Advanced Organocatalytic Synthesis of Bioactive Chiral Chroman Compounds for Commercial API Production
Advanced Organocatalytic Synthesis of Bioactive Chiral Chroman Compounds for Commercial API Production
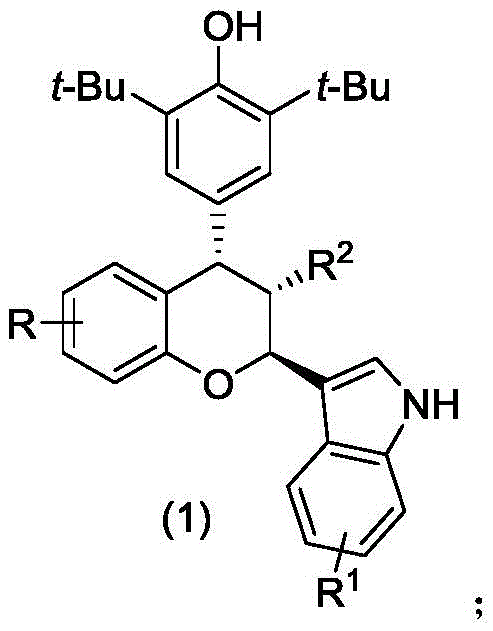
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN112920173A, which discloses a novel class of chiral chroman compounds exhibiting potent cytotoxic activity against aggressive adenocarcinoma cell lines. This patent introduces a highly efficient asymmetric organocatalytic strategy that overcomes the historical limitations of traditional synthesis methods, such as harsh reaction conditions and poor stereocontrol. By leveraging a specific chiral phosphoric acid catalyst system, the disclosed methodology achieves exceptional enantioselectivity and diastereoselectivity under remarkably mild conditions. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, more sustainable, and commercially viable manufacturing processes for high-value oncology intermediates. The ability to access these structurally diverse chroman derivatives with high purity opens new avenues for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral chroman derivatives has been plagued by significant technical hurdles that impede their widespread adoption in commercial API manufacturing. Traditional methodologies often rely on transition metal catalysts or stoichiometric chiral auxiliaries, which introduce severe complications regarding product purity and regulatory compliance. The presence of residual heavy metals necessitates expensive and time-consuming purification steps to meet stringent pharmacopeial standards, thereby inflating the overall cost of goods. Furthermore, many conventional routes require extreme reaction conditions, such as high temperatures or strongly acidic/basic environments, which can lead to decomposition of sensitive functional groups and safety hazards in a plant setting. These violent reaction processes not only increase the risk of operational accidents but also result in inconsistent yields and reduced enantiomeric excess, ultimately compromising the quality of the final pharmaceutical intermediate.
The Novel Approach
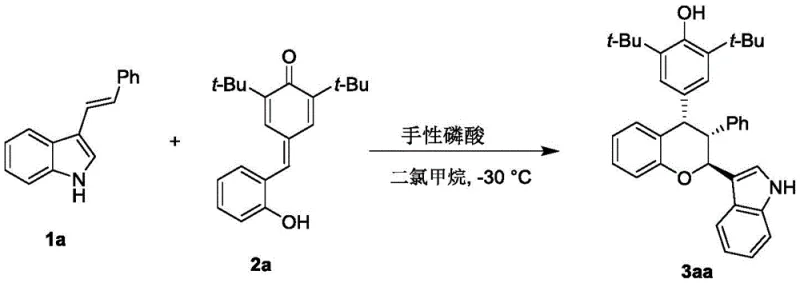
In stark contrast, the innovative approach detailed in the patent utilizes a chiral phosphoric acid catalyzed cascade reaction between p-methylene benzoquinones and 3-indolenes. This organocatalytic strategy operates under exceptionally mild conditions, specifically at a low temperature of -30°C in dichloromethane, ensuring the integrity of sensitive substrates and minimizing side reactions. The use of molecular sieves as additives further enhances the reaction efficiency by sequestering water, driving the equilibrium towards the desired product. This method eliminates the need for toxic transition metals, inherently simplifying the downstream processing and waste treatment protocols. The result is a streamlined, one-pot synthesis that delivers chiral chroman compounds with excellent yields and superior stereochemical control, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for green chemistry compliance.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The core of this technological advancement lies in the precise activation mode of the chiral phosphoric acid catalyst, which functions through a dual hydrogen-bonding mechanism. The catalyst, typically a binaphthyl skeleton derivative substituted with bulky groups like 9-anthracenyl, creates a well-defined chiral pocket around the reacting species. It simultaneously activates the electrophilic p-methylene benzoquinone and the nucleophilic 3-indolene through hydrogen bonding interactions. This bifunctional activation lowers the energy barrier for the carbon-carbon bond formation while rigidly controlling the spatial orientation of the approaching molecules. Such precise stereocontrol is crucial for establishing the multiple chiral centers found in the chroman scaffold with high fidelity. The specific geometry of the catalyst ensures that the reaction proceeds through a highly organized transition state, leading to the observed high diastereomeric and enantiomeric ratios.
