Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
The landscape of modern medicinal chemistry is increasingly defined by the strategic incorporation of fluorine atoms into heterocyclic scaffolds to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN111423381B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. These nitrogen-containing five-membered heterocycles are pivotal structural motifs found in numerous bioactive molecules, ranging from potent H1 histamine receptor antagonists like Alcaftadine to complex natural products such as Naamidine A. The introduction of the trifluoromethyl group specifically modulates the electronegativity and lipophilicity of the parent molecule, properties that are critical for optimizing drug candidates. As a leading entity in the fine chemical sector, understanding this patented methodology is essential for any reliable pharmaceutical intermediate supplier aiming to deliver high-value building blocks to the global market.
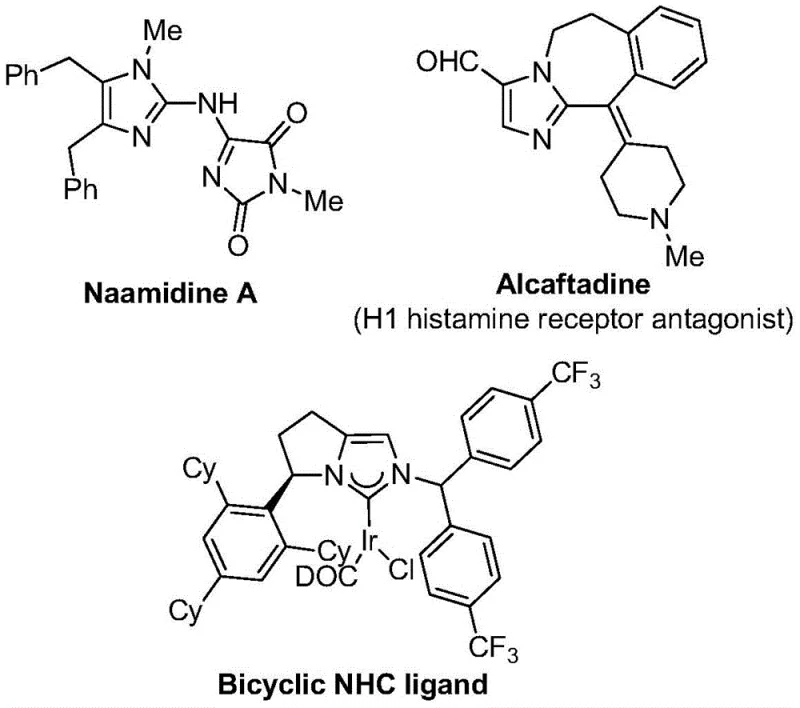
The significance of this technology extends beyond mere academic interest; it represents a tangible shift towards more sustainable and economically viable manufacturing processes. By leveraging a transition metal palladium-catalyzed carbonylation series reaction, this method circumvents the limitations of earlier synthetic routes. For R&D directors and process chemists, the ability to access these diversified trifluoromethylated imidazoles through a robust, one-pot protocol offers a streamlined pathway for library synthesis and lead optimization. The patent highlights that the reaction proceeds with high efficiency and good substrate compatibility, utilizing cheap and easily obtained starting materials. This aligns perfectly with the industry's drive towards cost reduction in API manufacturing, ensuring that complex heterocyclic cores can be produced without prohibitive expense or logistical bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with challenges that hinder large-scale adoption. Literature reports indicate that prevailing methods often rely on the direct reaction of a synthon containing a trifluoromethyl group with a suitable substrate, frequently utilizing reagents such as trifluorodiazoethane. While effective in small-scale laboratory settings, trifluorodiazoethane is known for its instability and potential safety hazards, posing significant risks during commercial scale-up of complex polymer additives or pharmaceutical ingredients. Furthermore, other commonly used synthons like trifluoroethylimidoyl halides have not been widely exploited due to perceived limitations in reactivity or availability. Traditional approaches often necessitate harsh reaction conditions, including elevated temperatures and pressures, which not only increase energy consumption but also limit the tolerance for sensitive functional groups. This lack of functional group compatibility restricts the chemical space accessible to medicinal chemists, forcing them to compromise on molecular design or engage in lengthy protection-deprotection sequences that erode overall yield and efficiency.
The Novel Approach
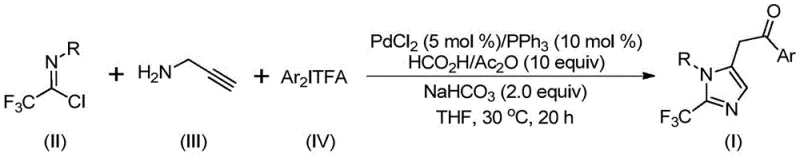
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN111423381B introduces a paradigm shift by employing a palladium-catalyzed multicomponent reaction. This novel approach utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the fundamental building blocks. The reaction is elegantly driven by a carbon monoxide substitute generated in situ from formic acid and acetic anhydride, eliminating the need for handling toxic CO gas directly. The entire transformation occurs under remarkably mild conditions, specifically at 30°C, which is a testament to the high catalytic activity of the system. This gentle thermal profile preserves the integrity of sensitive substituents, allowing for the successful incorporation of diverse functional groups such as nitro, halogen, and alkoxy moieties. The result is a versatile platform capable of synthesizing variously substituted imidazole compounds with trifluoromethyl groups at the 2-position, thereby broadening the utility of this method for creating high-purity OLED material precursors or specialized agrochemical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
For the discerning R&D Director, understanding the mechanistic underpinnings of this transformation is crucial for troubleshooting and further optimization. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the base, sodium bicarbonate, yielding a trifluoroacetamidine intermediate. This species then undergoes isomerization to set the stage for the metal-catalyzed cycle. The palladium catalyst, typically PdCl2 coordinated with triphenylphosphine, facilitates the activation of the alkyne moiety in the propargylamine derivative. Through a palladation event, an alkenyl palladium intermediate is generated, which subsequently isomerizes to a more stable alkyl palladium species. The critical carbonylation step follows, where the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond to form an acyl palladium intermediate. This acyl species then engages with the diaryl iodonium salt via oxidative addition, accessing a high-valent tetravalent palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst. This intricate dance of organometallic steps ensures high atom economy and selectivity.
