Advanced Ezetimibe Intermediate Manufacturing: Leveraging Novel Chiral Auxiliaries for Industrial Scale-Up
Advanced Ezetimibe Intermediate Manufacturing: Leveraging Novel Chiral Auxiliaries for Industrial Scale-Up
The pharmaceutical landscape for lipid-lowering agents continues to evolve, driven by the relentless demand for higher purity and more cost-effective manufacturing processes for blockbuster drugs like Ezetimibe. A pivotal advancement in this domain is detailed in patent CN101935309A, which discloses a robust methodology for preparing Ezetimibe and its critical intermediates. This technology represents a significant departure from conventional synthetic routes by introducing a novel chiral auxiliary system based on a tricyclic indeno-oxazole scaffold. For R&D directors and procurement strategists, this patent offers a compelling value proposition: a pathway that not only achieves exceptional stereochemical control with ee values reaching 99.97% but also simplifies the downstream processing required to isolate the final Active Pharmaceutical Ingredient (API). The core innovation lies in replacing traditional, less rigid chiral inducers with a sterically demanding auxiliary that enforces high fidelity during the formation of the azetidinone ring, thereby addressing long-standing challenges in impurity profiling and yield optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ezetimibe has relied heavily on the use of (S)-4-benzyl-2-oxazolone as a chiral auxiliary to establish the critical stereocenters at the 3 and 4 positions of the azetidinone ring. While effective to a degree, this conventional approach suffers from inherent limitations regarding steric bulk and conformational flexibility. The benzyl group, being relatively flexible and less sterically encumbered compared to fused ring systems, can sometimes lead to suboptimal diastereoselectivity during the cyclization steps. This often necessitates rigorous and costly purification protocols, such as repeated recrystallizations or preparative chromatography, to remove unwanted diastereomers that compromise the optical purity of the final drug substance. Furthermore, the recovery and recycling of the benzyl-oxazolone auxiliary can be inefficient, leading to increased raw material costs and a larger environmental footprint due to waste generation. These factors collectively inflate the cost of goods sold (COGS) and introduce supply chain vulnerabilities associated with the consistent availability of high-purity starting materials.
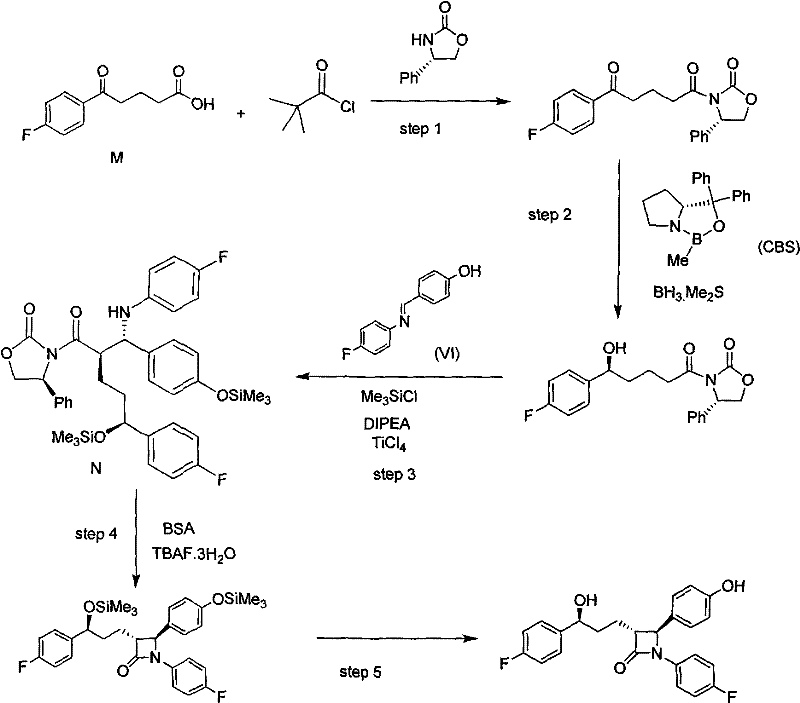
The Novel Approach
In stark contrast, the methodology outlined in CN101935309A introduces a paradigm shift by utilizing (3aS, 8aR)-3,3a,8,8a-tetrahydro-2H-indeno[1,2-d]oxazole-2-one as the chiral auxiliary. This tricyclic structure imposes a much more rigid and sterically demanding environment around the reaction center. The increased steric hindrance effectively shields one face of the molecule during nucleophilic attacks, driving the formation of the desired stereoisomer with remarkable precision. This structural rigidity translates directly into process efficiency; the patent reports yields reaching 90% in key steps with optical purity exceeding 99.97%, effectively eliminating the need for extensive chiral separations later in the process. Moreover, the indeno-oxazole auxiliary is designed for recoverability, with the patent noting a recycling yield of approximately 95%. This closed-loop capability significantly mitigates raw material costs and aligns with modern green chemistry principles, making the process highly attractive for sustainable commercial manufacturing.
