acting simultaneously as the terminal oxidant and the source of the acyloxy group, which dramatically simplifies the reagent profile. This approach expands the substrate scope significantly, tolerating a wide array of electronic and steric environments including halogens, esters, and heterocycles, thereby enabling the synthesis of diverse libraries of indole derivatives essential for drug discovery and development.
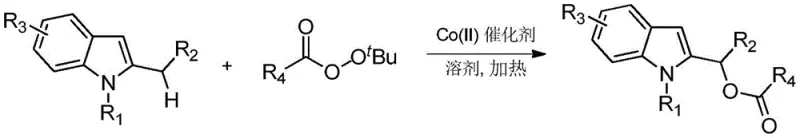
Mechanistic Insights into Co(II)-Catalyzed C-H Acyloxylation
The mechanistic elegance of this transformation lies in the redox versatility of the cobalt center, which facilitates a radical-mediated pathway distinct from traditional two-electron organometallic cycles. The reaction is initiated by the homolytic cleavage of the weak O-O bond in the tert-butyl peroxyester, potentially assisted by the Co(II) species, generating an acyloxy radical and a tert-butoxy radical. These reactive intermediates engage in a selective hydrogen atom transfer (HAT) process at the benzylic-like C2-alpha position of the indole substrate. The resulting carbon-centered radical is subsequently trapped by another equivalent of the acyloxy radical or undergoes oxidation by the cobalt species to form a cationic intermediate, which is finally quenched to yield the desired C2 alpha acyloxy indole product. This radical manifold bypasses the need for pre-functionalized directing groups, offering a step-economical advantage that is highly attractive for process chemistry.
From an impurity control perspective, the regiospecificity of this cobalt-catalyzed system is paramount for ensuring high product quality. Unlike electrophilic aromatic substitution which might favor the electron-rich C3 position, the radical mechanism described here is governed by bond dissociation energies and steric accessibility, inherently favoring the C2-alpha methylene or methine protons. This intrinsic selectivity minimizes the formation of regioisomeric byproducts, simplifying the purification workflow and reducing the burden on analytical quality control labs. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups often present in complex drug candidates, ensuring that the final intermediate retains the integrity required for subsequent coupling reactions in the API synthesis sequence.
How to Synthesize C2 Alpha Acyloxy Indole Efficiently
The practical implementation of this cobalt-catalyzed methodology is designed for ease of operation in standard laboratory and pilot plant settings. The procedure typically involves charging a reaction vessel with the 2-substituted indole substrate, a suitable organic solvent such as toluene or 1,2-dichloroethane, the tert-butyl peroxyester oxidant, and a catalytic amount of the cobalt salt. The mixture is then heated under stirring for a defined period, allowing the catalytic cycle to turnover efficiently. Following the reaction, a simple workup involving filtration and solvent removal followed by column chromatography yields the pure product. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with 2-substituted indole derivative, organic solvent, tert-butyl peroxyester, and Co(II) catalyst under controlled molar ratios.
- Heat the mixture in an oil bath at temperatures between 50-120°C for 5-20 hours to facilitate the oxidative acyloxylation reaction.
- Upon completion, dilute with ethyl acetate, filter, concentrate under reduced pressure, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from precious metal catalysts like palladium to earth-abundant cobalt represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for expensive hypervalent iodine reagents and reducing the complexity of waste treatment associated with heavy metal residues, the overall cost of goods sold (COGS) can be substantially lowered. Additionally, the simplified reagent profile enhances supply chain resilience, as the key materials are commodity chemicals with stable global availability, reducing the risk of production delays due to raw material shortages.
- Cost Reduction in Manufacturing: The replacement of costly palladium catalysts and stoichiometric hypervalent iodine oxidants with inexpensive cobalt salts and peroxyesters drives down raw material expenses significantly. The elimination of iodobenzene byproducts reduces the load on purification steps, leading to higher overall process efficiency and lower solvent consumption. This streamlined approach minimizes the number of unit operations required, directly translating to reduced utility costs and labor hours per kilogram of product produced.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-strategic raw materials ensures a robust and continuous supply chain for high-purity pharmaceutical intermediates. Unlike processes dependent on scarce noble metals which are subject to geopolitical price volatility, cobalt and organic peroxides are sourced from stable industrial supply chains. This stability allows for more accurate long-term planning and inventory management, ensuring that critical API production schedules are met without interruption.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metal waste streams facilitate easier scale-up from gram to multi-ton quantities while adhering to strict environmental regulations. The process generates fewer hazardous byproducts, simplifying effluent treatment and lowering the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis route. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional iodine-based oxidation?
A: The cobalt-catalyzed method eliminates the need for expensive hypervalent iodine reagents like PhI(OAc)2, thereby avoiding the generation of iodobenzene byproducts which complicate purification. It also utilizes cheaper earth-abundant cobalt catalysts instead of precious metals like palladium.
Q: Does this process offer better regioselectivity for C2-alpha functionalization?
A: Yes, the method demonstrates high regiospecificity for the C2-alpha position on 2-substituted indoles, overcoming the selectivity issues often seen in C2 vs C3 functionalization in prior art methods.
Q: What types of substituents are tolerated on the indole ring?
A: The process exhibits excellent functional group tolerance, accommodating various groups such as halogens (F, Cl, Br, I), alkyls, esters, cyano, and heterocycles like pyrimidine on the indole scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2 Alpha Acyloxy Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed technology in accelerating the development of next-generation antiviral therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of C2 alpha acyloxy indole intermediate delivered meets the highest international quality standards required by regulatory bodies.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By leveraging our expertise in process optimization and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
