Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as a Sustainable Carbon Source
Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as a Sustainable Carbon Source
The pharmaceutical and fine chemical industries are constantly seeking sustainable, cost-effective routes to access privileged heterocyclic scaffolds, particularly those containing fluorine motifs which are critical for modulating metabolic stability and bioavailability. A groundbreaking approach detailed in Chinese Patent CN113880781A introduces a novel methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds by utilizing glucose as a renewable carbon source. This innovation represents a paradigm shift from traditional petrochemical-dependent pathways to biomass-utilizing cascades, addressing both economic and environmental pressures faced by modern chemical manufacturing. The significance of the 1,2,4-triazole core cannot be overstated, as it serves as a fundamental building block for a vast array of bioactive molecules, including antifungal agents, anti-inflammatory drugs, and kinase inhibitors. By leveraging the natural abundance of glucose, this process not only lowers the barrier to entry for producing these high-value intermediates but also aligns with the global push towards green chemistry principles in API production.
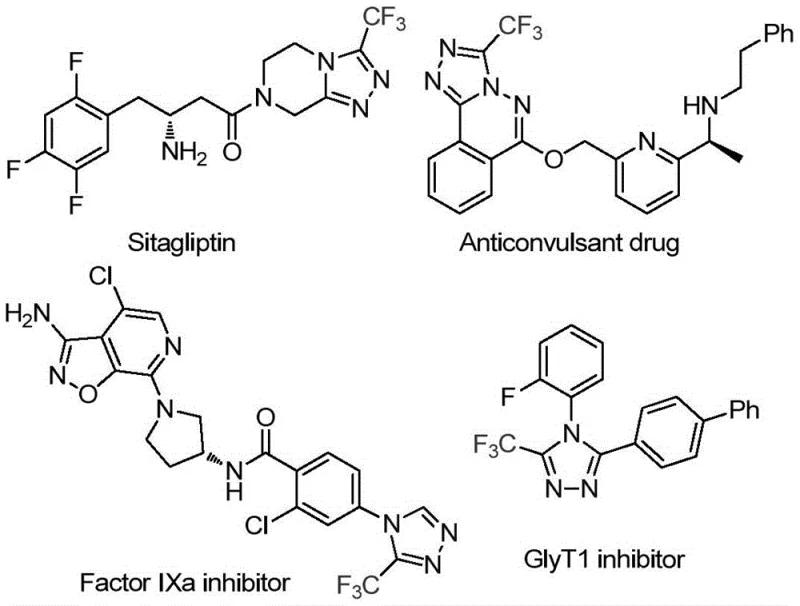
The strategic importance of this synthetic route is further highlighted by the prevalence of the triazole motif in commercial drugs. As illustrated in the structural diversity of known pharmaceuticals, the ability to efficiently install a trifluoromethyl group at the 3-position of the triazole ring offers medicinal chemists a powerful tool for optimizing drug candidates. The trifluoromethyl group is renowned for its ability to enhance lipophilicity and block metabolic hotspots, thereby extending the half-life of therapeutic agents. Consequently, developing a reliable pharmaceutical intermediate supplier capability for these specific scaffolds is of paramount interest to R&D directors looking to accelerate their pipeline development. The method described in the patent provides a direct, one-pot solution that bypasses multi-step sequences typically required to construct this heterocyclic system, thereby reducing waste generation and processing time significantly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied heavily on the condensation of amidrazones with carboxylic acid derivatives or the cyclization of hydrazones with nitriles under harsh conditions. These conventional protocols often necessitate the use of expensive, moisture-sensitive reagents and require stringent anhydrous and anaerobic conditions to prevent side reactions or reagent decomposition. Furthermore, the starting materials, such as specific trifluoroacetyl derivatives or activated nitriles, are frequently derived from non-renewable petrochemical sources and can be cost-prohibitive for large-scale manufacturing. The purification processes associated with these traditional methods are often cumbersome, involving complex workups to remove heavy metal catalysts or toxic byproducts, which poses significant challenges for supply chain heads concerned with environmental compliance and waste disposal costs. Additionally, the limited substrate scope of many classical methods restricts the ability to introduce diverse functional groups, hindering the rapid exploration of structure-activity relationships during the drug discovery phase.
