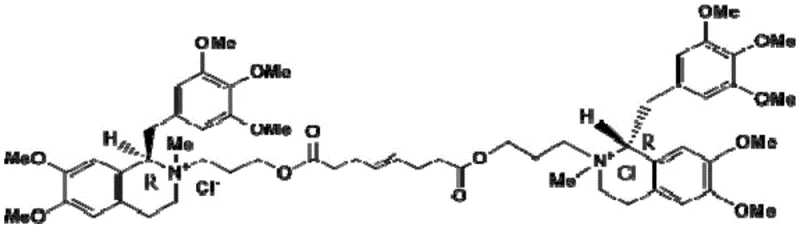
Advanced Synthesis of Mivacurium Chloride Intermediate for Commercial Scale-Up
Advanced Synthesis of Mivacurium Chloride Intermediate for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for producing complex muscle relaxant intermediates. A pivotal advancement in this domain is detailed in patent CN114057642B, which discloses a highly efficient synthetic method for the key intermediate of Mivacurium Chloride, specifically 6,7-dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline. This innovation addresses long-standing challenges associated with the thermal instability of precursors and the hazardous nature of traditional cyclization reagents. By shifting from harsh high-temperature reflux conditions to a mild, controlled low-temperature protocol, this technology offers a transformative approach for reliable pharmaceutical intermediate suppliers aiming to enhance production safety and product quality. The strategic implementation of 1-(3-dimethylaminopropyl)-3-(ethylcarbodiimide hydrochloride) (EDCI) as a dehydrating agent, coupled with trifluoromethanesulfonic anhydride for cyclization, represents a significant leap forward in process chemistry optimization.
The Limitations of Conventional Methods vs. The Novel Approach
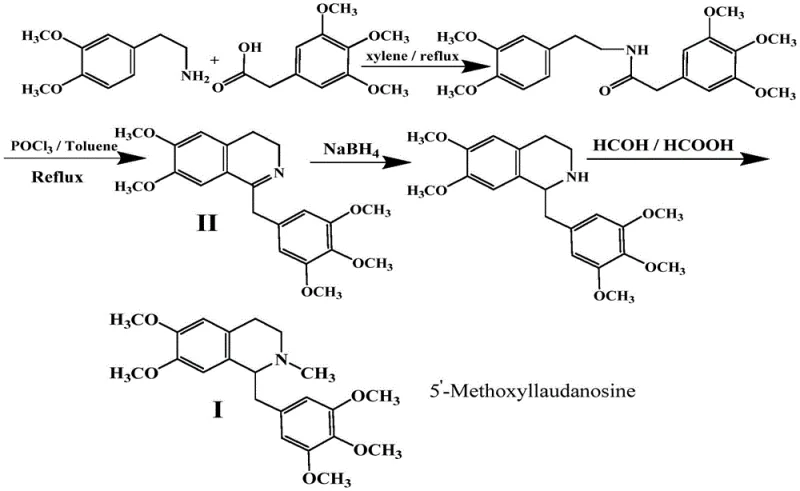
The Limitations of Conventional Methods
Historically, the synthesis of benzylisoquinoline derivatives like the Mivacurium intermediate has relied heavily on aggressive reaction conditions that pose substantial risks to both operational safety and product integrity. Traditional protocols, such as those referenced in earlier patents like CN1203061C, typically involve the reflux dehydration of phosphorus oxychloride in toxic solvents like toluene or xylene at elevated temperatures often exceeding 140°C. These high-thermal energy inputs are detrimental because the primary amine precursor, 3,4-dimethoxyphenethylamine, is inherently unstable and prone to degradation under such harsh thermal stress, leading to the formation of difficult-to-remove impurities. Furthermore, the use of phosphorus oxychloride generates hydrogen chloride gas during the reaction, which causes severe corrosion to standard stainless steel reactor equipment and necessitates expensive scrubbing systems to mitigate environmental pollution. The reliance on flammable, explosive, and toxic solvents like xylene further complicates the safety profile, requiring rigorous fire and explosion prevention measures that drive up capital expenditure and operational complexity for manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data utilizes a sophisticated two-step sequence that operates entirely within a mild temperature window of 0-30°C. This paradigm shift eliminates the need for high-energy reflux, thereby preserving the structural integrity of the sensitive 3,4-dimethoxyphenethylamine starting material and preventing thermal degradation pathways. The first step employs EDCI as a highly efficient dehydrating agent to couple 3,4,5-trimethoxyphenylacetic acid with the amine, forming the requisite amide bond without the generation of corrosive byproducts. The subsequent cyclization step leverages trifluoromethanesulfonic anhydride and 2-fluoropyridine to close the isoquinoline ring under similarly mild conditions. This methodology not only drastically simplifies the equipment requirements by removing the need for specialized high-temperature reflux setups but also significantly enhances the environmental profile of the process by avoiding the release of hydrogen chloride gas and minimizing the use of hazardous volatile organic compounds.
