Revolutionizing Triazole Synthesis: Scalable Production of High-Purity 3-Trifluoromethyl Intermediates for Pharmaceutical Applications
The patent CN114920707B introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative approach leverages dimethylformamide (DMF) as both solvent and carbon source under ambient atmospheric conditions, eliminating the stringent anhydrous and anaerobic requirements that have historically plagued traditional triazole syntheses. The process demonstrates exceptional operational simplicity through its one-pot reaction protocol that operates effectively at 120°C for 12 hours without specialized equipment or inert gas handling. By utilizing molecular iodine as a promoter and commercially available starting materials including trifluoroethyliminohydrazide and standard DMF solvent, this method achieves remarkable substrate versatility across diverse aryl substitutions while maintaining consistent product quality. The elimination of complex purification steps and compatibility with standard laboratory apparatus positions this technology as a transformative solution for manufacturing critical pharmaceutical intermediates with enhanced supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles typically require transition metal catalysts such as palladium or copper complexes under strictly anhydrous and anaerobic conditions, creating significant operational complexities and cost burdens for pharmaceutical manufacturers. These methods often necessitate specialized glovebox equipment, rigorous solvent drying protocols, and multiple purification steps to remove metal residues that could compromise final product purity. The sensitivity to moisture and oxygen frequently results in batch inconsistencies and reduced yields when scaling from laboratory to production environments. Furthermore, conventional approaches typically employ expensive fluorinated building blocks that require multi-step preparation, increasing both raw material costs and process development timelines. The need for cryogenic temperatures or high-pressure reactors in some methodologies further complicates manufacturing scalability while introducing additional safety considerations that impact facility design and operational costs.
The Novel Approach
This patented methodology overcomes these limitations through an elegant iodine-promoted tandem cyclization that operates efficiently in air at 120°C using DMF as a dual-function reagent. The process eliminates all transition metal catalysts while maintaining high functional group tolerance across diverse aryl substitutions including halogenated, alkylated, and heteroaromatic systems. By utilizing DMF both as solvent and carbon source, the reaction achieves remarkable atom economy without requiring additional reagents or specialized equipment. The ambient atmospheric conditions enable straightforward implementation in standard manufacturing facilities without inert gas infrastructure, significantly reducing capital expenditure requirements. The simplified workup procedure involving basic filtration followed by column chromatography purification ensures consistent product quality while minimizing waste streams. This approach demonstrates exceptional scalability from laboratory to commercial production through its robust reaction profile that maintains consistent yields across different batch sizes.
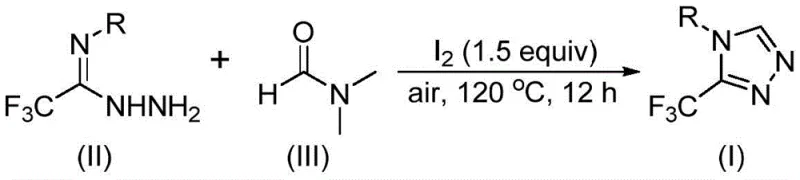
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The reaction mechanism operates through two distinct but complementary pathways that both utilize DMF as a carbon source. In the formyl group pathway, the carbonyl oxygen of DMF activates the imine nitrogen of trifluoroethyliminohydrazide through hydrogen bonding, facilitating nucleophilic attack that forms a hydrazone intermediate. Subsequent intramolecular cyclization eliminates dimethylamine to yield the triazole core structure. The N-methyl pathway involves initial iodination of DMF's methyl group followed by dehydrohalogenation to generate an iminium species that undergoes nucleophilic addition with the hydrazide nitrogen. This forms an azadiene intermediate that subsequently undergoes intramolecular cyclization and oxidative aromatization to produce the final triazole product. Both pathways proceed concurrently under the reaction conditions, providing complementary routes that enhance overall reaction efficiency and substrate scope.
Impurity control is achieved through the inherent selectivity of the iodine-mediated cyclization process which minimizes side reactions typically associated with metal-catalyzed systems. The absence of transition metals eliminates potential metal contamination pathways while the air-stable reaction conditions prevent oxidation byproducts that commonly occur in traditional syntheses requiring inert atmospheres. The well-defined reaction profile maintains consistent product distribution across different substrate substitutions as demonstrated by the patent's comprehensive examples showing uniform purity levels across diverse aryl groups. The simplified purification protocol using standard column chromatography effectively removes any minor byproducts without requiring specialized techniques like chelation or extraction steps that could introduce additional impurities.
