Advanced Cobalt Catalysis for Scalable Production of High-Purity C2 Alpha Acyloxy Indoles
Advanced Cobalt Catalysis for Scalable Production of High-Purity C2 Alpha Acyloxy Indoles
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly indole derivatives which serve as critical building blocks for bioactive molecules. Patent CN109721590B introduces a groundbreaking methodology for the preparation of C2α acyloxy indoles utilizing a cobalt-catalyzed C-H functionalization strategy. This innovation addresses significant bottlenecks in traditional synthetic routes by employing earth-abundant Cobalt(II) catalysts and tert-butyl peroxyesters as dual oxidants and acyloxy sources. For R&D directors and procurement specialists, this technology represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates. The ability to directly functionalize the C2-alpha position of 2-substituted indoles without pre-functionalization steps offers substantial improvements in step economy and overall process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the direct C-H functionalization of indoles at the C2-alpha position was heavily reliant on methodologies that suffered from severe substrate limitations and environmental drawbacks. Existing literature, such as methods utilizing TBAI (tetrabutylammonium iodide) with PhI(OAc)2 or Palladium(II) acetate systems, were restricted primarily to 2,3-disubstituted indoles, thereby excluding a vast array of simpler 2-substituted precursors that are commercially vital. Furthermore, these traditional oxidative protocols invariably generate stoichiometric amounts of iodobenzene byproducts, which complicate downstream purification processes and increase waste disposal costs. The reliance on expensive noble metal catalysts like palladium and hypervalent iodine reagents also imposes a significant financial burden on large-scale production, limiting the economic feasibility of synthesizing these valuable intermediates for widespread therapeutic applications.
The Novel Approach
The disclosed invention overcomes these historical constraints by introducing a robust Cobalt(II) catalytic system that enables the regioselective acyloxylation of 2-substituted indoles. As illustrated in the reaction scheme below, this method utilizes readily available tert-butyl peroxyesters which serve simultaneously as the oxidant and the source of the acyloxy group, streamlining the reagent profile. 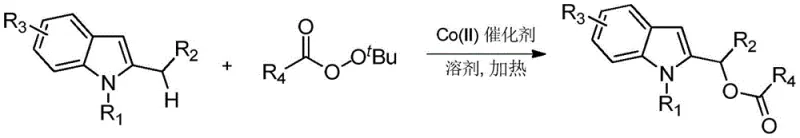 This novel approach expands the substrate scope significantly, allowing for the successful transformation of indoles bearing diverse substituents including halogens, esters, and heterocycles at the nitrogen position. By eliminating the formation of iodobenzene byproducts and replacing costly palladium with inexpensive cobalt salts, this process offers a cleaner, more atom-economical route that is inherently better suited for the rigorous demands of modern API manufacturing and supply chain optimization.
This novel approach expands the substrate scope significantly, allowing for the successful transformation of indoles bearing diverse substituents including halogens, esters, and heterocycles at the nitrogen position. By eliminating the formation of iodobenzene byproducts and replacing costly palladium with inexpensive cobalt salts, this process offers a cleaner, more atom-economical route that is inherently better suited for the rigorous demands of modern API manufacturing and supply chain optimization.
Mechanistic Insights into Co(II)-Catalyzed C-H Acyloxylation
The mechanistic underpinning of this transformation involves a radical-mediated pathway initiated by the homolytic cleavage of the peroxide bond in the tert-butyl peroxyester, facilitated by the Co(II) catalyst. This generation of acyloxy radicals allows for the direct abstraction of a hydrogen atom from the C2-alpha position of the indole ring, followed by a rapid rebound mechanism that installs the acyloxy functionality with high regioselectivity. Unlike electrophilic aromatic substitution which often targets the C3 position, this radical pathway is uniquely tuned to favor the C2-alpha site, even in the presence of competing reactive sites on the indole scaffold. The tolerance for various electronic environments, evidenced by the successful conversion of electron-deficient substrates like 5-fluoropyrimidine substituted indoles, suggests a mechanism that is resilient to electronic deactivation, ensuring consistent performance across a wide library of potential drug candidates.
