Advanced One-Pot Synthesis of Nifuratel for Industrial Scale Pharmaceutical Manufacturing
Introduction to Advanced Nifuratel Synthesis Technology
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational simplicity, particularly for broad-spectrum anti-infective agents like Nifuratel. Patent CN103232445A introduces a transformative methodology that addresses critical bottlenecks in the traditional manufacturing of this vital gynecological therapeutic. By innovatively adopting a post-treatment strategy utilizing ethanol to remove water, this novel approach effectively circumvents the need for high-vacuum reduced pressure distillation, a step that has historically plagued production yields and equipment complexity. The technical breakthrough lies in the seamless integration of intermediate preparation and final condensation, resulting in a streamlined workflow that significantly enhances process controllability. This advancement not only ensures a final product purity exceeding 99.8% but also establishes a foundation for more sustainable and cost-effective manufacturing practices. For global supply chain stakeholders, this represents a pivotal shift towards more reliable sourcing of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates such as 3-methylthio-2-hydroxypropyl hydrazine has relied heavily on high-vacuum underpressure distillation for purification. While this traditional technique can achieve high intermediate purity, it introduces severe operational challenges that hinder industrial scalability. The requirement for maintaining high vacuum conditions creates significant difficulties in process control, often leading to fluctuating yields and increased energy consumption. Furthermore, the complexity of the equipment needed to sustain such vacuum levels elevates capital expenditure and maintenance costs, creating a barrier for efficient mass production. These conventional routes often involve multiple isolation steps that increase the risk of material loss and environmental contamination, thereby reducing the overall economic viability of the manufacturing process. Consequently, producers have long sought a method that could maintain quality standards without the burdensome infrastructure of high-vacuum systems.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the purification landscape by implementing an ethanol-assisted water removal technique. This method allows for the effective dehydration of the reaction mixture under much milder vacuum conditions, specifically controlling the vacuum degree at less than or equal to 0.09MPa. By eliminating the dependency on extreme high-vacuum distillation, the process becomes inherently safer and more adaptable to standard industrial reactors. Additionally, the synthesis employs a one-pot methodology for the final condensation step, where the hydrolysis of 5-nitrofurfural diethyl acetal and its subsequent reaction with the oxazolidinone intermediate occur in a unified sequence. This consolidation of steps drastically reduces solvent usage and processing time, while simultaneously boosting the overall yield to levels surpassing 30%. 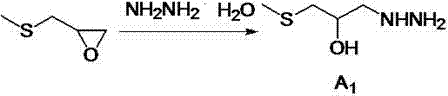
Mechanistic Insights into One-Pot Condensation and Cyclization
The core of this synthetic advancement relies on the precise orchestration of nucleophilic substitution and cyclization reactions under controlled thermal conditions. In the initial phase, methylthio propylene oxide undergoes a single substitution reaction with 80% hydrazine hydrate at temperatures ranging between 80°C and 85°C. The hydrazine hydrate serves a dual function here, acting both as a reactant and as the reaction solvent, which simplifies the stoichiometry and reduces the need for additional organic solvents. Following this, the intermediate undergoes a ring-closure reaction with diethyl carbonate in the presence of a strong base catalyst like sodium methylate. This cyclization is critical for forming the 2-oxazolidinone core structure, which is essential for the biological activity of the final drug. The mechanistic efficiency is further enhanced by the direct use of the reaction solution without intermediate isolation, preserving the reactive species for the subsequent condensation.