Furthermore, the mechanistic pathway inherently suppresses the formation of unwanted byproducts and impurities, which is a critical consideration for process chemists. By avoiding radical pathways or high-energy intermediates common in thermal cyclizations, the reaction maintains a clean profile throughout the conversion. The mild acidic nature of the phosphoric acid prevents the polymerization of the reactive quinone methide intermediates, a common side reaction that plagues other methods. This impurity control mechanism translates directly to simplified purification workflows, often requiring only standard silica gel column chromatography to achieve analytical purity. For manufacturing teams, this means reduced solvent consumption and shorter cycle times, directly impacting the bottom line through improved operational efficiency and resource utilization.
How to Synthesize Chiral Chroman Efficiently
The practical implementation of this synthesis route is designed for ease of operation, utilizing standard laboratory equipment and readily available reagents. The process begins with the preparation of the reaction mixture in anhydrous dichloromethane, where the substrates are combined with activated molecular sieves to ensure a dry environment. The addition of the chiral catalyst initiates the transformation at a controlled low temperature, allowing for precise monitoring of the reaction progress via thin-layer chromatography. Upon completion, the workup involves simple filtration to remove the solid additives followed by concentration and purification. This straightforward protocol minimizes the need for specialized handling or exotic reagents, facilitating rapid technology transfer from the lab to the pilot plant.
- Prepare the reaction mixture by combining p-methylene benzoquinone and 3-indolene substrates in dichloromethane solvent with molecular sieves as an additive.
- Add the chiral phosphoric acid catalyst (preferably a binaphthyl skeleton derivative) to the mixture and maintain the reaction temperature at -30°C.
- Stir the reaction until TLC indicates completion, then filter to remove additives, concentrate the filtrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers profound advantages that address key pain points in the global supply chain for fine chemicals and API intermediates. The shift from metal-based catalysis to organocatalysis fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating expensive and potentially toxic metal catalysts, manufacturers can significantly reduce raw material costs and avoid the regulatory burdens associated with heavy metal clearance testing. This simplification of the supply chain enhances reliability, as the sourcing of organocatalysts is generally more stable and less subject to geopolitical fluctuations compared to precious metals. Additionally, the mild reaction conditions contribute to substantial cost savings in terms of energy consumption and safety infrastructure requirements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for costly scavenging resins and extensive purification steps required to meet residual metal limits. This streamlining of the downstream process leads to significant reductions in solvent usage and waste disposal costs. Furthermore, the high atom economy of the reaction ensures that a greater proportion of the starting materials are converted into the final product, maximizing material efficiency. The use of common solvents like dichloromethane and standard purification techniques like silica gel chromatography further ensures that the process remains economically competitive on a large scale.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable organocatalysts and commodity chemicals like p-methylene benzoquinones ensures a resilient supply chain. Unlike complex ligand-metal systems that may have long lead times or single-source dependencies, the reagents for this process are widely available from multiple vendors. This diversification of supply sources mitigates the risk of production delays caused by raw material shortages. The simplicity of the reaction setup also allows for flexible manufacturing scheduling, enabling producers to respond quickly to fluctuating market demands without extensive retooling or specialized training for operators.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions, specifically the low temperature of -30°C, presents a manageable engineering challenge that is easily addressed with standard industrial cooling systems. The absence of hazardous reagents and the generation of minimal waste align perfectly with modern environmental, health, and safety (EHS) standards. This green chemistry profile facilitates easier permitting and regulatory approval for new manufacturing facilities. The process is inherently scalable, allowing for seamless transition from gram-scale laboratory synthesis to multi-ton commercial production without the loss of selectivity or yield that often accompanies scale-up efforts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral chroman synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using chiral phosphoric acid over transition metal catalysts for chroman synthesis?
A: Chiral phosphoric acid organocatalysis eliminates the risk of heavy metal contamination, which is critical for pharmaceutical intermediates. It operates under milder conditions (-30°C) compared to many metal-catalyzed processes, reducing safety risks and energy consumption while maintaining high enantioselectivity.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is suitable for industrial large-scale production. The reaction conditions are conventional, the process is mild and safe, and it utilizes easily available raw materials, making it highly viable for commercial scale-up.
Q: What is the biological activity profile of the synthesized chiral chroman compounds?
A: The synthesized compounds exhibit strong cytotoxic activity against various adenocarcinoma cell lines, including MDA-MB-231 (breast cancer), HepG2 (liver cancer), and SGC-7901 (gastric cancer), addressing the limitation of prior art compounds which showed weak activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in advancing oncology drug development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from concept to market. Our state-of-the-art facilities are equipped to handle the specific requirements of low-temperature organocatalysis, and our rigorous QC labs enforce stringent purity specifications to guarantee the highest quality intermediates. We are committed to delivering consistent supply and technical excellence, supporting your R&D goals with reliable manufacturing capabilities.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific therapeutic programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your path to clinical success with our expert manufacturing solutions.