From an impurity control perspective, the mechanism offers distinct advantages. The use of diaryl iodonium salts as arylating agents is particularly beneficial because they are highly reactive yet generate benign byproducts compared to traditional aryl halides which might require harsher conditions to activate. The mild temperature of 30°C significantly suppresses side reactions such as polymerization of the alkyne or decomposition of the trifluoromethyl group, which are common pitfalls in high-temperature heterocycle synthesis. Furthermore, the specific stoichiometry recommended in the patent, with a molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate of 0.05:0.1:2, creates an optimized environment that minimizes the formation of homocoupling byproducts. The post-treatment procedure, involving simple filtration followed by silica gel mixing and column chromatography, effectively removes palladium residues and inorganic salts, ensuring the delivery of high-purity pharmaceutical intermediates that meet stringent regulatory specifications for residual metals.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a practical setting requires adherence to the specific protocols outlined in the patent to maximize yield and reproducibility. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. By combining the palladium catalyst system with the specific trio of reactants in a solvent like tetrahydrofuran (THF), manufacturers can achieve conversions that are both rapid and complete within a standard workday. The detailed standardized synthesis steps below provide a clear roadmap for replicating the high yields (up to 97% in optimized examples) reported in the intellectual property documentation. This level of efficiency is critical for reducing lead time for high-purity pharmaceutical intermediates in a fast-paced drug development environment.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the final 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented technology are profound. The shift towards this specific catalytic manifold addresses several chronic pain points in the sourcing of complex heterocyclic building blocks. By transitioning to a process that relies on commodity chemicals rather than exotic, custom-synthetized reagents, organizations can secure a more resilient supply chain. The stability of the starting materials, such as trifluoroethylimidoyl chloride and propargylamine, ensures that inventory can be managed effectively without the degradation issues associated with unstable diazo compounds. This reliability translates directly into reduced risk of production delays and ensures continuity of supply for downstream API manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the utilization of inexpensive and widely available raw materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from corresponding aromatic amines, which are bulk commodities, while diaryl iodonium salts can be prepared simply from aryl boronic acids and aryl iodides. The elimination of expensive and hazardous reagents like trifluorodiazoethane removes the need for specialized containment infrastructure and safety protocols, leading to substantial cost savings. Additionally, the low catalyst loading (5 mol% PdCl2) and the use of common solvents like THF further drive down the operational expenditure, making the cost reduction in electronic chemical manufacturing or pharma intermediate production significant and sustainable.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain stability. Because the reaction proceeds efficiently at 30°C, it does not require energy-intensive heating or cooling systems, reducing the facility's dependence on complex utility infrastructure. The broad substrate scope means that a single manufacturing platform can produce a wide variety of derivatives simply by swapping the aryl groups on the starting materials. This flexibility allows suppliers to respond rapidly to changing customer demands without retooling entire production lines. The ability to scale this reaction from gram levels to industrial quantities ensures that as a drug candidate moves from clinical trials to commercial launch, the supply of the key intermediate can grow seamlessly to meet demand.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns with green chemistry principles. The use of formic acid and acetic anhydride as a carbon monoxide surrogate avoids the handling of toxic CO gas cylinders, significantly improving workplace safety and simplifying regulatory compliance. The post-treatment involves straightforward filtration and chromatography, which generates less hazardous waste compared to quenching reactive organometallic reagents. The high reaction efficiency and yield minimize the amount of unreacted starting material that needs to be recovered or disposed of. This streamlined workflow facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining a smaller environmental footprint, a key metric for modern sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is vital for making informed decisions about process integration and vendor selection.
Q: What are the advantages of this palladium-catalyzed method over traditional trifluoromethylation techniques?
A: Unlike traditional methods that often rely on hazardous or expensive reagents like trifluorodiazoethane, this novel approach utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine. Furthermore, it operates under exceptionally mild conditions (30°C) compared to the harsh thermal requirements of conventional syntheses, significantly enhancing safety and operational simplicity.
Q: Does this synthesis method support a wide range of substrate functional groups?
A: Yes, the method demonstrates excellent substrate compatibility. It tolerates various substituents on the aryl rings, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl groups. This versatility allows for the design and synthesis of diverse imidazole derivatives tailored for specific drug discovery programs.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The patent explicitly states that the method is scalable to the gram level and provides a strong possibility for industrial large-scale production. The use of inexpensive catalysts (PdCl2/PPh3), common solvents (THF), and a simple post-treatment procedure involving filtration and chromatography makes it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing palladium-catalyzed reactions to ensure maximum efficiency and minimal metal residue, adhering to stringent purity specifications required by top-tier pharmaceutical clients. With our rigorous QC labs and state-of-the-art manufacturing facilities, we are uniquely positioned to deliver high-purity pharmaceutical intermediates that meet the exacting standards of the international market.
We invite you to leverage our expertise to accelerate your drug development timelines. Whether you require custom synthesis of novel trifluoromethyl imidazole derivatives or scale-up of existing routes, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