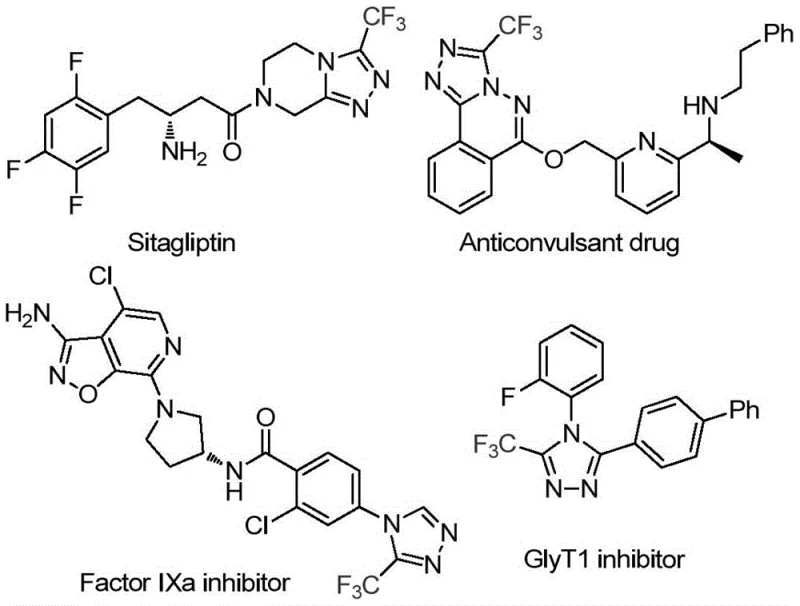
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
This patent discloses a streamlined manufacturing process for producing high-purity trifluoromethyl triazoles with exceptional operational simplicity and scalability potential. The methodology represents a significant improvement over conventional approaches by eliminating specialized equipment requirements while maintaining excellent functional group tolerance across diverse substrate classes. Detailed standardized synthesis procedures have been developed to ensure consistent product quality at commercial scale, with specific protocols optimized for different aryl substitution patterns. The following section provides comprehensive step-by-step guidance for implementing this technology in pharmaceutical manufacturing environments.
- Combine trifluoroethyliminohydrazide (II), DMF (III), and molecular iodine (1.5 equiv) in air atmosphere at room temperature with magnetic stirring
- Heat reaction mixture to 120°C under ambient conditions and maintain for 12 hours with continuous agitation
- Perform post-treatment via filtration, silica gel mixing, and column chromatography purification to obtain target triazole compound (I)
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The elimination of specialized infrastructure requirements reduces capital investment barriers while enhancing manufacturing flexibility across diverse production facilities. The use of commercially available starting materials creates resilient supply chains less vulnerable to single-source dependencies, while the simplified process flow enables rapid scale-up from development to commercial production volumes. These advantages collectively contribute to more reliable delivery schedules and improved cost structures without compromising on quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive catalyst procurement costs along with complex metal removal processes that require additional equipment and validation steps. By utilizing DMF as both solvent and reactant, the process achieves superior atom economy while reducing raw material expenses through simplified sourcing requirements. The air-stable reaction conditions eliminate costly inert gas infrastructure and associated operational expenses, creating significant cost savings throughout the manufacturing lifecycle without requiring capital-intensive facility modifications.
- Enhanced Supply Chain Reliability: The reliance on universally available starting materials including standard DMF solvent and commercially accessible iodine reagent creates robust supply chains less susceptible to single-source vulnerabilities. The simplified process flow enables seamless technology transfer between manufacturing sites while maintaining consistent product quality across different production environments. This operational flexibility allows for rapid response to demand fluctuations through distributed manufacturing capabilities without requiring specialized equipment or reagent sourcing networks.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production through its robust reaction profile that maintains consistent yields across different batch sizes without requiring process reoptimization. The elimination of hazardous metal catalysts significantly reduces waste streams while simplifying waste treatment protocols, aligning with increasingly stringent environmental regulations. The simplified purification requirements minimize solvent consumption and energy usage compared to traditional methods, contributing to more sustainable manufacturing operations while meeting global regulatory standards for pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented triazole synthesis methodology. These answers are derived directly from the patent's technical specifications and experimental data to provide accurate information for procurement and R&D decision-making processes.
Q: Why does this method eliminate the need for anhydrous/anaerobic conditions?
A: The reaction utilizes molecular iodine as promoter in air atmosphere, with DMF serving as both solvent and carbon source. This eliminates moisture-sensitive catalysts and inert gas requirements while maintaining high conversion rates through iodine-mediated activation pathways.
Q: How does DMF function as dual carbon source in this synthesis?
A: DMF provides two distinct carbon incorporation pathways: the formyl group undergoes condensation to form hydrazone intermediates followed by cyclization, while the N-methyl group generates azadiene species through iodine-promoted dehydrohalogenation, enabling versatile substrate scope without additional reagents.
Q: What ensures consistent purity in commercial-scale production?
A: The process avoids transition metal catalysts that require complex removal steps, while the air-stable reaction conditions prevent oxidation byproducts. Standardized column chromatography purification with defined solvent systems achieves stringent purity specifications required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
Our patented technology represents a significant advancement in the manufacturing of critical triazole intermediates for pharmaceutical applications, offering unparalleled operational simplicity combined with exceptional product quality. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our dedicated technical teams ensure seamless technology transfer and process optimization for your specific manufacturing requirements, providing comprehensive support from initial feasibility assessment through full-scale commercial production.
Leverage our expertise to accelerate your development timeline through our Customized Cost-Saving Analysis service tailored to your specific production needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your target compounds.
