Advanced Metal-Free Synthesis of Isatin Derivatives for Commercial Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of Isatin Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and environmentally benign synthetic routes for critical heterocyclic scaffolds. Patent CN109456249B introduces a transformative methodology for the synthesis of isatin derivatives, a privileged structure found in numerous bioactive molecules exhibiting anti-HIV, anti-inflammatory, and antifungal properties. This innovation addresses the longstanding challenges associated with traditional oxidative cyclization methods by replacing expensive transition metal catalysts with a simple, cost-effective Brønsted acid system. By utilizing molecular oxygen as the terminal oxidant and trifluoroacetic acid as the catalyst, this process offers a streamlined pathway that aligns perfectly with modern green chemistry principles and stringent regulatory requirements for impurity control in active pharmaceutical ingredients.
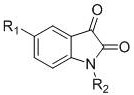
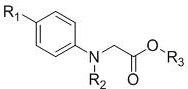
The core innovation lies in the direct oxidative cyclization of N-alkyl-N-aryl glycine ester derivatives. Unlike conventional approaches that rely on harsh conditions or precious metals, this method operates under mild thermal conditions, typically around 60°C, using 1,2-dichloroethane as the solvent. The structural versatility of this approach allows for the introduction of various alkyl, alkoxy, and aryl groups at the R1 and R2 positions, enabling the rapid generation of diverse libraries of isatin analogs. For R&D directors focused on lead optimization, this flexibility is invaluable, as it permits the exploration of structure-activity relationships without being constrained by incompatible functional group tolerances often seen in metal-catalyzed C-H activation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isatin core has relied heavily on transition metal catalysis, particularly using copper or palladium complexes to facilitate intramolecular C-H oxidative cyclization. While effective in academic settings, these methods present significant hurdles for industrial scale-up. The use of stoichiometric or even catalytic amounts of heavy metals introduces severe contamination risks, necessitating elaborate and expensive purification steps to meet the strict ppm-level limits imposed by global health authorities. Furthermore, many traditional protocols require strong, hazardous oxidants such as pyridinium chlorochromate (PCC) or excess hydrogen peroxide, which pose safety risks regarding thermal runaway and generate substantial toxic waste streams. These factors collectively inflate the cost of goods sold (COGS) and complicate the supply chain logistics for high-purity intermediates.
The Novel Approach
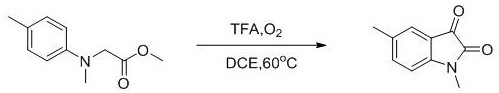
The methodology disclosed in CN109456249B represents a paradigm shift by employing a metal-free, acid-catalyzed oxidative cyclization strategy. By leveraging the intrinsic reactivity of N-alkyl-N-aryl glycine esters under acidic conditions in the presence of oxygen, the process achieves ring closure without the need for external metal promoters. This approach drastically simplifies the reaction setup and workup procedures. The reliance on molecular oxygen, an abundant and atom-economical oxidant, eliminates the need for purchasing and handling dangerous chemical oxidants. As demonstrated in the specific reaction schemes, substrates such as N-methyl-N-p-tolylglycine methyl ester can be efficiently converted to 1,5-dimethyl-2,3-indolinedione with respectable yields, showcasing the practical viability of this route for commercial production.
Mechanistic Insights into Acid-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between acid catalysis and aerobic oxidation. The process initiates with the protonation of the carbonyl oxygen of the glycine ester moiety by trifluoroacetic acid, which enhances the electrophilicity of the carbonyl carbon. This activation facilitates an intramolecular electrophilic aromatic substitution or a related cyclization event, depending on the specific electronic environment of the aromatic ring. The presence of oxygen is critical, serving as the hydrogen acceptor to restore aromaticity and form the dicarbonyl system characteristic of the isatin scaffold. This mechanism avoids the formation of radical intermediates often associated with metal-catalyzed pathways, thereby reducing the generation of complex side products and oligomeric impurities that are difficult to separate.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of metal species means there is no risk of metal-ligand complexation leading to persistent colored impurities or catalytic decomposition of the product during storage. Furthermore, the mild acidic conditions ensure that acid-sensitive protecting groups elsewhere in a complex molecule might remain intact, although the primary substrates here are relatively simple esters. The reaction proceeds through a well-defined ionic pathway rather than a chaotic radical chain reaction, resulting in a cleaner crude reaction profile. This cleanliness translates directly to higher recovery rates during the final column chromatography or crystallization steps, maximizing the overall mass balance and reducing solvent consumption per kilogram of product produced.
