Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical limitations in existing synthetic routes. This technology leverages a unique molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this scaffold cannot be overstated, as evidenced by its presence in major therapeutic agents such as Sitagliptin and various anticonvulsant drugs, where the trifluoromethyl group plays a pivotal role in modulating lipophilicity and binding affinity.
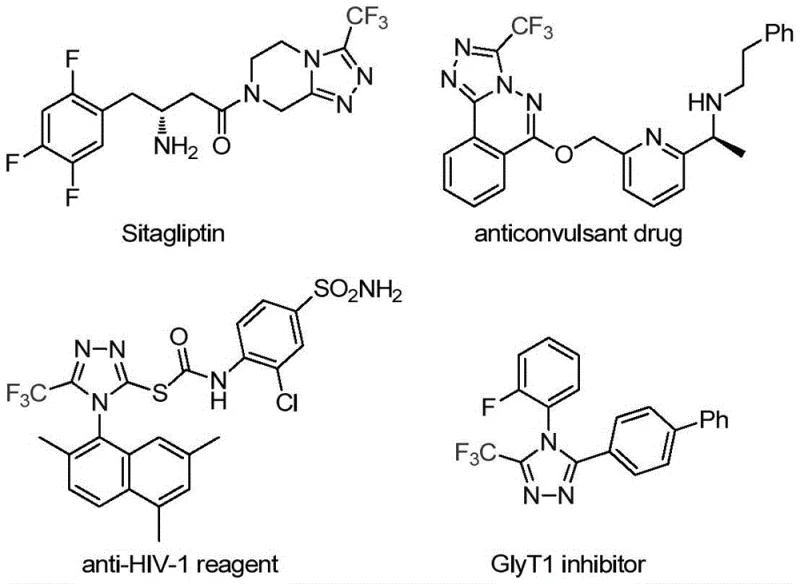
For R&D directors and process chemists, the ability to access these privileged structures efficiently is paramount. The disclosed method operates under remarkably mild conditions, typically between 70°C and 90°C, utilizing tetrahydrofuran (THF) as the preferred solvent. This represents a significant departure from classical approaches that often demand extreme temperatures or the handling of unstable intermediates. By establishing a reliable protocol that tolerates a wide array of functional groups—including halogens, alkyls, and electron-withdrawing nitro groups—this patent provides a versatile platform for the rapid generation of diverse chemical libraries essential for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale manufacturing. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These pathways frequently suffer from poor atom economy and require the use of hazardous hydrazine derivatives, which pose significant safety risks in an industrial setting due to their potential explosivity and toxicity. Furthermore, alternative multi-component reactions involving diazonium salts and trifluorodiazoethane often necessitate stringent anhydrous conditions and complex purification procedures to remove metal residues, thereby inflating the cost of goods sold (COGS) and complicating supply chain logistics for high-purity API intermediates.
The Novel Approach
The methodology described in patent CN113307778A offers a transformative solution by employing a dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate. This innovative route bypasses the need for dangerous hydrazine precursors, instead utilizing stable and commercially accessible trifluoroethylimidoyl chloride and functionalized isonitriles. The reaction proceeds via a streamlined one-pot process where the catalysts activate the substrates for efficient ring closure. This not only simplifies the operational workflow but also drastically reduces the generation of hazardous waste. The ability to achieve high conversion rates with excellent yields across a broad spectrum of substrates demonstrates the robustness of this chemistry, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where purity and safety are non-negotiable.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent quality. The reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex. Simultaneously, cuprous acetate acts as a promoter, facilitating the [3+2] cycloaddition between the activated isonitrile and the trifluoroethylimidoyl chloride. This synergistic interaction lowers the activation energy barrier, allowing the formation of the five-membered triazole ring to proceed smoothly at moderate temperatures. The presence of triethylamine serves as a base to neutralize acidic byproducts, driving the equilibrium towards product formation while maintaining the integrity of the catalytic cycle.
