Scalable Synthesis of 2-Trifluoromethyl Imidazoles via Mild Palladium-Catalyzed Carbonylation for Commercial API Production
Scalable Synthesis of 2-Trifluoromethyl Imidazoles via Mild Palladium-Catalyzed Carbonylation for Commercial API Production
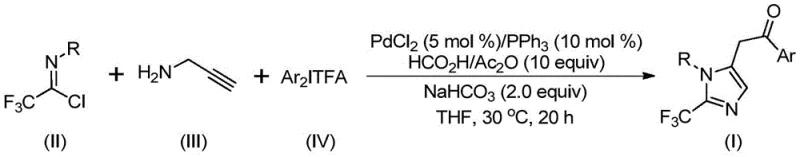
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical bottlenecks in current medicinal chemistry workflows. This technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing trifluoroacetimidoyl chloride, propargylamine, and diaryl iodonium salts as key building blocks. Unlike traditional methods that often require extreme temperatures or toxic reagents, this novel approach operates efficiently at a mild 30°C, demonstrating exceptional substrate compatibility and high reaction yields. For R&D directors and procurement managers alike, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with improved cost-efficiency and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with challenges that hinder large-scale commercialization. Conventional pathways often rely on direct reactions between trifluoromethyl synthons and suitable substrates, which frequently necessitate harsh reaction conditions, such as elevated temperatures or strong acidic environments that degrade sensitive functional groups. Furthermore, many existing methods suffer from poor atom economy and limited substrate scope, restricting the diversity of molecules that can be accessed for drug discovery campaigns. The reliance on unstable or expensive trifluoromethyl sources also introduces significant variability in batch-to-batch consistency, complicating the regulatory approval process for active pharmaceutical ingredients. These inefficiencies translate directly into higher manufacturing costs and extended lead times, creating a substantial burden for supply chain heads managing complex API portfolios.
The Novel Approach
The methodology outlined in the patent revolutionizes this landscape by introducing a mild, multi-component coupling strategy that bypasses the limitations of prior art. By employing cheap and easily obtained starting materials—specifically trifluoroacetimidoyl chloride, propargylamine, and diaryl iodonium salts—the process achieves high conversion rates under remarkably gentle conditions. The reaction proceeds at 30°C in common organic solvents like tetrahydrofuran, eliminating the need for energy-intensive heating protocols. This approach not only simplifies the operational workflow but also dramatically expands the utility of the method, allowing for the design and synthesis of diversified imidazole compounds with various substitutions at the 1 and 5 positions. The ability to tolerate a wide range of functional groups, including halogens, nitro groups, and alkyl chains, makes this a versatile platform for generating libraries of bioactive molecules.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific targets. The reaction initiates with the formation of a trifluoroacetamidine compound through an intermolecular carbon-nitrogen bond promoted by the alkaline additive, sodium bicarbonate. Following this initial condensation, an isomerization occurs, setting the stage for the palladium catalytic cycle. The palladium catalyst, generated in situ from palladium chloride and triphenylphosphine, facilitates the palladation of the alkyne moiety in the propargylamine derivative, yielding a key alkenyl palladium intermediate. This intermediate subsequently undergoes isomerization to form an alkyl palladium species, which is primed for the critical carbonylation step.
The source of the carbonyl group is elegantly managed through the decomposition of formic acid and acetic anhydride, which releases carbon monoxide in situ, thereby avoiding the handling of hazardous CO gas cylinders. This acyl palladium intermediate then reacts with the diaryl iodonium salt via oxidative addition to generate a tetravalent palladium species. The cycle concludes with a reductive elimination step that forge the final carbon-carbon bond, releasing the desired 2-trifluoromethyl-substituted imidazole product and regenerating the active palladium catalyst. This intricate dance of organometallic steps ensures high selectivity and minimizes the formation of unwanted byproducts, resulting in a clean impurity profile that is highly desirable for pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield. The protocol involves mixing the palladium catalyst system with the three primary substrates in an aprotic solvent, followed by a controlled reaction period. The simplicity of the workup procedure, involving filtration and standard column chromatography, further enhances its appeal for rapid iteration during process development. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroacetimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture.
- Stir the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift towards milder reaction conditions and commercially available starting materials fundamentally alters the cost structure of producing these valuable heterocyclic intermediates. By eliminating the need for specialized high-pressure equipment or exotic reagents, manufacturers can significantly reduce capital expenditure and operational overhead. Furthermore, the robustness of the reaction across a wide variety of substrates ensures consistent output quality, reducing the risk of batch failures that can disrupt downstream production schedules.
- Cost Reduction in Manufacturing: The utilization of inexpensive and widely available raw materials, such as propargylamine and aromatic amines used to prepare the imidoyl chloride, drives down the direct material costs associated with production. Additionally, the use of formic acid and acetic anhydride as a safe, in situ source of carbon monoxide removes the logistical and safety costs associated with storing and handling high-pressure carbon monoxide gas. The mild reaction temperature of 30°C also translates to substantial energy savings compared to processes requiring reflux or cryogenic conditions, contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: Because the key starting materials are commodity chemicals or easily synthesized from common precursors, the supply chain for this process is inherently more resilient against market fluctuations. The high substrate compatibility means that a single manufacturing line can be adapted to produce a diverse range of derivatives without extensive retooling, providing flexibility to meet changing customer demands. This adaptability reduces lead times for high-purity pharmaceutical intermediates, allowing partners to respond more agilely to clinical trial requirements or market launches.
- Scalability and Environmental Compliance: The patent explicitly notes the potential for industrial large-scale production, supported by the simplicity of the post-treatment process which avoids complex purification steps. The reaction generates minimal hazardous waste, and the avoidance of toxic heavy metals beyond the catalytic amount of palladium (which can be recovered) aligns with increasingly stringent environmental regulations. This green chemistry profile facilitates easier regulatory approval and supports corporate sustainability goals, making it an attractive option for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route.
Q: What are the key advantages of this palladium-catalyzed method over traditional imidazole synthesis?
A: This method operates at a mild temperature of 30°C, utilizes cheap and readily available starting materials like propargylamine, and demonstrates excellent functional group tolerance, allowing for the synthesis of diverse derivatives without harsh conditions.
Q: Can this process be scaled for industrial manufacturing of API intermediates?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides the possibility for industrial large-scale production due to its simple operation, high reaction efficiency, and use of stable catalysts.
Q: What is the role of the diaryl iodonium salt in this reaction mechanism?
A: The diaryl iodonium salt acts as an arylating agent that undergoes oxidative addition to the palladium center, facilitating the formation of the final carbon-carbon bond and completing the catalytic cycle to yield the 5-aryl substituted imidazole product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN111423381B for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole intermediate meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for success.
