Advanced Supported Catalyst Technology For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
Advanced Supported Catalyst Technology For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
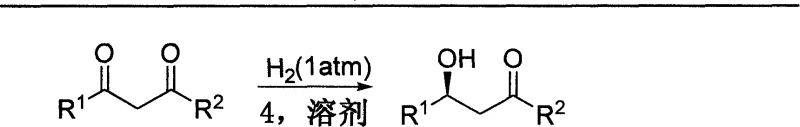
The landscape of chiral synthesis is undergoing a transformative shift driven by the urgent need for safer, more sustainable, and cost-effective manufacturing processes. A pivotal development in this domain is documented in patent CN100389877C, which introduces a novel supported catalyst system designed for the preparation of chiral secondary alcohols under normal pressure conditions. This technology addresses critical bottlenecks in the production of high-value pharmaceutical intermediates, particularly those required for antidepressants and other bioactive compounds. By utilizing polyethylene glycol macromolecules as carriers and specific chiral phosphine ligands coordinated with ruthenium, this innovation enables asymmetric catalytic hydrogenation without the need for hazardous high-pressure autoclaves. For R&D directors and process chemists, this represents a significant leap forward in operational safety and efficiency, allowing for the generation of optically pure compounds with exceptional stereoselectivity while drastically simplifying downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral secondary alcohols often rely on homogeneous catalytic systems, such as the renowned Noyori Binap-Ru catalysts, which, while effective, present substantial industrial challenges. These conventional processes typically necessitate the use of high-pressure hydrogenation equipment, introducing significant safety risks and capital expenditure requirements for specialized reactors capable of withstanding extreme conditions. Furthermore, the separation of the homogeneous catalyst from the final product is notoriously difficult and costly, often requiring complex chromatographic purification or extensive washing steps that reduce overall yield. The inability to recover the expensive noble metal catalyst leads to high operational costs and generates heavy metal waste, posing environmental compliance issues. Additionally, the potential for toxic ruthenium residues to remain in the final active pharmaceutical ingredient is a major regulatory concern that can delay product approval and compromise patient safety.
The Novel Approach
In stark contrast, the technology described in patent CN100389877C offers a robust alternative by employing a soluble polymer-supported catalyst that operates efficiently at ambient pressure. This approach combines the high activity and selectivity of homogeneous catalysis with the ease of separation characteristic of heterogeneous systems. The use of polyethylene glycol as a support medium ensures that the catalyst remains soluble during the reaction, facilitating excellent mass transfer and interaction with the substrate, yet allows for simple precipitation and filtration upon completion. This dual nature eliminates the need for high-pressure vessels, thereby reducing energy consumption and infrastructure costs significantly. Moreover, the catalyst can be recovered and reused multiple times without degradation of its stereochemical performance, providing a sustainable solution that aligns with green chemistry principles while ensuring consistent product quality.

Mechanistic Insights into PEG-Supported Ruthenium Catalysis
The success of this catalytic system lies in the unique physicochemical properties of the polyethylene glycol support and its interaction with the chiral ligand environment. Unlike rigid insoluble supports that can sterically hinder the active site and reduce enantioselectivity, the flexible PEG chains create a solvated microenvironment that mimics homogeneous conditions. This "quasi-homogeneous" state prevents the aggregation of ruthenium species into inactive dimers, a common deactivation pathway in unsupported systems, thereby maintaining high turnover frequencies even at low catalyst loadings. The specific coordination of the chiral diphosphine ligand to the ruthenium center is preserved effectively, ensuring that the steric and electronic factors required for high enantiomeric excess are not compromised by the polymer backbone. This delicate balance allows the reaction to proceed with rapid kinetics at moderate temperatures, typically between 50°C and 60°C, achieving complete conversion in a fraction of the time required by traditional supported catalysts.
Furthermore, the mechanism of product isolation is elegantly simple yet highly effective for impurity control. Upon completion of the hydrogenation, the addition of a non-solvent such as diethyl ether causes the polymer-supported catalyst to precipitate out of the solution, leaving the chiral alcohol product in the supernatant. This phase separation is driven by the solubility characteristics of the PEG chain, which collapses in the presence of the anti-solvent. This physical separation method ensures that the final product stream is virtually free of catalyst residues, with ICP analysis confirming ruthenium levels below 10 ppm. This intrinsic purification step reduces the burden on subsequent refining processes, such as distillation or crystallization, and minimizes the risk of metal contamination in the final API. The robustness of the ligand attachment to the polymer also ensures that leaching of the chiral phosphine is negligible, preserving the optical purity of the product over multiple recycling cycles.