Mechanistic Insights into Indeno-Oxazole Mediated Asymmetric Synthesis
The success of this synthetic route hinges on the precise orchestration of steric and electronic effects facilitated by the indeno-oxazole auxiliary. The mechanism begins with the acylation of the auxiliary with 5-(4-fluorophenyl)-5-carbonyl valeric acid to form the keto-amide intermediate (Compound IV). The rigidity of the indeno-fused ring system locks the amide bond into a specific conformation that directs subsequent reactions. When this intermediate undergoes asymmetric reduction to install the 3'-hydroxyl chiral center, the bulky auxiliary framework works in concert with the reducing agent (such as CBS catalyst or chloroborane species) to ensure hydride delivery occurs exclusively from the less hindered face. This synergistic effect is crucial for establishing the (S)-configuration at the hydroxyl bearing carbon with the reported high enantiomeric excess. The subsequent coupling reaction with the substituted imine (Compound III) further benefits from this pre-organized geometry, where the auxiliary directs the incoming nucleophile to form the correct relative stereochemistry at the azetidinone ring junction.

Impurity control in this process is inherently built into the molecular design. By maximizing the diastereomeric ratio at the earliest stages of the synthesis, the formation of difficult-to-separate isomers is suppressed at the source. Traditional routes often struggle with 'epimerization' risks during the cyclization or deprotection phases; however, the stability of the indeno-oxazole adduct minimizes these risks. The robust nature of the intermediate compounds allows for standard aqueous workups and crystallizations to effectively purge minor impurities without sacrificing yield. For quality control teams, this means a cleaner impurity profile in the final API, reducing the burden on analytical laboratories and accelerating batch release times. The mechanistic clarity provided by this route ensures that scale-up from kilogram to tonnage quantities maintains the same high fidelity observed in laboratory settings.
How to Synthesize Ezetimibe Intermediate Efficiently
The synthesis of the key intermediate Compound I involves a sequence of highly controlled reactions that leverage the unique properties of the indeno-oxazole auxiliary. The process initiates with the activation of the valeric acid derivative followed by coupling with the chiral auxiliary under mild basic conditions. Subsequent asymmetric reduction establishes the side-chain chirality, followed by a Lewis acid-mediated coupling with the fluorophenyl-imine species to construct the core azetidinone scaffold. The final steps involve a carefully managed cyclization and deprotection sequence to reveal the active pharmacophore. Detailed standard operating procedures for each unit operation, including specific temperature ramps and quenching protocols, are essential for maintaining the high stereochemical integrity described in the patent literature.
- Preparation of Compound IV via condensation of 5-(4-fluorophenyl)-5-carbonyl valeric acid with the indeno-oxazole auxiliary.
- Asymmetric reduction of Compound IV to Compound II using chloroborane reagents to establish the 3'-hydroxyl chiral center.