In the final stage, the mechanism involves the acid-catalyzed hydrolysis of 5-nitrofurfural diethyl acetal to generate the reactive aldehyde species in situ. This generated aldehyde immediately reacts with the amino group of the oxazolidinone intermediate to form the Schiff base linkage characteristic of Nifuratel. Conducting this reaction at mild temperatures between 20°C and 25°C prevents the degradation of the sensitive nitrofuran moiety, which is prone to decomposition under harsh acidic or thermal conditions. The use of dilute sulfuric acid in an ethanolic medium provides the optimal pH environment for hydrolysis while maintaining the solubility of the organic components. This careful balance of reaction parameters ensures that impurities are minimized, leading to the observed high purity profiles. 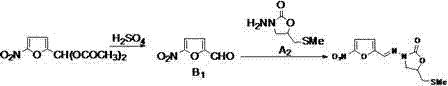
How to Synthesize Nifuratel Efficiently
Implementing this synthesis route requires strict adherence to temperature controls and reagent addition sequences to maximize yield and safety. The process begins with the gradual addition of methylthio propylene oxide to heated hydrazine hydrate, followed by a specific dehydration protocol using absolute ethanol to remove residual water without high vacuum stress. Subsequent steps involve the preparation of a sodium methylate catalyst solution and the controlled addition of diethyl carbonate to effect cyclization. The final convergence of the furan and oxazolidinone fragments is achieved through a carefully managed one-pot condensation, followed by recrystallization using acetic acid and ethanol to achieve pharmaceutical grade purity. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- React methylthio propylene oxide with 80% hydrazine hydrate at 80-85°C, followed by vacuum distillation and ethanol dehydration to obtain intermediate A1.
- Perform ring-closure reaction of intermediate A1 with diethyl carbonate in sodium methylate methanol solution at 60-70°C to form intermediate A2.
- Hydrolyze 5-nitrofurfural diethyl acetal with dilute sulfuric acid, then conduct a one-pot condensation with intermediate A2 at 20-25°C to yield crude Nifuratel.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages by decoupling production efficiency from complex, high-maintenance infrastructure. The elimination of high-vacuum distillation equipment translates directly into reduced capital expenditure for manufacturing facilities, allowing for more flexible allocation of resources towards capacity expansion or quality control systems. Moreover, the simplification of post-treatment operations reduces the labor intensity and technical expertise required on the production floor, thereby lowering operational overheads. The reliance on domestically available raw materials within major chemical manufacturing hubs ensures a stable supply chain that is less susceptible to geopolitical disruptions or logistics bottlenecks. This resilience is crucial for maintaining continuous supply to downstream pharmaceutical formulators who depend on consistent API availability.
- Cost Reduction in Manufacturing: The removal of energy-intensive high-vacuum steps and the consolidation of reaction vessels significantly lower utility costs and equipment depreciation. By avoiding the need for specialized vacuum pumps and complex sealing systems, the facility can operate with standard reactor setups, leading to drastic simplifications in the production line. The one-pot nature of the final step minimizes solvent consumption and waste generation, further contributing to substantial cost savings in raw material procurement and waste disposal. These cumulative efficiencies create a highly competitive cost structure that allows for better pricing flexibility in the global market.
- Enhanced Supply Chain Reliability: The use of common, commercially available reagents such as hydrazine hydrate and diethyl carbonate mitigates the risk of raw material shortages that often plague specialty chemical synthesis. The robustness of the reaction conditions, which tolerate standard industrial variations in temperature and pressure, ensures consistent batch-to-batch quality and reliable delivery schedules. This stability is paramount for long-term supply agreements, as it reduces the likelihood of production delays caused by equipment failure or difficult purification hurdles. Consequently, partners can rely on a steady flow of high-purity intermediates to support their own manufacturing timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that translate seamlessly from pilot plants to multi-ton commercial reactors. The reduction in hazardous waste streams, particularly through the avoidance of complex distillation residues, aligns with increasingly stringent environmental regulations and sustainability goals. Simplified workup procedures mean less solvent waste and lower emissions, facilitating easier compliance with local environmental protection standards. This eco-friendly profile not only reduces regulatory risk but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Nifuratel synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality assurances associated with this technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or procurement strategies.
Q: How does the new synthesis method improve upon traditional high vacuum distillation?
A: The novel method replaces difficult-to-control high vacuum distillation with an 'ethanol with water' post-treatment. This simplifies equipment requirements, improves yield stability, and enhances safety for industrial scale-up.
Q: What is the purity level achievable with this one-pot synthesis route?
A: The patented process consistently achieves a product purity higher than 99.8% after recrystallization and beating purification, meeting stringent pharmaceutical standards for active ingredients.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials, avoids complex purification steps, and employs simple reaction controls, making it highly conducive to industrialized production and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nifuratel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering Nifuratel and its intermediates with stringent purity specifications, supported by our rigorous QC labs that validate every batch against international pharmacopoeia standards. Our capability to implement the one-pot synthesis technology described in patent CN103232445A allows us to offer a superior product profile that combines high quality with supply chain resilience.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this process. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
