Revolutionizing Acetophenone Production via Solvent-Free Mechanochemical Oxidation
The landscape of fine chemical synthesis is undergoing a paradigm shift driven by the urgent need for greener, safer, and more energy-efficient manufacturing processes. Patent CN110483222B introduces a groundbreaking methodology for the selective oxidation of ethylbenzene compounds to produce acetophenone derivatives, utilizing solid-phase ball milling technology. This innovation addresses critical bottlenecks in the production of pharmaceutical intermediates and agrochemical precursors by eliminating the reliance on volatile organic solvents during the reaction phase. By leveraging mechanochemical forces at room temperature, this technique not only reduces energy consumption but also mitigates the safety hazards associated with traditional thermal oxidation methods. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding this technological leap is essential for future-proofing supply chains and achieving sustainability goals.
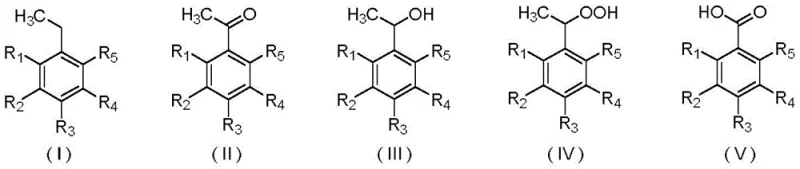
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of acetophenone and its derivatives has relied heavily on liquid-phase oxidation using molecular oxygen or harsh chemical oxidants. These conventional routes are plagued by significant inefficiencies, primarily due to the stability of molecular oxygen which necessitates high reaction temperatures and the use of large quantities of transition metal catalysts or co-catalysts. Furthermore, the liquid-phase environment often leads to incomplete decomposition of intermediate peroxides, causing them to accumulate and creating severe safety risks regarding potential explosions. The extensive use of organic solvents in these traditional processes also generates substantial hazardous waste, complicating disposal and increasing the environmental footprint, which is increasingly untenable for modern cost reduction in fine chemical manufacturing strategies.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a solvent-free, solid-state mechanochemical reaction that operates effectively at ambient temperatures ranging from 20 to 30°C. By utilizing a ball mill to generate shear, friction, and impact forces, the reactants are activated mechanically rather than thermally, allowing for precise control over the oxidation process without the need for external heating. This method significantly enhances the selectivity towards the desired acetophenone products while minimizing the formation of unwanted by-products like benzoic acids or alcohols. The elimination of bulk organic solvents during the reaction phase not only simplifies the workflow but also aligns perfectly with the principles of green chemistry, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Metalloporphyrin-Catalyzed Mechanochemical Oxidation
The core of this innovative synthesis lies in the utilization of biomimetic metalloporphyrin catalysts, which serve as model compounds for Cytochrome P450 enzymes found in biological systems. These catalysts, represented structurally in Formula VI, possess exceptional catalytic activity, enabling the transformation of substrates at incredibly low molar ratios ranging from 1:200,000 to 1:500. The mechanical energy provided by the ball milling process facilitates the interaction between the solid oxidant, the substrate, and the catalyst surface, promoting the formation of active oxygen species that selectively attack the ethyl group. This mechanistic pathway avoids the radical chain reactions typical of thermal oxidation, thereby preventing the runaway reactions that lead to safety incidents and poor selectivity in traditional methods.
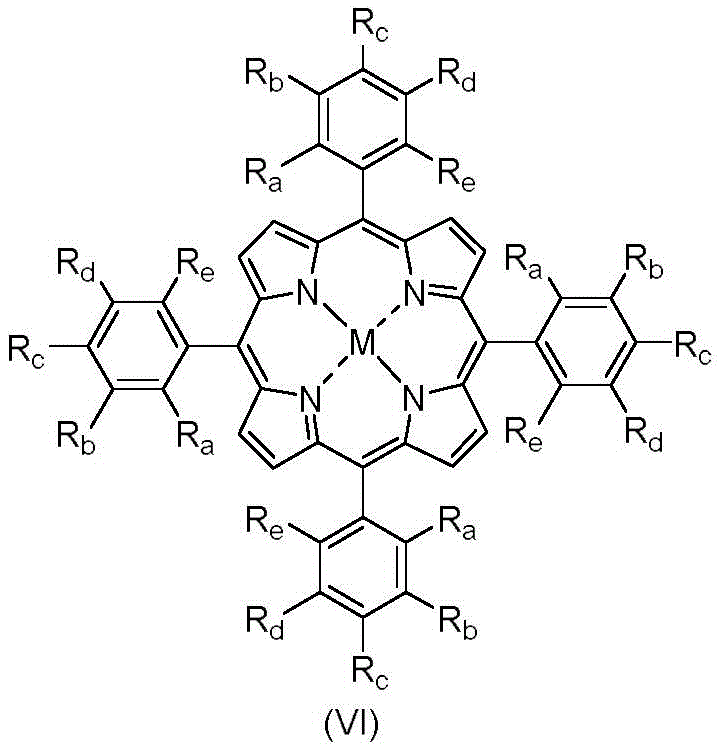
Furthermore, the impurity control mechanism inherent in this solid-state system is superior to liquid-phase alternatives. The physical confinement of reactants within the ball mill jar, combined with the specific electronic properties of the metalloporphyrin center (such as Cobalt or Manganese), directs the oxidation specifically to the benzylic position. This high degree of regioselectivity ensures that side reactions, such as ring oxidation or over-oxidation to carboxylic acids, are suppressed. For R&D directors focused on purity and impurity profiles, this means a cleaner crude reaction mixture that requires less intensive downstream purification, ultimately preserving the yield of high-value intermediates and reducing the loss of material during isolation steps.
