Optimizing Terbutaline Sulfate Production: A Technical Breakthrough for Global Pharmaceutical Supply Chains
Optimizing Terbutaline Sulfate Production: A Technical Breakthrough for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe synthetic routes for critical active pharmaceutical ingredients (APIs) and their intermediates. A significant advancement in this domain is detailed in patent CN110950765A, which discloses a novel preparation method for terbutaline sulfate, a widely used beta-2 adrenergic agonist for treating asthma and other obstructive airway diseases. This technical insight report analyzes the strategic value of this patented process, highlighting its departure from hazardous traditional methods towards a more sustainable and commercially viable manufacturing protocol. By leveraging 3,5-dihydroxyacetophenone as a cost-effective starting material and employing a sequence of benzyl protection, copper-mediated functionalization, and reductive amination, this route addresses longstanding pain points in yield optimization and operational safety. For R&D directors and procurement strategists, understanding the nuances of this chemistry is essential for securing a reliable terbutaline sulfate supplier capable of meeting stringent global quality standards while minimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of terbutaline sulfate has been plagued by significant technical and safety hurdles that impede efficient commercial scale-up of complex pharmaceutical intermediates. Early methodologies, such as those described in US3937838, relied on multi-step sequences involving diazomethane, a highly explosive and toxic reagent that poses severe safety risks in large-scale manufacturing environments. Furthermore, alternative routes reported by research institutes often utilized methyllithium, a pyrophoric metal reagent requiring rigorous anhydrous conditions and specialized handling equipment, alongside selenium dioxide for oxidation steps. The use of selenium dioxide is particularly problematic due to its high toxicity and the generation of hazardous selenium-containing waste streams, which complicate environmental compliance and waste treatment protocols. These conventional approaches not only suffer from long reaction sequences and low overall yields but also introduce substantial variability in product purity, making the purification of the final API difficult and costly. Consequently, these legacy processes are increasingly viewed as unsustainable for modern green chemistry initiatives and cost-sensitive supply chains.
The Novel Approach
In stark contrast to these hazardous legacy routes, the methodology outlined in patent CN110950765A introduces a streamlined and safer synthetic strategy that fundamentally reshapes the production landscape. This novel approach initiates with the readily available 3,5-dihydroxyacetophenone, bypassing the need for expensive or dangerous carboxylic acid derivatives. The core innovation lies in the substitution of toxic oxidants with a copper bromide and dimethyl sulfoxide (DMSO) system, which effectively converts the ketone to the requisite aldehyde intermediate under mild conditions. By avoiding high-temperature and high-pressure reactions, this process significantly lowers the barrier to entry for manufacturing facilities, allowing for the use of standard reactor setups. The elimination of heavy metal contaminants like selenium and the avoidance of pyrophoric reagents like methyllithium drastically simplifies the downstream purification process, leading to higher purity profiles and reduced solvent consumption. This shift represents a paradigm change in cost reduction in pharmaceutical intermediates manufacturing, offering a pathway that is not only chemically elegant but also economically superior.
Mechanistic Insights into Copper-Mediated Bromination and Reductive Amination
The chemical elegance of this synthesis is rooted in its precise control over functional group transformations, particularly during the critical alpha-functionalization and amination stages. The process begins with the protection of the phenolic hydroxyl groups using benzyl chloride and potassium carbonate in acetone, creating a robust 3,5-dibenzyloxyacetophenone scaffold that prevents unwanted side reactions during subsequent steps. The pivotal transformation occurs in the second step, where copper(II) bromide mediates the alpha-bromination of the ketone in a mixed solvent system of chloroform and ethyl acetate. This is immediately followed by a Kornblum-type oxidation using DMSO and sodium bicarbonate, which cleanly converts the alpha-bromo ketone into the corresponding phenylacetaldehyde derivative. This two-step one-pot style sequence is crucial for maintaining high throughput and minimizing the isolation of unstable intermediates. Following this, the aldehyde undergoes reductive amination with tert-butylamine in ethanol, forming an imine intermediate that is subsequently reduced by potassium borohydride to yield the protected amino-alcohol. This choice of reducing agent is strategic, as it offers excellent chemoselectivity without affecting the benzyl protecting groups or the aromatic ring.
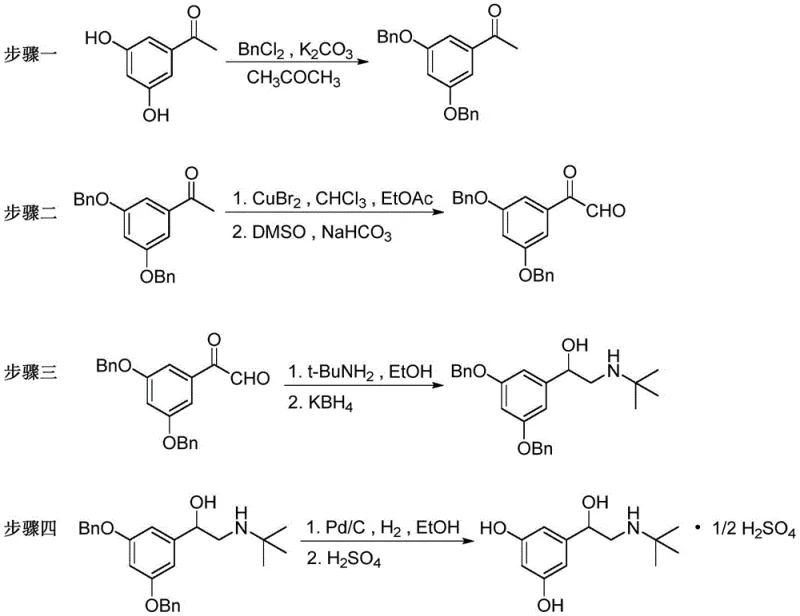
The final stage involves the removal of the benzyl protecting groups via catalytic hydrogenation using palladium on carbon (Pd/C) under a hydrogen atmosphere, followed by salt formation with sulfuric acid. This deprotection step is highly efficient, occurring at mild temperatures between 20-50°C and low pressures of 0.1-0.5 MPa, ensuring the integrity of the chiral center and the overall molecular structure. From an impurity control perspective, the use of crystallization steps after the protection and amination stages serves as a powerful purification tool, removing unreacted starting materials and side products before they can propagate through the synthesis. The avoidance of harsh acidic or basic hydrolysis conditions further preserves the stereochemical purity of the molecule, which is critical for the biological activity of the final terbutaline sulfate API. This mechanistic clarity provides R&D teams with a predictable and controllable process, facilitating easier technology transfer and regulatory filing.