The Novel Approach
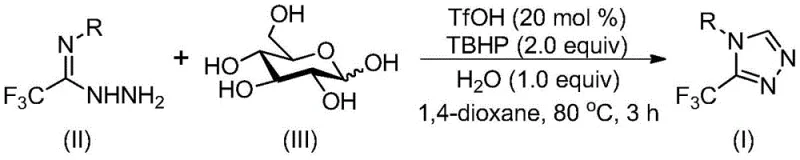
In stark contrast, the methodology disclosed in patent CN113880781A utilizes a biomimetic cascade strategy that transforms simple, abundant glucose into the requisite aldehyde intermediate in situ. This novel approach employs trifluoromethanesulfonic acid (TfOH) as a potent organocatalyst to promote the cleavage of glucose, followed by condensation with trifluoroacetimidoyl hydrazide. The reaction proceeds through an intramolecular nucleophilic addition and subsequent oxidative aromatization using tert-butyl hydroperoxide (TBHP) as a terminal oxidant. Crucially, this process operates under mild conditions (70-90°C) and tolerates the presence of water, which is actually beneficial for the reaction efficiency rather than detrimental. This tolerance eliminates the need for expensive drying agents and inert atmosphere equipment, drastically simplifying the operational complexity. The use of 1,4-dioxane as a solvent ensures good solubility for both the polar sugar and the organic substrates, facilitating a homogeneous reaction environment that enhances mass transfer and overall conversion rates.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
The mechanistic pathway of this transformation is a sophisticated example of acid-catalyzed cascade chemistry that maximizes atom economy. Initially, the strong Brønsted acid catalyst, trifluoromethanesulfonic acid, activates the glucose molecule, promoting its fragmentation to generate reactive aldehyde species. These aldehydes immediately undergo condensation with the trifluoroacetimidoyl hydrazide to form a hydrazone intermediate. This step is critical as it sets the stage for the subsequent ring closure. Following hydrazone formation, an intramolecular nucleophilic attack occurs where the nitrogen atom attacks the electrophilic carbon, leading to the formation of the dihydro-triazole ring system. The final step involves the oxidation of this dihydro-intermediate by TBHP, which restores aromaticity to the triazole ring, yielding the stable 3-trifluoromethyl-1,2,4-triazole product. This seamless integration of bond-breaking (glucose cleavage) and bond-forming (cyclization and oxidation) steps in a single pot minimizes the isolation of unstable intermediates and reduces the overall process mass intensity.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. Because the reactive aldehyde is generated in situ and consumed immediately by the hydrazide, the concentration of free aldehyde remains low, thereby suppressing potential polymerization or self-condensation side reactions that often plague glucose-based chemistry. Furthermore, the oxidative aromatization step driven by TBHP is highly selective for the dihydro-triazole intermediate, ensuring that the final product is formed with high purity. The compatibility of this system with various substituents on the hydrazide nitrogen (R group) allows for the synthesis of a wide library of derivatives without compromising the integrity of sensitive functional groups. For R&D directors, understanding this mechanism is vital for troubleshooting and optimizing the process for specific target molecules, as it highlights the delicate balance between acid strength, oxidant loading, and temperature required to drive the cascade to completion efficiently.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly attractive for both laboratory scale-up and commercial production. The protocol involves charging a reactor with the trifluoroacetimidoyl hydrazide, glucose, catalytic amount of trifluoromethanesulfonic acid, and aqueous TBHP in 1,4-dioxane. The mixture is then heated to 80°C for approximately 3 hours, after which the reaction is quenched and worked up. The simplicity of the workup, involving filtration and standard column chromatography, ensures that high-purity products can be obtained with minimal effort. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid catalyst, and tert-butyl hydroperoxide oxidant in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 70-90°C and stir for 2-4 hours to facilitate the cascade cyclization and aromatization.
- Upon completion, filter the mixture and purify the crude product via silica gel column chromatography to isolate the high-purity triazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this glucose-based synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver for cost reduction is the substitution of expensive, specialized aldehyde precursors with glucose, a commodity chemical available in massive quantities globally at a fraction of the cost. This shift not only lowers the raw material bill of materials but also insulates the supply chain from the volatility associated with petrochemical pricing. Moreover, the elimination of strict anhydrous and anaerobic requirements translates to significant capital expenditure savings, as manufacturers do not need to invest in specialized dry rooms or inert gas manifolds, allowing existing general-purpose reactors to be utilized for production.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of biomass feedstocks and the avoidance of precious metal catalysts. By replacing costly transition metal catalysts with an inexpensive organo-acid (TfOH), the process eliminates the need for expensive metal scavenging steps and the associated regulatory testing for residual metals in the final API. This reduction in downstream processing complexity leads to substantial cost savings in terms of both consumables and labor hours. Additionally, the high atom economy of the cascade reaction means less waste is generated per kilogram of product, further reducing waste disposal fees and improving the overall process mass intensity metrics.
- Enhanced Supply Chain Reliability: Sourcing reliability is dramatically improved because glucose is a universally available agricultural product with a stable and robust supply network, unlike many specialized fine chemical intermediates that may be sourced from single suppliers. The robustness of the reaction conditions, specifically the tolerance to water and air, reduces the risk of batch failures due to environmental excursions, ensuring consistent delivery schedules. This reliability is crucial for maintaining continuous manufacturing operations and meeting the just-in-time delivery expectations of downstream pharmaceutical clients who depend on a steady flow of high-quality intermediates for their own production lines.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration at the gram level with potential for ton-scale expansion, facilitated by the use of common solvents and safe oxidants. From an environmental standpoint, the use of TBHP as an oxidant generates tert-butanol as a byproduct, which is relatively benign and easier to handle than heavy metal waste streams. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the facility more attractive to eco-conscious investors and partners. The simplified waste profile reduces the burden on effluent treatment plants and lowers the overall environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-mediated triazole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using glucose in this synthesis?
A: Using glucose eliminates the need for expensive, pre-functionalized aldehyde starting materials. As a widely available biomass feedstock, glucose significantly reduces raw material costs and simplifies the supply chain compared to traditional petrochemical-derived synthons.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key innovations of patent CN113880781A is that the reaction proceeds efficiently without the need for rigorous anhydrous or oxygen-free environments. The addition of water actually promotes the reaction efficiency, making the process much more robust for industrial scale-up.
Q: What is the typical yield range for this trifluoromethyl triazole synthesis?
A: The patent data demonstrates high reaction efficiency with yields varying based on the substrate. For example, naphthyl-substituted derivatives achieved yields as high as 99%, while other aryl substitutions ranged effectively between 36% and 84%, proving the method's versatility across different functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biomass-derived synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like the glucose-based triazole synthesis are translated into robust, GMP-compliant manufacturing processes. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can accelerate your drug development timeline while optimizing your supply chain economics.