- Coupling of Compound II with substituted imine (Compound III) under Lewis acid catalysis to form the protected intermediate Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic advantages that extend beyond mere technical elegance. The primary driver for value creation is the drastic simplification of the purification train. In traditional manufacturing, a significant portion of operational expenditure is dedicated to separating closely related stereoisomers and removing residual chiral auxiliaries. By achieving near-perfect stereoselectivity upfront, this process eliminates several purification units, thereby reducing solvent consumption, energy usage, and labor hours. This streamlining directly correlates to a lower cost base, allowing for more competitive pricing in the global marketplace. Additionally, the ability to recover and reuse the expensive chiral auxiliary at high efficiency creates a buffer against raw material price volatility, ensuring more stable long-term costing models for the final API.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the high recovery rate of the chiral auxiliary and the reduction in waste disposal costs. By recycling the indeno-oxazole scaffold with approximately 95% efficiency, the effective cost per kilogram of the chiral inducer is significantly amortized over multiple batches. Furthermore, the high yields reported (up to 90% in final conversion steps) mean that less starting material is required to produce the same amount of product, directly lowering the variable cost of production. The elimination of complex chromatographic separations also reduces the capital expenditure required for specialized equipment, making the process viable for a wider range of manufacturing facilities.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on scarce or difficult-to-synthesize reagents. This route utilizes readily available starting materials and robust reaction conditions that are less sensitive to minor fluctuations in temperature or moisture compared to highly sensitive organometallic alternatives. The mild conditions described, such as reactions proceeding at temperatures ranging from -30°C to 30°C, are easily achievable in standard glass-lined or stainless steel reactors without requiring cryogenic infrastructure. This operational flexibility ensures that production schedules can be maintained consistently, reducing the risk of batch failures or delays that could disrupt the supply of this critical cardiovascular medication to patients worldwide.
- Scalability and Environmental Compliance: As regulatory bodies impose stricter limits on solvent residues and heavy metal contaminants, this metal-free or low-metal approach offers a distinct compliance advantage. The process avoids the use of toxic transition metals often found in alternative hydrogenation catalysts, simplifying the validation process for regulatory filings. From an environmental standpoint, the reduced solvent load and the ability to recycle key reagents align with sustainability goals, minimizing the ecological footprint of the manufacturing site. This 'green' profile not only satisfies regulatory requirements but also enhances the brand reputation of the manufacturer as a responsible supplier in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's potential within your existing manufacturing portfolio. Understanding these nuances is critical for making informed decisions about technology transfer and process validation.
Q: What is the primary advantage of the indeno-oxazole auxiliary over traditional benzyl-oxazolone?
A: The indeno-oxazole auxiliary possesses a rigid tricyclic structure that provides significantly greater steric hindrance compared to the flexible benzyl group. This enhanced steric environment drives stereoselectivity to exceptional levels (ee > 99.97%) and allows for efficient recovery and recycling of the auxiliary.
Q: How does this process impact the cost of goods for Ezetimibe manufacturing?
A: By achieving high yields (up to 90% in final steps) and enabling the recovery of the expensive chiral auxiliary at approximately 95% efficiency, the process drastically reduces raw material consumption. Furthermore, mild reaction conditions minimize energy costs and equipment wear.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to mild operating temperatures, simple work-up procedures involving standard filtration and crystallization, and the avoidance of hazardous or difficult-to-remove transition metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven scale-up capabilities. Our team specializes in the development and manufacturing of complex pharmaceutical intermediates, including those required for the synthesis of Ezetimibe. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield metrics demonstrated in the lab are faithfully reproduced at the plant level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm the >99.9% ee targets mandated by this advanced synthetic route.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing process can be integrated into your supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to request specific COA data and route feasibility assessments to validate the commercial viability of this technology for your operations. Let us collaborate to deliver high-quality, cost-effective Ezetimibe intermediates that meet the highest standards of the global pharmaceutical industry.
