Advanced Catalytic Synthesis of 3-Trifluoromethyl Triazoles: Enabling Commercial Scale-Up for Pharmaceutical Manufacturing
The groundbreaking patent CN113307778A introduces a novel synthetic pathway for 3-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This method addresses critical limitations in existing synthetic routes by employing a dual molybdenum-copper catalytic system that operates under remarkably mild conditions (70–90°C) while achieving high reaction efficiency. The process utilizes readily accessible starting materials including trifluoroethylimidoyl chloride and functionalized isonitriles, enabling the construction of pharmacologically relevant triazole scaffolds found in major drug classes such as antidiabetics and antivirals. Crucially, the methodology demonstrates exceptional scalability from laboratory to commercial production volumes, with documented gram-scale reactions that maintain consistent product quality. This innovation directly responds to the pharmaceutical industry's demand for robust synthetic routes that balance structural complexity with manufacturing practicality, particularly for compounds containing the valuable trifluoromethyl moiety which enhances metabolic stability and bioavailability in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethylated triazoles suffer from multiple critical drawbacks that hinder their industrial adoption. Conventional cyclization methods using trifluoroacetyl hydrazine and amidines often require harsh reaction conditions exceeding 120°C, leading to significant decomposition of sensitive intermediates and reduced yields of the desired products. Alternative routes involving hydrazinolysis of trifluoromethyl oxazolinones typically generate complex mixtures of regioisomers due to poor selectivity control, necessitating extensive purification procedures that compromise overall process efficiency. The copper-catalyzed multi-component reactions employing diazonium salts present additional challenges including hazardous reagent handling and strict anhydrous requirements that complicate large-scale implementation. Furthermore, existing methodologies demonstrate limited substrate scope with poor tolerance for functional groups commonly found in pharmaceutical intermediates, restricting their applicability in drug discovery programs. These combined limitations result in higher production costs, inconsistent quality profiles, and extended development timelines that directly impact pharmaceutical supply chain reliability.
The Novel Approach
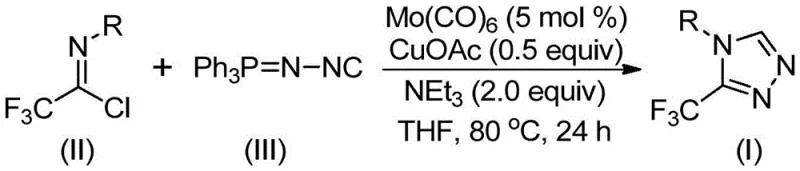
The patented methodology overcomes these historical challenges through an innovative dual catalytic system featuring molybdenum hexacarbonyl and cuprous acetate that operates under significantly milder conditions (70–90°C). This approach leverages the synergistic interaction between the two catalysts to facilitate a controlled [3+2] cycloaddition mechanism between trifluoroethylimidoyl chloride and functionalized isonitriles, ensuring high regioselectivity and minimal byproduct formation. The process utilizes commercially available starting materials with broad functional group tolerance, enabling the synthesis of diverse triazole derivatives without requiring specialized reagents or equipment. Notably, the reaction proceeds efficiently in standard organic solvents like THF at moderate temperatures, eliminating the need for extreme thermal conditions that cause decomposition in conventional methods. The simplified workup procedure involving filtration and standard column chromatography further enhances process robustness while maintaining high product purity essential for pharmaceutical applications. This combination of mild conditions, operational simplicity, and scalability represents a paradigm shift in triazole synthesis methodology.
Mechanistic Insights into Mo/Cu Dual Catalytic Triazole Formation
The reaction mechanism begins with the formation of a molybdenum-isocyanide complex that activates the functionalized isonitrile toward nucleophilic attack. This activation enables a copper-promoted [3+2] cycloaddition with trifluoroethylimidoyl chloride to form a key five-membered ring intermediate through a concerted transition state that ensures regiochemical control. The dual catalyst system operates through complementary pathways where molybdenum facilitates isonitrile coordination while copper mediates the electrophilic activation of the imidoyl chloride component. This synergistic interaction lowers the activation energy barrier for the cycloaddition step, allowing the reaction to proceed efficiently at 80°C rather than requiring elevated temperatures typical of uncatalyzed processes. The subsequent elimination of triphenylphosphine oxide occurs through hydrolysis under mild aqueous conditions, generating the final triazole product without requiring additional reagents or harsh processing steps that could compromise product integrity.