Mechanistic Insights into EDCI-Mediated Coupling and Triflic Anhydride Cyclization
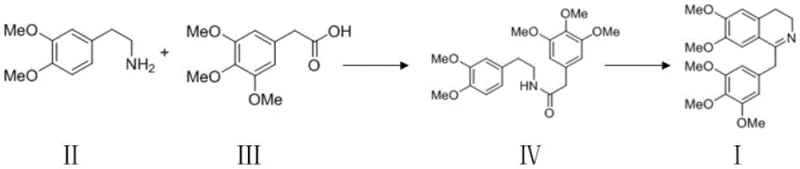
The core of this technological breakthrough lies in the precise mechanistic control exerted during the amide formation and subsequent ring closure. In the initial dehydration step, EDCI activates the carboxylic acid group of 3,4,5-trimethoxyphenylacetic acid to form an O-acylisourea intermediate, which is highly susceptible to nucleophilic attack by the amine group of 3,4-dimethoxyphenethylamine. This activation occurs efficiently at low temperatures (0-5°C initially, warming to 24-30°C), ensuring that the reaction kinetics favor amide bond formation over side reactions that might occur at higher thermal energies. The careful stoichiometric balance, optimized at a molar ratio of approximately 1:1.2 for acid to amine, ensures complete conversion of the acid while minimizing excess reagent waste, which is critical for maintaining high purity in the crude product before the cyclization step even begins.
Following amide formation, the cyclization mechanism involves the activation of the amide oxygen by trifluoromethanesulfonic anhydride (Tf2O) in the presence of 2-fluoropyridine as a base. This generates a highly reactive imidate species that undergoes an intramolecular electrophilic aromatic substitution to close the dihydroisoquinoline ring. The use of 2-fluoropyridine is crucial as it acts as a non-nucleophilic base to scavenge the triflic acid generated during the activation, preventing protonation of the amine which would inhibit the cyclization. Operating this step at 0-30°C prevents the decomposition of the activated intermediate and suppresses polymerization or oligomerization side reactions that are common in high-temperature Friedel-Crafts type cyclizations. This precise control over the electronic environment of the reaction mixture is what enables the achievement of exceptional purity levels, reported as high as 99.88%, which is vital for meeting the stringent specifications required for pharmaceutical active ingredients.
How to Synthesize 6,7-Dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and stoichiometric ratios defined in the patent to maximize yield and purity. The process begins with the dissolution of the acid and amine in a suitable solvent such as dichloromethane or DMF, followed by the controlled addition of EDCI under ice-water cooling to manage the exotherm. Once the amide is formed and isolated, it is redissolved in dry dichloromethane under nitrogen protection for the cyclization step, where Tf2O and 2-fluoropyridine are added sequentially at low temperature. The detailed standardized synthesis steps, including specific workup procedures like aqueous washes and column chromatography purification using a methanol/dichloromethane system, are critical for removing trace impurities and achieving the reported >91% total yield.
- Dehydrate 3,4,5-trimethoxyphenylacetic acid and 3,4-dimethoxyphenethylamine using EDCI at 0-30°C to form the amide intermediate.
- Cyclize the resulting amide using trifluoromethanesulfonic anhydride and 2-fluoropyridine at 0-30°C to obtain the dihydroisoquinoline core.
- Purify the final product via column chromatography using a methanol/dichloromethane eluent system to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of high-temperature reflux and corrosive reagents like phosphorus oxychloride significantly reduces the maintenance burden on production equipment, extending the lifecycle of reactors and piping systems while lowering the frequency of costly shutdowns for repairs. Furthermore, the avoidance of toxic solvents like xylene and toluene simplifies waste management protocols and reduces the regulatory compliance costs associated with handling hazardous materials, thereby streamlining the overall manufacturing workflow. The mild reaction conditions also imply a lower energy footprint, as there is no need for sustained heating or complex cooling systems beyond standard chillers, contributing to a more sustainable and cost-effective production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The shift to a low-temperature process eliminates the need for expensive high-grade alloys resistant to hydrogen chloride corrosion, allowing for the use of standard glass-lined or stainless steel equipment which significantly lowers capital investment. Additionally, the high selectivity of the EDCI and Tf2O reagents minimizes the formation of byproducts, reducing the load on downstream purification units and increasing the overall throughput of the facility without requiring additional separation columns or extensive recrystallization steps.
- Enhanced Supply Chain Reliability: By avoiding the use of restricted toxic solvents and hazardous reagents that often face supply volatility due to environmental regulations, this method ensures a more stable and continuous supply of raw materials. The robustness of the reaction at ambient to low temperatures also reduces the risk of batch failures due to thermal runaway or equipment malfunction, guaranteeing consistent delivery schedules for downstream API manufacturers who rely on just-in-time inventory models for their production lines.
- Scalability and Environmental Compliance: The simplicity of the operation, which does not require complex pressure vessels or specialized high-temperature reflux condensers, makes this process inherently easier to scale from pilot plant to commercial tonnage production. The absence of HCl gas emissions and the reduction in volatile organic compound usage facilitate easier compliance with increasingly strict environmental protection laws, ensuring long-term operational viability and reducing the risk of regulatory fines or production halts due to non-compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis pathway. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield optimization, safety parameters, and purity standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the critical temperature controls in the new synthesis route?
A: The process strictly maintains temperatures between 0-30°C for both the dehydration and cyclization steps to prevent degradation of the unstable 3,4-dimethoxyphenethylamine raw material.
Q: How does this method improve upon traditional POCl3 reflux methods?
A: Unlike traditional methods requiring high-temperature reflux with toxic solvents like xylene and corrosive phosphorus oxychloride, this route operates at mild temperatures, eliminating HCl gas release and reducing equipment corrosion risks.
Q: What purity levels can be achieved with this synthetic protocol?
A: The patented method demonstrates the capability to achieve product purity levels as high as 99.88%, with a total two-step yield exceeding 91%, ensuring high quality for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving neuromuscular blocking agents like Mivacurium Chloride. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the >99% purity benchmarks required for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle the specific solvent and reagent requirements of this low-temperature synthesis, guaranteeing a supply chain that is both resilient and responsive to your evolving needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