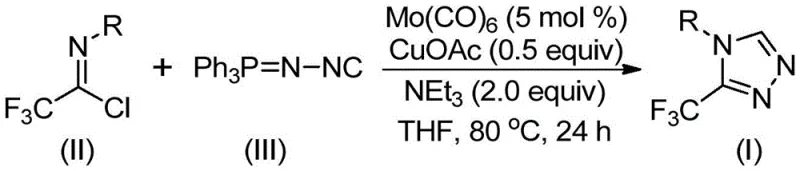
Following the cycloaddition, the intermediate undergoes a hydrolysis-like elimination of triphenylphosphine oxide, a step that is critical for aromatization and final product stability. This mechanism explains the high tolerance for various substituents on the aromatic ring, as the electronic properties of the R-group do not significantly impede the initial coordination steps. For quality control teams, this mechanistic clarity allows for better prediction of impurity profiles. By controlling the stoichiometry of the catalysts—specifically maintaining a molar ratio of approximately 1:1.5:0.05:0.5:2 for the imidoyl chloride, isonitrile, Mo catalyst, Cu catalyst, and base respectively—manufacturers can minimize side reactions and ensure the production of high-purity 3-trifluoromethyl-1,2,4-triazoles suitable for sensitive downstream applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis requires careful attention to reagent quality and reaction monitoring to maximize yield. The protocol outlined in the patent is designed for simplicity, utilizing standard laboratory equipment such as Schlenk tubes for small-scale optimization before transitioning to larger reactors. The use of molecular sieves is recommended to maintain anhydrous conditions, which protects the moisture-sensitive catalysts and prevents hydrolysis of the imidoyl chloride prior to reaction. Detailed standardized synthesis steps follow below to guide process engineers in replicating these results.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C and stir for 18-30 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this novel synthetic route offers substantial strategic benefits regarding raw material sourcing and inventory management. The starting materials, specifically trifluoroethylimidoyl chloride and functionalized isonitriles, are derived from widely available commodity chemicals, reducing dependency on niche suppliers who might dictate volatile pricing structures. This accessibility ensures a more resilient supply chain, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of expensive and difficult-to-handle reagents like diazonium salts translates directly into lower operational expenditures, allowing for more competitive pricing models in the global marketplace for fine chemical intermediates.
- Cost Reduction in Manufacturing: The adoption of this Mo/Cu co-catalyzed method eliminates the need for costly purification steps associated with removing heavy metal contaminants often found in traditional palladium or rhodium-catalyzed processes. Since cuprous acetate and molybdenum hexacarbonyl are relatively inexpensive and used in catalytic amounts, the overall catalyst loading cost is significantly minimized. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature reflux methods, contributing to a leaner manufacturing budget and improved margin potential for bulk orders of these specialized heterocycles.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or pressure, which enhances batch-to-b consistency. This reliability is critical for supply chain heads who must guarantee uninterrupted delivery to downstream API manufacturers. The ability to source generic solvents like THF and common bases like triethylamine further simplifies logistics, as these materials are stocked by nearly all major chemical distributors worldwide, ensuring that production timelines remain unaffected by regional supply disruptions.
- Scalability and Environmental Compliance: The patent data indicates that the reaction can be successfully expanded from milligram scales to gram-level preparations without loss of efficiency, suggesting a clear path to kilogram and metric-ton scale-up. This scalability is accompanied by a favorable environmental profile; the avoidance of explosive hydrazines and the use of less toxic solvents align with modern green chemistry principles. Consequently, waste treatment costs are reduced, and regulatory compliance becomes more straightforward, facilitating faster approval for commercial production facilities in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this synthesis route for their specific projects.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require hazardous hydrazine derivatives or harsh cyclization conditions, this patented approach utilizes mild temperatures (70-90°C) and commercially available starting materials, significantly improving operational safety and substrate tolerance.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the reaction efficiency remains high and the procedure is simple enough to be expanded from milligram scale to gram-level reactions, providing a viable pathway for subsequent metric-ton scale production.
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method exhibits broad substrate tolerance, successfully accommodating various aryl groups including phenyl, naphthyl, and phenethyl, as well as substituents like methyl, methoxy, fluoro, chloro, and nitro groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry with precision, delivering materials that accelerate your drug development timelines.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your specific pipeline needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that our capabilities align perfectly with your project goals for high-purity pharmaceutical intermediates.