How to Synthesize Chiral Secondary Alcohol Efficiently
The implementation of this catalytic technology involves a streamlined sequence of operations that can be readily integrated into existing manufacturing workflows. The process begins with the preparation of the supported ligand, followed by the complexation with the ruthenium precursor, and finally the hydrogenation reaction itself. Each step is optimized for scalability and reproducibility, utilizing commonly available reagents and standard laboratory or plant equipment. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, temperature controls, and solvent choices necessary to achieve optimal performance. By adhering to these protocols, manufacturers can ensure consistent batch-to-batch quality while maximizing the economic benefits of catalyst recovery and reuse.
- Prepare the chiral hydroxybiphenyl bisphosphine ligand by demethylating the methoxy precursor using boron tribromide at low temperature followed by warming to room temperature.
- Attach the chiral ligand to the polyethylene glycol carrier by reacting the hydroxy-ligand with mesylated PEG in the presence of a base like cesium carbonate in DMF.
- Complex the supported ligand with a ruthenium precursor such as (COD)Ru(allyl)2 and hydrobromic acid in acetone to generate the final active catalyst species.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this supported catalyst technology translates into tangible strategic advantages that extend beyond mere technical performance. The elimination of high-pressure reaction requirements fundamentally alters the risk profile of the manufacturing process, allowing for production in standard glass-lined or stainless steel reactors rather than specialized autoclaves. This flexibility significantly reduces capital investment barriers and expands the pool of qualified contract manufacturing organizations capable of executing the synthesis. Additionally, the ability to recycle the catalyst multiple times directly impacts the cost of goods sold by amortizing the expense of the expensive ruthenium and chiral ligand over a much larger volume of product. This efficiency gain is compounded by the reduction in waste disposal costs associated with heavy metal contaminants, aligning production with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the drastic simplification of the reaction conditions and the reusability of the catalytic system. By operating at normal pressure, the energy consumption associated with gas compression and high-temperature maintenance is significantly reduced, leading to lower utility costs per kilogram of product. Furthermore, the quantitative recovery of the catalyst means that the effective loading of precious metals per batch is minimized, decoupling production costs from the volatile market prices of ruthenium. The simplified workup procedure also reduces solvent usage and labor hours required for purification, contributing to an overall leaner and more cost-competitive manufacturing process that enhances margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness and stability of the supported catalyst, which can be prepared in advance and stored or generated in situ with high reliability. The use of commercially available polyethylene glycol carriers and standard chemical reagents ensures that raw material sourcing is not subject to the bottlenecks often associated with exotic or proprietary ligands. Moreover, the high yield and selectivity of the reaction minimize the need for reprocessing or scrapping off-spec batches, ensuring a steady and predictable output of critical intermediates. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery schedules of downstream pharmaceutical customers who require uninterrupted supply for their own formulation lines.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the absence of mass transfer limitations typically encountered in heterogeneous catalysis, as the soluble nature of the support ensures uniform reaction kinetics regardless of vessel size. The process inherently generates less hazardous waste due to the lack of heavy metal contamination in the effluent streams, simplifying wastewater treatment and reducing the environmental footprint of the facility. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate sustainability profile, which is increasingly becoming a key criterion for supplier selection by major multinational pharmaceutical companies committed to responsible sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this supported catalyst system. These insights are derived directly from the experimental data and comparative studies presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for process engineers and quality assurance teams who are evaluating the feasibility of integrating this technology into their current production portfolios. The answers reflect the proven capabilities of the system in terms of activity, selectivity, and operational safety.
Q: What are the primary advantages of this supported catalyst over traditional homogeneous systems?
A: The primary advantages include the ability to operate at normal atmospheric pressure rather than high pressure, simplified product separation via precipitation, and the capability to recycle the expensive ruthenium catalyst multiple times without significant loss of activity or stereoselectivity.
Q: How does the polyethylene glycol support affect the reaction kinetics?
A: The polyethylene glycol support renders the catalyst soluble in the reaction medium, creating a quasi-homogeneous environment that maximizes contact between the catalyst and substrate while preventing the formation of inactive ruthenium dimers, thus maintaining high catalytic activity.
Q: What is the expected residual metal content in the final product?
A: Due to the efficient separation of the polymer-supported catalyst from the product solution, the residual ruthenium content in the final chiral alcohol product is typically less than 10 ppm, meeting stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Secondary Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to drive innovation and efficiency in the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of chiral intermediate meets the highest global standards. Our commitment to technical excellence allows us to leverage innovations like the PEG-supported ruthenium catalyst to deliver superior value to our partners, combining high optical purity with cost-effective manufacturing solutions.
We invite you to collaborate with us to explore how this cutting-edge technology can optimize your specific synthesis routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating the potential economic impact of switching to this ambient pressure process. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral synthesis can become a strategic asset for your organization's growth and competitiveness in the global market.