How to Synthesize 1,5-Dimethyl-2,3-indolinedione Efficiently
The synthesis of specific isatin derivatives, such as 1,5-dimethyl-2,3-indolinedione, follows a standardized protocol that balances reaction time and yield optimization. The procedure involves charging a reaction vessel with the appropriate N-alkyl-N-aryl glycine ester substrate, dissolving it in 1,2-dichloroethane, and adding a catalytic amount of trifluoroacetic acid. The mixture is then stirred under an oxygen atmosphere at 60°C. While the patent indicates reaction times can vary significantly from 30 to 150 hours depending on the steric and electronic nature of the substituents, monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical documentation below.
- Charge a reaction vessel with N-alkyl-N-aryl glycine ester derivative, 1,2-dichloroethane solvent, and 20 mol% trifluoroacetic acid under an oxygen atmosphere.
- Stir the reaction mixture at a controlled temperature of 60°C for a duration ranging from 30 to 150 hours depending on the substrate substituents.
- Upon completion, remove the solvent via reduced pressure distillation and purify the crude residue using silica gel column chromatography to isolate the pure isatin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthetic route offers compelling economic and logistical benefits. The most immediate impact is the drastic reduction in raw material costs associated with catalysts. By eliminating the need for palladium or copper salts, which are subject to volatile market pricing and geopolitical supply constraints, manufacturers can stabilize their input costs. Additionally, the simplified downstream processing—specifically the removal of heavy metal scavenging resins and filtration steps—reduces both the time and consumables required for purification. This streamlining of the manufacturing process enhances the overall throughput of the production facility, allowing for faster turnaround times on custom synthesis orders.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous stoichiometric oxidants fundamentally alters the cost structure of isatin derivative production. Without the need for specialized metal removal technologies or the disposal of toxic heavy metal waste, the operational expenditure (OPEX) is significantly lowered. Furthermore, the use of molecular oxygen as the oxidant is virtually cost-free compared to purchasing proprietary oxidizing reagents, contributing to substantial long-term savings in chemical procurement budgets.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as trifluoroacetic acid, 1,2-dichloroethane, and oxygen gas ensures a robust and resilient supply chain. Unlike specialized ligands or organometallic complexes that may have single-source suppliers and long lead times, the reagents for this process are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions (60°C) and the absence of explosive or highly toxic reagents make this process inherently safer for scale-up to multi-ton quantities. From an environmental standpoint, the reduction in heavy metal waste and the use of oxygen align with increasingly stringent global environmental regulations. This compliance reduces the burden on waste treatment facilities and minimizes the environmental footprint of the manufacturing site, supporting corporate sustainability goals and reducing regulatory overhead.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is critical for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this acid-catalyzed method over traditional metal-catalyzed routes?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts such as copper or palladium. This removes the need for complex and costly heavy metal removal steps during downstream processing, significantly simplifying purification and ensuring higher purity profiles suitable for pharmaceutical applications.
Q: What oxidant is utilized in this synthetic pathway?
A: This process utilizes molecular oxygen (O2) as the terminal oxidant, typically supplied via an oxygen balloon. This is a stark contrast to traditional methods that often require hazardous stoichiometric oxidants like PCC or hydrogen peroxide, making the process greener and safer for large-scale operations.
Q: What yields can be expected for substituted isatin derivatives?
A: According to the patent data, yields vary based on the electronic nature of the substituents. For instance, electron-donating groups like methyl or methoxy generally provide moderate to good yields ranging from 56% to 63%, while bulkier or electron-withdrawing groups may result in slightly lower yields around 35% to 45%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isatin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient, high-quality intermediate synthesis in the development of next-generation therapeutics. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of isatin derivatives we produce, adhering to the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain for isatin-based projects. Whether you require custom synthesis of novel analogs or reliable tonnage supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
