Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing significant bottlenecks in current manufacturing processes. This technology leverages a unique molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this chemical scaffold cannot be overstated, as evidenced by its presence in major therapeutic agents such as Sitagliptin and various anticonvulsant drugs, highlighting the urgent demand for reliable pharmaceutical intermediate suppliers capable of delivering these complex structures efficiently.
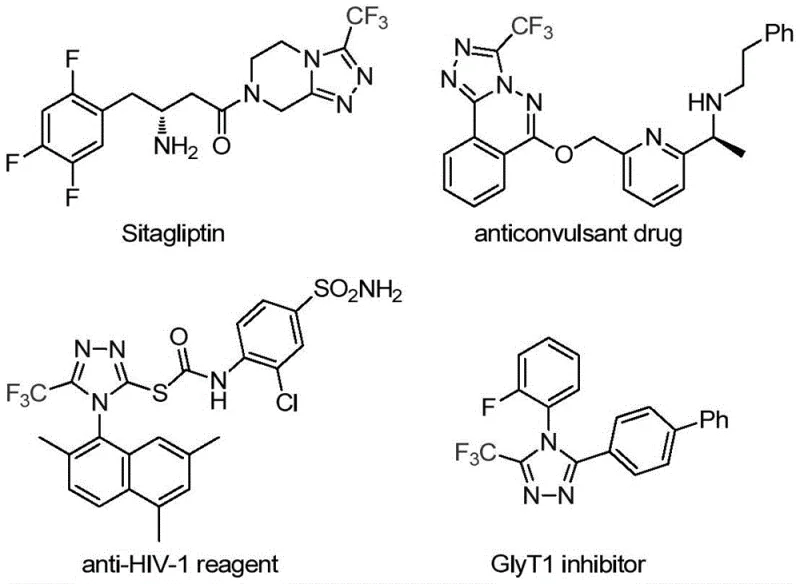
Traditionally, the construction of the 1,2,4-triazole ring has relied on methodologies that often suffer from severe limitations regarding safety, cost, and operational complexity. Conventional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl substituted 1,2,4-oxazolinones. These legacy routes frequently necessitate the handling of hazardous hydrazine derivatives, which pose significant safety risks and regulatory hurdles in large-scale manufacturing environments. Furthermore, alternative multi-component reactions involving diazonium salts and trifluorodiazoethane often require stringent temperature controls and generate substantial quantities of toxic waste, complicating the environmental compliance profile for cost reduction in API manufacturing. The reliance on unstable intermediates in these older methods frequently leads to inconsistent batch quality and lower overall yields, creating supply chain vulnerabilities for downstream drug producers.
In stark contrast, the novel approach detailed in the patent utilizes a transition metal-catalyzed strategy that fundamentally reshapes the synthetic landscape for these heterocycles. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (specifically Ph3P=N-NC) as the primary building blocks, the method bypasses the need for dangerous hydrazine reagents entirely. The reaction proceeds under remarkably mild thermal conditions, typically between 70°C and 90°C, which significantly reduces energy consumption compared to high-temperature pyrolysis methods. This shift not only enhances operator safety but also improves the impurity profile of the final product, a critical factor for R&D directors focused on purity specifications. The use of commercially available and inexpensive starting materials further underscores the economic viability of this process, making it an attractive option for the commercial scale-up of complex pharmaceutical additives.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic transformation lies in the synergistic interaction between the molybdenum and copper catalytic centers, which orchestrate the assembly of the five-membered triazole ring with high precision. The mechanism initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex that primes the carbon-nitrogen triple bond for nucleophilic attack. Simultaneously, the cuprous acetate acts as a Lewis acid promoter, facilitating the [3+2] cycloaddition between the activated isonitrile and the trifluoroethylimidoyl chloride. This dual-catalyst system ensures that the reaction barrier is sufficiently lowered to proceed at moderate temperatures while maintaining excellent regioselectivity for the 3-trifluoromethyl substitution pattern.