How to Synthesize Terbutaline Sulfate Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize yield and purity. The patent specifies the use of acetone for the initial protection step, which offers a balance of solubility and ease of removal, while the bromination utilizes a chloroform-ethyl acetate mixture to optimize the reactivity of copper bromide. For the reductive amination, ethanol is preferred as a green solvent that supports both the condensation and reduction phases effectively. Operators must strictly adhere to the specified temperature ranges, such as maintaining 50-80°C for protection and 30-80°C for bromination, to prevent thermal degradation or incomplete conversion. The detailed standardized synthesis steps, including specific workup procedures like washing with concentrated hydrochloric acid to remove copper residues and recrystallization protocols, are essential for achieving the high-quality standards required for pharmaceutical applications. For a comprehensive guide on executing these reactions with precision, please refer to the technical protocol below.
- Perform benzyl protection on 3,5-dihydroxyacetophenone using benzyl chloride and potassium carbonate in acetone to obtain 3,5-dibenzyloxyacetophenone.
- Execute alpha-bromination with copper bromide followed by oxidation with DMSO and sodium bicarbonate to generate the key aldehyde intermediate.
- Conduct reductive amination with tert-butylamine and potassium borohydride in ethanol to form the protected amino-alcohol intermediate.
- Finalize the synthesis through catalytic hydrogenation using Pd/C to remove benzyl groups, followed by salt formation with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial cost savings derived from the utilization of commodity-grade raw materials. By sourcing 3,5-dihydroxyacetophenone and common reagents like benzyl chloride and potassium carbonate, manufacturers can insulate themselves from the price volatility associated with exotic or specialized starting materials. Furthermore, the elimination of toxic selenium reagents removes the need for expensive hazardous waste disposal services and complex effluent treatment systems, leading to significant operational expenditure reductions. The mild reaction conditions also imply lower energy consumption, as there is no requirement for cryogenic cooling or extreme heating, thereby reducing the utility burden on the manufacturing plant. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring that the final product remains competitive in the global market.
- Cost Reduction in Manufacturing: The replacement of high-cost, high-risk reagents with inexpensive alternatives directly impacts the bottom line. For instance, substituting selenium dioxide with a copper bromide/DMSO system eliminates the procurement costs of toxic oxidants and the associated safety infrastructure. Additionally, the simplified purification process reduces solvent usage and processing time, further driving down the cost of goods sold (COGS). This economic efficiency allows suppliers to offer more competitive pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures a stable supply of inputs, mitigating the risk of production stoppages due to raw material shortages. Unlike routes dependent on custom-synthesized intermediates or regulated substances, this method leverages a robust global chemical market. This reliability is crucial for maintaining consistent delivery schedules to downstream API manufacturers and pharmaceutical companies, fostering long-term partnerships based on trust and dependability.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from pilot plants to multi-ton commercial production. The absence of explosive diazomethane or pyrophoric methyllithium simplifies safety audits and regulatory approvals, accelerating the time-to-market for new generic formulations. Moreover, the greener profile of the synthesis aligns with increasingly strict environmental regulations, future-proofing the manufacturing asset against evolving sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, scalability, and quality control measures. Understanding these aspects is vital for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: How does this new process improve safety compared to traditional selenium dioxide oxidation?
A: The patented method replaces highly toxic selenium dioxide oxidants with a safer copper bromide and DMSO system. This eliminates the handling risks associated with volatile and toxic selenium compounds, significantly reducing environmental hazards and operator exposure risks during industrial scale-up.
Q: What are the key advantages regarding raw material availability for this synthesis route?
A: The process utilizes 3,5-dihydroxyacetophenone as a starting material, which is cheap and easily available compared to complex precursors used in older patents. Additionally, reagents like benzyl chloride, copper bromide, and potassium borohydride are commodity chemicals, ensuring stable supply chains and preventing procurement bottlenecks.
Q: Does this method require high-pressure or high-temperature conditions?
A: No, the reaction conditions are notably mild. The benzyl protection occurs at 50-80°C, bromination at 30-80°C, and hydrogenation at low pressure (0.1-0.5 MPa) and moderate temperature (20-50°C). This mild profile reduces energy consumption and allows for the use of standard glass-lined or stainless steel reactors without specialized high-specification equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity terbutaline sulfate and related intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By integrating the innovative processes described in patent CN110950765A, we can offer our partners a superior product profile with improved impurity control and consistent batch-to-batch reproducibility.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or seeking a Customized Cost-Saving Analysis for your upcoming projects, we are ready to provide the data-driven support you need. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can drive value for your organization.