From an impurity control perspective, the specificity of this cobalt-catalyzed cycle minimizes the formation of regioisomeric byproducts that typically plague non-selective oxidation reactions. The use of tert-butyl peroxyesters ensures that the only major organic byproduct is tert-butanol, which is volatile and easily removed during the concentration phase, unlike the persistent iodobenzene residues found in older methods. This purity profile is critical for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals are enforced. The ability to achieve high yields, such as the 76% isolated yield observed in the synthesis of methyl (1-(pyrimidin-2-yl)-1H-indol-2-yl) benzoate (Compound 1a shown below), demonstrates the practical viability of this mechanism for producing high-purity C2 alpha acyloxy indole intermediates. 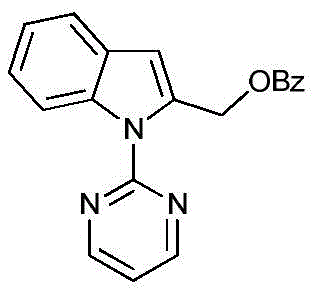
How to Synthesize C2 Alpha Acyloxy Indole Efficiently
The operational simplicity of this cobalt-catalyzed protocol makes it highly attractive for process chemists aiming to translate laboratory discoveries into pilot plant operations. The procedure involves a straightforward one-pot reaction where the 2-substituted indole derivative, solvent, oxidant, and catalyst are combined in a standard reactor vessel. Detailed standardized synthesis steps for implementing this technology in your facility are provided in the guide below.
- Charge a Schlenk bottle with 2-substituted indole derivative, solvent, tert-butyl peroxyester, and Co(II) catalyst under controlled molar ratios.
- Heat the reaction mixture in an oil bath at temperatures between 50-120°C for 5-20 hours to facilitate the C-H acyloxylation.
- Upon completion, dilute with ethyl acetate, filter, concentrate, and purify via silica gel column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology translates into tangible strategic advantages regarding cost stability and material availability. The shift from precious metal catalysts to base metal cobalt significantly reduces the raw material cost baseline, insulating the production budget from the volatile pricing trends associated with palladium and rhodium. Furthermore, the elimination of hypervalent iodine reagents removes a major cost driver and safety hazard, as peroxyesters are generally more manageable and cost-effective on a metric ton scale. This transition supports a more resilient supply chain by diversifying the sourcing of key reagents away from specialized, high-cost vendors towards commodity chemical suppliers.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant cobalt salts drastically lowers the catalyst loading costs, while the avoidance of PhI(OAc)2 eliminates the expense associated with stoichiometric hypervalent iodine oxidants. Additionally, the simplified workup procedure, which avoids the difficult removal of iodobenzene byproducts, reduces solvent consumption and processing time, leading to substantial overall cost savings in the manufacturing of complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available industrial solvents like toluene and ethyl acetate alongside commodity cobalt salts, this process mitigates the risk of supply disruptions often caused by the scarcity of specialized reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to failed runs or extensive reprocessing, thereby securing a steady flow of high-purity intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The mild reaction temperatures ranging from 50-120°C and the absence of toxic heavy metal waste streams make this process highly scalable and environmentally compliant. The generation of benign tert-butanol as a byproduct simplifies waste treatment protocols, aligning with increasingly stringent global environmental regulations and facilitating easier permitting for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed C-H functionalization technology. These insights are derived directly from the experimental data and scope defined in the patent documentation to assist decision-makers in evaluating the feasibility of this route for their specific projects.
Q: What are the advantages of this cobalt catalyzed method over traditional palladium or iodine mediated routes?
A: This method eliminates the use of expensive palladium catalysts and hypervalent iodine oxidants like PhI(OAc)2, which generate difficult-to-remove iodobenzene byproducts. It utilizes cheap Co(II) salts and tert-butyl peroxyesters, offering better atom economy and simplified purification.
Q: Does this process support a broad range of substrate functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating substrates with halogens (F, Cl, Br, I), esters, ethers, and heterocycles like pyrimidine, making it suitable for diverse pharmaceutical intermediate synthesis.
Q: Is this method scalable for industrial production of indole derivatives?
A: The reaction operates under mild thermal conditions (50-120°C) using common organic solvents like toluene or DCE, and avoids sensitive reagents, indicating strong potential for commercial scale-up and robust supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2 Alpha Acyloxy Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed technology in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of C2 alpha acyloxy indole delivered meets the highest standards required for global pharmaceutical registration.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can optimize your specific project requirements. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your target molecules. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