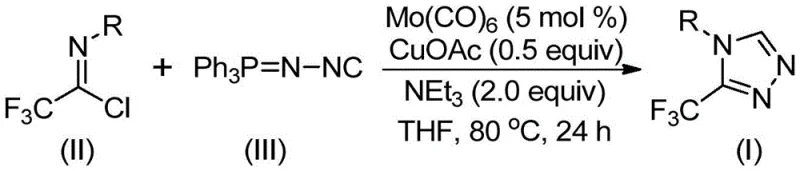
Following the initial cycloaddition, the intermediate undergoes a crucial elimination step where triphenylphosphine oxide is expelled from the molecular framework, driven by the thermodynamic stability of the resulting aromatic triazole system. This elimination is assisted by trace water present in the reaction system or added during workup, ensuring the clean formation of the final heterocyclic product. From an impurity control perspective, this mechanism is highly advantageous because the byproducts, primarily triphenylphosphine oxide and metal salts, are chemically distinct from the product and can be easily removed during standard purification protocols. The tolerance of the catalytic system towards various functional groups on the aryl ring of the imidoyl chloride—such as halogens, alkyls, and alkoxy groups—demonstrates the versatility of this method for generating diverse libraries of high-purity OLED material precursors or pharmaceutical intermediates without compromising yield.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this patented methodology offers a streamlined pathway for laboratory and pilot-scale synthesis, minimizing the need for specialized equipment while maximizing throughput. The procedure is designed to be operationally simple, requiring only standard Schlenk techniques and common organic solvents, which facilitates rapid adoption by process chemistry teams aiming for reducing lead time for high-purity pharmaceutical intermediates. The following guide outlines the standardized protocol derived from the patent examples, ensuring reproducibility and consistency across different batches.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and perform column chromatography purification to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu co-catalyzed route presents a compelling value proposition centered on cost stability and operational reliability. The primary driver for cost reduction in manufacturing stems from the elimination of expensive and hazardous reagents like hydrazines and diazonium salts, replacing them with stable, commodity-grade chemicals that are readily sourced from the global market. This shift mitigates the risk of price volatility associated with specialty reagents and reduces the overhead costs related to hazardous waste disposal and safety containment systems. Furthermore, the high atom economy of the cycloaddition reaction ensures that a greater proportion of the input mass is converted into the desired product, directly improving the material cost basis per kilogram of output.
Enhanced supply chain reliability is another critical benefit, as the robustness of the reaction conditions allows for consistent production schedules regardless of minor fluctuations in ambient conditions. The use of tetrahydrofuran (THF) as the preferred solvent, a widely available industrial chemical, prevents bottlenecks that might arise from relying on exotic or regulated solvents. Additionally, the scalability of the process, demonstrated effectively at the gram level in the patent data, suggests a smooth trajectory for scaling to multi-kilogram or tonnage production without the need for extensive process re-engineering. This scalability ensures that partners can secure long-term supply contracts with confidence, knowing that the manufacturing process is resilient and adaptable to increasing market demand.
From an environmental and regulatory standpoint, the simplified post-treatment process significantly lowers the burden of waste management. The reaction mixture can be processed through simple filtration followed by standard silica gel chromatography, avoiding complex extraction sequences that generate large volumes of aqueous waste. This aligns with modern green chemistry principles and helps manufacturing facilities maintain strict environmental compliance standards. The ability to synthesize a wide range of substituted derivatives using the same core protocol also means that production lines can be quickly switched between different product variants, offering substantial flexibility in responding to diverse client requirements for custom synthesis projects.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, providing clarity for technical teams evaluating its integration into their existing workflows. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this Mo/Cu catalytic system over traditional methods?
A: Unlike traditional methods that rely on harsh hydrazine cyclization or unstable diazonium salts, this novel approach utilizes mild conditions (70-90°C) and commercially available starting materials. The dual-metal catalysis ensures high reaction efficiency and broad substrate tolerance, significantly simplifying the synthetic route for complex heterocycles.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to gram-level reactions with high yields. The simplicity of the post-treatment process, involving basic filtration and standard column chromatography, indicates strong potential for commercial scale-up without requiring specialized high-pressure equipment.
Q: Which solvents provide the optimal conversion rates for this reaction?
A: While various aprotic solvents like acetonitrile and DMF are compatible, tetrahydrofuran (THF) is identified as the preferred solvent. THF effectively dissolves all starting materials and promotes the highest conversion rates, ensuring efficient formation of the 3-trifluoromethyl-substituted 1,2,4-triazole core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this innovative Mo/Cu catalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your development timelines while maximizing operational efficiency.
