Advanced Cobalt-Catalyzed Synthesis of C2 Alpha Acyloxy Indoles for Commercial Pharmaceutical Production
Advanced Cobalt-Catalyzed Synthesis of C2 Alpha Acyloxy Indoles for Commercial Pharmaceutical Production
The synthesis of functionalized indole derivatives represents a cornerstone in modern medicinal chemistry, particularly for the development of antiviral agents targeting HIV, HPV, and influenza. Patent CN109721590B introduces a transformative methodology for preparing C2 alpha acyloxy indoles using a cost-effective cobalt catalytic system. This innovation addresses critical bottlenecks in the manufacturing of high-purity pharmaceutical intermediates by replacing expensive noble metal catalysts with abundant earth-abundant cobalt. For R&D directors and procurement strategists, this shift signifies a move towards more sustainable and economically viable supply chains for complex heterocyclic scaffolds. The technology leverages tert-butyl peroxyesters not only as oxidants but also as the acyloxy source, streamlining the reaction stoichiometry and minimizing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct C-H functionalization at the C2 alpha position of indoles has been plagued by significant synthetic challenges and economic inefficiencies. Traditional approaches often rely on hypervalent iodine reagents, such as PhI(OAc)2, in conjunction with catalysts like TBAI or Palladium(II) acetate. As illustrated in the comparative reaction schemes, these legacy methods suffer from severe substrate limitations, typically restricted to 2,3-disubstituted indoles, thereby excluding a vast array of simpler 2-substituted precursors. Furthermore, the use of iodobenzene diacetate generates stoichiometric amounts of iodobenzene byproducts, which are notoriously difficult to separate from the desired product, complicating downstream purification and increasing solvent consumption. The reliance on palladium also introduces substantial cost volatility and potential heavy metal contamination risks, which are unacceptable for GMP-grade API intermediate production.
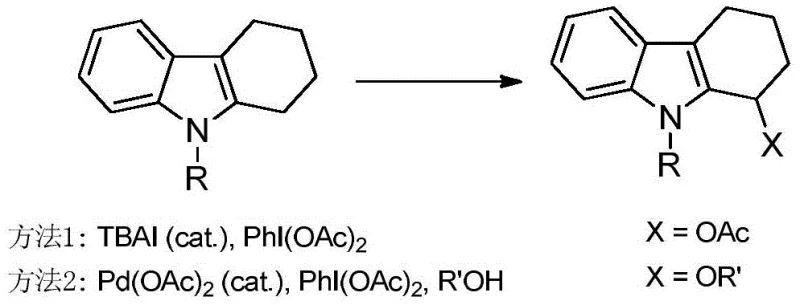
The Novel Approach
In stark contrast, the disclosed cobalt-catalyzed protocol offers a robust and versatile solution that overcomes these historical barriers. By utilizing Co(II) salts such as Co(acac)2 or Co(OAc)2 alongside tert-butyl peroxyesters, the reaction achieves high yields and exceptional regioselectivity for 2-substituted indoles. This method effectively bypasses the need for pre-functionalization, adhering to the principles of atom economy and step efficiency. The reaction conditions are remarkably mild, operating effectively between 50°C and 120°C in common organic solvents like toluene or dichloroethane. This flexibility allows for the tolerance of diverse functional groups, including halogens, esters, and heterocycles, enabling the rapid synthesis of a broad library of derivatives without the need for protecting group strategies. The elimination of iodobenzene waste and expensive palladium catalysts fundamentally alters the cost structure of producing these valuable building blocks.
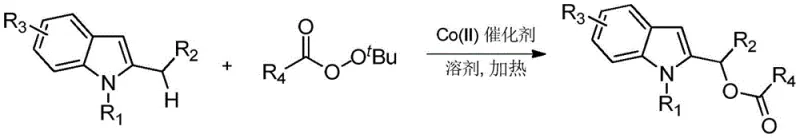
Mechanistic Insights into Cobalt-Catalyzed C-H Acyloxylation
The mechanistic pathway of this transformation likely involves a radical-mediated process initiated by the homolytic cleavage of the peroxide bond facilitated by the cobalt center. The Co(II) species activates the tert-butyl peroxyester to generate an acyloxy radical, which subsequently attacks the electron-rich indole ring. The unique electronic environment of the 2-substituted indole directs this attack specifically to the C2 alpha position, ensuring the observed regiospecificity. This radical mechanism differs significantly from the electrophilic substitution pathways often seen in iodine-mediated reactions, explaining the broader substrate scope and milder conditions. Understanding this radical manifold is crucial for process chemists aiming to optimize reaction parameters for large-scale batches, as it dictates the sensitivity to oxygen and the choice of solvent to stabilize radical intermediates.