How to Synthesize Acetophenone Derivatives Efficiently
Implementing this mechanochemical protocol requires precise attention to the physical parameters of the ball milling equipment and the stoichiometry of the solid reagents. The process begins with the uniform mixing of the ethylbenzene substrate, the specific metalloporphyrin catalyst, a solid oxidant like t-butyl hydroperoxide or hydrogen peroxide, and a dispersant such as anhydrous sodium sulfate. The detailed standardized synthesis steps below outline the optimal conditions for rotation speed, duration, and post-reaction workup to ensure maximum conversion and selectivity, serving as a foundational guide for process chemists looking to adopt this technology.
- Load the ethylbenzene substrate, metalloporphyrin catalyst, oxidant (such as t-butyl hydroperoxide), and a solid dispersant into an agate ball mill jar.
- Seal the jar and perform ball milling at room temperature (20-30°C) with a rotation speed between 100 and 800 rpm for a duration of 3 to 24 hours, pausing periodically to release gas.
- Post-process the reaction mixture by dissolving in absolute ethanol, filtering, and recovering the product via distillation to obtain high-purity acetophenone derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free ball milling technology presents a compelling value proposition centered on operational efficiency and risk mitigation. The most immediate benefit is the drastic reduction in raw material costs associated with organic solvents, as the reaction proceeds in a solid state, thereby eliminating the need for purchasing, storing, and recovering large volumes of volatile liquids. Additionally, the room temperature operation significantly lowers energy expenditures related to heating and cooling systems, contributing to substantial cost savings in manufacturing overheads without compromising on production throughput or product quality standards.
- Cost Reduction in Manufacturing: The elimination of organic solvents during the reaction phase removes the capital and operational expenses tied to solvent recovery units and waste treatment facilities. By avoiding the use of expensive transition metal catalysts in high loadings and instead utilizing highly efficient metalloporphyrins, the overall cost of goods sold is optimized. This qualitative shift in process design allows for a leaner manufacturing model where resource utilization is maximized, directly impacting the bottom line through reduced variable costs per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Operating at room temperature with stable solid reagents reduces the dependency on specialized high-pressure or high-temperature reactor infrastructure, making the supply chain more resilient to equipment failures. The use of commercially available analytical grade reagents and simple agate ball mill jars ensures that raw material sourcing is straightforward and less susceptible to geopolitical or logistical disruptions. This simplicity in equipment and reagent requirements guarantees a more consistent and reliable supply of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The mechanochemical nature of this process is inherently scalable, as ball milling technology can be adapted from laboratory scales to industrial continuous flow mills without significant changes to the reaction chemistry. The low peroxide content and absence of toxic solvent emissions ensure strict compliance with increasingly stringent environmental regulations, avoiding potential fines and shutdowns. This environmental compatibility enhances the long-term viability of the production facility and aligns with the corporate social responsibility goals of major multinational chemical consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented oxidation method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, scalability, and product quality for stakeholders evaluating this technology for integration into their existing manufacturing portfolios.
Q: How does this mechanochemical method improve safety compared to traditional oxidation?
A: Traditional liquid-phase oxidation often leads to the accumulation of unstable peroxide intermediates, posing explosion risks. This solid-state ball milling method ensures low peroxide content and operates at room temperature, drastically enhancing process safety.
Q: Is organic solvent required for the reaction phase in this new process?
A: No, the core reaction occurs in a solid-state environment without the need for organic solvents. Solvents like ethanol are only used in the post-reaction workup for extraction and purification, significantly reducing VOC emissions and waste disposal costs.
Q: What types of catalysts are compatible with this ball milling oxidation technique?
A: The process utilizes biomimetic metalloporphyrin catalysts, specifically those modeled after Cytochrome P450, containing central metals such as Cobalt, Manganese, Iron, or Nickel, which offer high catalytic activity at extremely low loading ratios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetophenone Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies such as the mechanochemical oxidation described in CN110483222B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of acetophenone derivative meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to engage with our technical procurement team to discuss how this innovative solvent-free technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, sustainability, and technical excellence.
