Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Oncology Applications
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Oncology Applications
The pharmaceutical industry is constantly seeking robust and scalable pathways to access complex chiral scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in Chinese Patent CN112209947A, which discloses a highly efficient synthesis method for chiral indoxazinone compounds. These molecules, characterized by their fused indole-oxazinone core, have garnered immense attention due to their potent biological profiles, particularly their cytotoxic activity against PC-3 human prostate cancer cells. The patent outlines a novel organocatalytic strategy that utilizes 2-indolemethanol and nitrones as primary starting materials, facilitated by a dual catalytic system involving chiral phosphoric acid and hexafluoroisopropanol. This approach represents a paradigm shift from traditional metal-catalyzed processes, offering a greener, safer, and more economically viable route for the production of high-purity pharmaceutical intermediates.
The structural versatility of the resulting indoxazinone derivatives allows for extensive medicinal chemistry optimization, making them invaluable assets in the discovery of new anticancer agents. The general structure of these privileged scaffolds is depicted below, showcasing the key substitution patterns that can be modulated to tune biological activity.
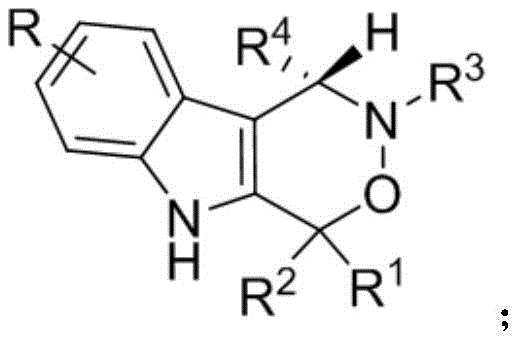
For procurement managers and supply chain directors, the implications of this technology are profound. By eliminating the need for expensive and toxic transition metal catalysts, the process not only reduces raw material costs but also simplifies the downstream purification workflow, thereby enhancing overall supply chain reliability. The ability to produce these complex heterocycles under mild conditions (20-30°C) further underscores the potential for energy-efficient large-scale manufacturing, aligning perfectly with modern sustainability goals in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral indole-fused ring systems has been fraught with significant technical and economic challenges. Traditional methodologies often rely on harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, many existing routes depend heavily on transition metal catalysis, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet stringent regulatory standards requires additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which drastically increase production costs and extend lead times. Additionally, conventional methods frequently suffer from poor enantioselectivity, necessitating difficult and yield-loss-prone resolution steps to isolate the biologically active enantiomer, ultimately compromising the atom economy and commercial viability of the process.
The Novel Approach
In stark contrast, the methodology described in patent CN112209947A offers a transformative solution by leveraging asymmetric organocatalysis. This novel approach employs a chiral phosphoric acid catalyst in conjunction with hexafluoroisopropanol (HFIP) to drive the cycloaddition reaction between 2-indolemethanol and nitrones. The reaction proceeds smoothly in toluene at ambient temperatures ranging from 20°C to 30°C, eliminating the energy demands associated with heating or cooling protocols. The use of anhydrous sodium sulfate as an additive further facilitates the reaction efficiency by managing water content without introducing corrosive reagents. This mild protocol not only ensures the integrity of the delicate indole moiety but also achieves exceptional enantioselectivity, often exceeding 90% ee across a broad range of substrates. The general reaction scheme illustrating this elegant transformation is shown below.
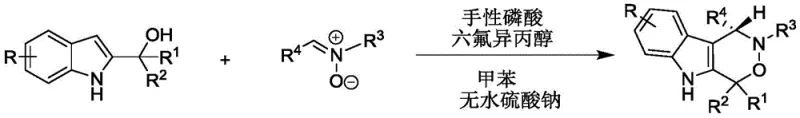
By adopting this streamlined one-pot synthesis, manufacturers can bypass the complexities of multi-step sequences and metal removal processes. The result is a high-yielding, operationally simple procedure that is inherently safer and more environmentally friendly, positioning it as an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthesis lies in the sophisticated interplay between the chiral phosphoric acid catalyst and the hydrogen-bonding donor solvent, hexafluoroisopropanol. The chiral phosphoric acid acts as a bifunctional catalyst, simultaneously activating the electrophilic nitrone and the nucleophilic 2-indolemethanol through a network of hydrogen bonds. This dual activation lowers the energy barrier for the cyclization step while imposing a rigid chiral environment that dictates the stereochemical outcome of the reaction. The specific structure of the catalyst plays a pivotal role; derivatives based on binaphthyl, octahydrobinaphthyl, or spiro skeletons, such as the one depicted below, provide the necessary steric bulk to enforce high facial selectivity during the bond-forming event.