From an impurity control perspective, the simplicity of the catalytic cycle minimizes the formation of complex side products. Unlike palladium-catalyzed cross-couplings which can suffer from beta-hydride elimination or homocoupling issues, this oxidative functionalization is clean and direct. The primary byproducts are tert-butanol and cobalt salts, both of which are easily removed during the aqueous workup or silica gel chromatography described in the patent examples. This purity profile is essential for meeting the stringent specifications required by regulatory bodies for pharmaceutical ingredients. The ability to synthesize compounds like methyl (1-(pyrimidin-2-yl)-1H-indol-2-yl) benzoate in high purity demonstrates the robustness of this mechanistic approach against competing degradation pathways.
How to Synthesize C2 Alpha Acyloxy Indole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory discovery and pilot plant operations. The procedure involves a straightforward one-pot reaction where the indole substrate, solvent, oxidant, and catalyst are combined and heated. Detailed standard operating procedures regarding exact molar ratios and specific temperature ramps are critical for maximizing yield and reproducibility. For comprehensive technical details on scaling this reaction, please refer to the standardized synthesis guide below which outlines the precise experimental conditions validated in the patent data.
- Charge a reaction vessel with 2-substituted indole derivative, organic solvent, tert-butyl peroxyester, and Co(II) catalyst.
- Heat the mixture in an oil bath at 50-120°C for 5-20 hours under stirring.
- Upon completion, dilute with ethyl acetate, filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this cobalt-catalyzed technology presents a compelling value proposition centered on cost stability and supply security. The transition from precious metals like palladium to base metals like cobalt drastically reduces the raw material cost baseline, insulating the supply chain from the volatile pricing of noble metals. Furthermore, the simplified workup procedure, which avoids the removal of stubborn iodobenzene residues, translates directly into reduced processing time and lower solvent usage. This efficiency gain is not merely theoretical; it manifests as shorter batch cycles and higher throughput in manufacturing facilities, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and hypervalent iodine reagents with inexpensive cobalt salts and peroxyesters results in significant raw material savings. Additionally, the elimination of complex purification steps required to remove iodobenzene byproducts reduces solvent consumption and waste disposal costs. This streamlined process flow lowers the overall cost of goods sold (COGS) for high-purity indole derivatives, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Cobalt catalysts and tert-butyl peroxyesters are commodity chemicals with stable and robust global supply chains, unlike specialized palladium complexes which can face availability constraints. The use of common solvents such as toluene and ethyl acetate further ensures that production is not hindered by solvent shortages. This reliability allows for consistent production scheduling and reduces the risk of delays caused by raw material procurement issues, ensuring a steady flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of highly toxic reagents make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The reduced generation of hazardous iodine-containing waste aligns with increasingly strict environmental regulations, lowering the compliance burden on manufacturing sites. This green chemistry profile not only mitigates regulatory risk but also enhances the corporate sustainability metrics of the supply chain, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's capabilities and limitations for potential adopters.
Q: What are the advantages of this cobalt-catalyzed method over traditional palladium or iodine-mediated routes?
A: This method eliminates the use of expensive palladium catalysts and hypervalent iodine reagents, which generate difficult-to-remove iodobenzene byproducts. It utilizes cheap cobalt salts and serves as both oxidant and acyloxy source, significantly simplifying purification and reducing environmental impact.
Q: Does this process offer better regioselectivity compared to prior art?
A: Yes, the patent explicitly highlights high regiospecificity for the C2 alpha position, overcoming the common issue of mixed C2/C3 functionalization found in earlier methods. This ensures a cleaner impurity profile critical for pharmaceutical intermediates.
Q: Is this method scalable for industrial production of API intermediates?
A: The reaction conditions are mild (50-120°C) and utilize commercially available solvents like toluene and ethyl acetate. The absence of air-sensitive or highly toxic reagents makes it highly suitable for commercial scale-up of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2 Alpha Acyloxy Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex heterocyclic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity C2 alpha acyloxy indoles that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in cobalt catalysis allows us to optimize this specific patent technology for maximum yield and minimal impurity formation.
We invite pharmaceutical partners to leverage our technical prowess to accelerate their drug development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how this innovative cobalt-catalyzed method can enhance your supply chain efficiency and reduce overall manufacturing costs.