Impurity control is achieved through multiple mechanistic features inherent to this catalytic system. The regioselective nature of the [3+2] cycloaddition prevents formation of undesired regioisomers that commonly plague traditional cyclization methods. The mild reaction temperature (80°C) minimizes thermal degradation pathways that generate decomposition products in conventional high-temperature processes. The water-mediated elimination step provides a clean pathway for byproduct removal without introducing additional impurities, while the use of molecular sieves maintains optimal reaction conditions by controlling moisture levels throughout the process. This multi-faceted approach to impurity management results in consistently high-purity products suitable for pharmaceutical applications without requiring specialized purification techniques beyond standard column chromatography.
How to Synthesize 3-Trifluoromethyl Triazoles Efficiently
This patented synthetic route represents a significant advancement in the manufacturing of trifluoromethylated triazole compounds, offering pharmaceutical developers a robust and scalable alternative to conventional methodologies. The process demonstrates exceptional operational simplicity while maintaining high levels of chemical precision required for active pharmaceutical ingredient intermediates. By leveraging commercially available catalysts and starting materials under mild reaction conditions, this method provides an ideal platform for both research-scale synthesis and commercial production. The following standardized procedure details the critical parameters necessary for successful implementation across various production scales, ensuring consistent product quality and process reliability essential for pharmaceutical manufacturing environments.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieve in THF under inert atmosphere
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) at room temperature with continuous stirring
- Heat reaction mixture to 80°C for 24 hours, then perform standard workup including filtration and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach delivers substantial value across procurement and supply chain operations by addressing fundamental challenges in pharmaceutical intermediate manufacturing. The methodology eliminates dependency on specialized equipment or hazardous reagents while maintaining compatibility with standard manufacturing infrastructure found in most chemical production facilities. By utilizing readily available catalysts and starting materials with broad commercial supply channels, the process significantly reduces sourcing risks associated with rare or controlled substances commonly required in alternative synthetic routes. The operational simplicity translates directly into reduced training requirements and faster technology transfer between development and production sites, enhancing overall supply chain agility while maintaining stringent quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and high-temperature processing requirements substantially reduces raw material costs while minimizing energy consumption during production. The simplified workup procedure avoids costly purification steps needed in conventional methods to remove regioisomeric impurities, resulting in higher overall process efficiency without requiring additional capital investment in specialized equipment.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials including molybdenum hexacarbonyl and cuprous acetate ensures consistent supply regardless of regional sourcing constraints. The robust nature of the reaction tolerates minor variations in raw material quality while maintaining product specifications, reducing batch failures and associated supply disruptions common in more sensitive synthetic processes.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory to commercial production volumes without requiring significant parameter adjustments or specialized engineering controls. The absence of hazardous reagents and byproducts simplifies waste stream management while meeting increasingly stringent environmental regulations governing pharmaceutical manufacturing operations worldwide.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement and R&D teams regarding implementation of this patented synthetic methodology. These responses are derived directly from experimental data and process observations documented in patent CN113307778A, providing evidence-based insights into the practical application of this innovative manufacturing approach for pharmaceutical intermediates.
Q: How does this method improve impurity control compared to conventional cyclization approaches?
A: The dual Mo/Cu catalytic system enables precise regioselectivity through controlled [3+2] cycloaddition, minimizing side products from competing reaction pathways. The mild conditions (80°C) prevent thermal degradation observed in traditional high-temperature cyclizations, while the water-mediated elimination step ensures clean removal of triphenylphosphine oxide byproducts.
Q: What substrate flexibility does this process offer for pharmaceutical intermediate development?
A: The method accommodates diverse aryl substituents (methyl, methoxy, fluoro, chloro) and phenethyl groups on the isonitrile component, with demonstrated tolerance for electron-donating and electron-withdrawing functional groups. This design flexibility allows rapid generation of compound libraries for structure-activity relationship studies without modifying core reaction parameters.
Q: How does the catalyst system contribute to supply chain reliability?
A: The use of commercially available molybdenum hexacarbonyl and cuprous acetate eliminates dependency on rare transition metals. The catalyst loading (5 mol% Mo, 0.5 equiv Cu) ensures consistent performance across batches while maintaining cost efficiency through simplified catalyst recovery protocols during standard workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Triazole Supplier
Our patented methodology represents a significant advancement in the synthesis of critical pharmaceutical intermediates containing the valuable trifluoromethyl triazole motif. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our specialized expertise in heterocyclic chemistry enables seamless technology transfer from laboratory development to full-scale manufacturing, ensuring consistent product quality that meets global regulatory requirements for active pharmaceutical ingredient intermediates.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs by contacting our technical procurement team. They will provide detailed COA data and route feasibility assessments demonstrating how this innovative process can enhance your supply chain reliability while delivering substantial operational efficiencies for your pharmaceutical manufacturing operations.