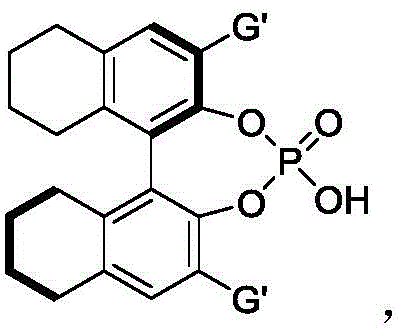
From an impurity control perspective, the mechanism ensures that side reactions are minimized. The mild acidity of the phosphoric acid prevents the polymerization of the indole methanol or the decomposition of the nitrone, which are common pitfalls in stronger acidic media. Furthermore, the presence of HFIP enhances the acidity of the catalyst via hydrogen bonding interactions, creating a synergistic effect that boosts both reaction rate and stereoselectivity. This precise control over the reaction trajectory results in a clean crude product profile, significantly reducing the burden on purification teams and ensuring that the final API intermediate meets the rigorous purity specifications required by global regulatory bodies.
How to Synthesize Chiral Indoxazinone Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the reaction mixture in toluene, where the stoichiometry of the reactants is carefully controlled to maximize conversion. The addition of the catalytic system initiates the transformation, which is monitored in real-time to ensure optimal endpoint determination. For a detailed breakdown of the standardized operating procedures, including specific molar ratios and workup protocols, please refer to the technical guide below.
- Mix 2-indolemethanol and nitrone in toluene with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol as catalysts and stir at 20-30°C.
- Monitor by TLC, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this organocatalytic route presents a compelling value proposition. The shift away from precious metal catalysts to readily available organic acids fundamentally alters the cost structure of the manufacturing process. By removing the dependency on volatile metal markets and expensive ligand systems, companies can achieve substantial cost savings in raw material procurement. Moreover, the simplified downstream processing eliminates the need for costly metal scavenging steps, further driving down the cost of goods sold (COGS) and improving overall margin potential for high-volume production campaigns.
- Cost Reduction in Manufacturing: The elimination of transition metals and the use of ambient temperature conditions significantly reduce energy consumption and waste treatment costs. The high atom economy of the reaction means that a larger proportion of the starting materials are incorporated into the final product, minimizing raw material waste. This efficiency translates directly into a more competitive pricing structure for the final pharmaceutical intermediate, allowing buyers to secure high-quality materials at a reduced total cost of ownership without compromising on purity or performance standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply chains. Since the reagents involved, such as toluene and anhydrous sodium sulfate, are commodity chemicals with stable global availability, the risk of supply disruptions due to raw material shortages is drastically minimized. This reliability allows procurement managers to forecast demand more accurately and maintain leaner inventory levels, thereby optimizing working capital and reducing the risk of obsolescence in a fast-moving market environment.
- Scalability and Environmental Compliance: The mild nature of the reaction makes it inherently safer for scale-up, reducing the risks associated with exothermic runaways or high-pressure operations. This safety profile facilitates easier regulatory approval for manufacturing sites and lowers insurance premiums. Additionally, the absence of heavy metals simplifies wastewater treatment and aligns with increasingly strict environmental regulations regarding industrial effluent. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate sustainability profile of the supply chain, a key metric for modern pharmaceutical sourcing strategies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for making informed sourcing decisions. The following questions address common inquiries regarding the scalability, biological relevance, and operational parameters of the patented method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on the feasibility of adopting this technology for commercial applications.
Q: What are the biological activities of the synthesized chiral indoxazinones?
A: The compounds exhibit cytotoxic activity against PC-3 human prostate cancer cells, with IC50 values as low as 40.08 μg/mL for specific derivatives, indicating potential for novel antitumor drug development.
Q: Why is this organocatalytic method superior to traditional synthesis?
A: Unlike conventional methods that often require violent conditions and transition metals, this process operates at mild temperatures (20-30°C) using chiral phosphoric acids, resulting in higher safety, lower costs, and excellent enantioselectivity.
Q: What is the substrate scope of this reaction?
A: The method demonstrates broad applicability, tolerating various substituents on the indole ring and nitrone component, including halogens, alkyl groups, and heteroaryl moieties, allowing for structural diversity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing advanced synthetic technologies like the one described in CN112209947A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering high-purity chiral indoxazinone intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your oncology drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing solutions can accelerate your project timelines and enhance your competitive advantage in the global marketplace.
