Scalable Synthesis of Phenylpropionic Acids Using Recyclable Heterogeneous Palladium Catalysts
Scalable Synthesis of Phenylpropionic Acids Using Recyclable Heterogeneous Palladium Catalysts
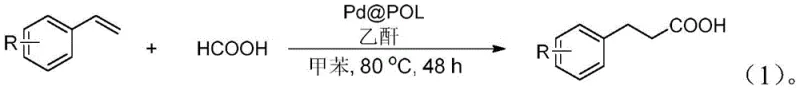
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign synthetic routes for high-value intermediates like phenylpropionic acid derivatives, which serve as crucial scaffolds in drug discovery and agrochemical development. A significant breakthrough in this domain is detailed in Chinese Patent CN112679336A, which discloses a novel method for synthesizing phenylpropionic acid compounds utilizing a heterogeneous palladium metal catalyst known as Pd@POL. This technology represents a paradigm shift from traditional homogeneous catalysis, addressing critical pain points regarding heavy metal contamination and catalyst recovery. By employing a polymer-supported palladium system, the process achieves efficient hydroformylation of styrenes using formic acid as a carbon source under relatively mild conditions. The strategic implementation of this heterogeneous system not only streamlines the downstream purification process but also aligns with the increasingly stringent regulatory requirements for residual metals in active pharmaceutical ingredients (APIs). For procurement and technical teams, understanding the mechanistic advantages and operational simplicity of this patented route is essential for evaluating its potential integration into existing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylpropionic acid derivatives has relied on several conventional methodologies, each fraught with significant operational and environmental drawbacks that hinder large-scale commercial viability. Traditional approaches often involve the reduction of cinnamic acid derivatives using reagents such as sodium amalgam or Raney nickel combined with hydrazine hydrate, processes that generate substantial hazardous waste and pose severe safety risks due to the handling of pyrophoric materials and toxic mercury byproducts. Furthermore, earlier palladium-catalyzed carbonylation methods typically utilized homogeneous catalysts like palladium acetate, which, while effective, necessitate complex and costly post-reaction treatments to remove trace palladium residues to meet pharmacopeial standards. These purification steps, often involving specialized scavengers or extensive chromatography, drastically increase the cost of goods sold (COGS) and extend the overall production lead time. Additionally, the use of high-pressure carbon monoxide in some legacy protocols introduces significant safety hazards and requires specialized high-pressure reactor infrastructure, limiting the accessibility of these methods for many contract manufacturing organizations.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN112679336A introduces a streamlined, heterogeneous catalytic system that fundamentally alters the economic and environmental landscape of phenylpropionic acid manufacturing. By utilizing the Pd@POL catalyst, the reaction proceeds efficiently at atmospheric pressure using formic acid and acetic anhydride as reagents in a toluene solvent system at a moderate temperature of 80°C. The heterogeneous nature of the catalyst allows for its physical separation from the reaction mixture via simple filtration, effectively eliminating the need for expensive and time-consuming metal scavenging processes. This innovation not only simplifies the workup procedure—requiring only standard aqueous washing and extraction—but also enables the direct reuse of the catalyst, as evidenced by recycling studies showing sustained activity over multiple cycles. The transition to this greener protocol significantly reduces the generation of heavy metal-contaminated waste streams, thereby lowering disposal costs and enhancing the overall sustainability profile of the manufacturing process, a key consideration for modern supply chain managers focused on ESG goals.
Mechanistic Insights into Pd@POL Catalyzed Hydroformylation
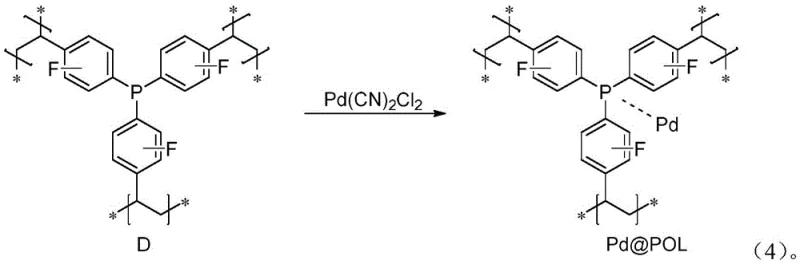
The core of this technological advancement lies in the unique architecture of the Pd@POL catalyst, which consists of palladium species coordinated within a cross-linked polymer matrix containing phosphine ligands. This structural design ensures that the active palladium centers are sterically protected and electronically tuned to facilitate the insertion of carbon monoxide (generated in situ from formic acid and acetic anhydride) into the styrene double bond. The polymer support acts as a nanoreactor, preventing the aggregation of palladium nanoparticles into inactive bulk metal, a common deactivation pathway in heterogeneous catalysis. Mechanistically, the reaction likely proceeds through a palladium-hydride species formed from the decomposition of formic acid, which inserts into the vinyl group of the styrene substrate. Subsequent migratory insertion of the in situ generated carbon monoxide and hydrolysis yields the desired phenylpropionic acid product while regenerating the active catalytic species. The robustness of the polymer backbone ensures that the ligand environment remains stable under the reaction conditions of 80°C for 48 hours, preventing leaching of palladium into the solution and ensuring the final product meets stringent purity specifications without additional purification.
Furthermore, the impurity profile of the resulting phenylpropionic acids is significantly improved compared to homogeneous methods, primarily due to the absence of soluble palladium complexes that can catalyze side reactions or remain trapped in the crystal lattice of the product. The controlled environment within the polymer matrix minimizes oligomerization of the styrene substrate, a common side reaction in acid-catalyzed processes, thereby enhancing the selectivity towards the linear phenylpropionic acid isomer. This high selectivity is crucial for pharmaceutical applications where isomeric purity can impact biological activity and regulatory approval. The ability to tune the electronic properties of the polymer support by varying the substituents on the aromatic rings of the ligand precursor offers a pathway for further optimization of the catalyst for specific substrates, providing R&D teams with a versatile platform for developing diverse libraries of phenylpropanoid derivatives. The stability of the catalyst under the reaction conditions also implies a longer operational lifetime, reducing the frequency of catalyst replacement and contributing to a more consistent and reliable manufacturing process.
How to Synthesize Phenylpropionic Acid Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory glassware and readily available reagents, making it highly accessible for both pilot-scale development and full commercial production. The process begins with the preparation of the reaction mixture by sequentially adding the Pd@POL catalyst, toluene, the specific styrene derivative, formic acid, and acetic anhydride into a reaction vessel equipped with a stirrer and condenser. The detailed standardized synthesis steps, including precise molar ratios, addition rates, and specific workup parameters for various substrates, are outlined in the comprehensive guide below to ensure reproducibility and optimal yield.
- Prepare the reaction mixture by adding Pd@POL catalyst, toluene solvent, styrene derivative, formic acid, and acetic anhydride into a reaction vessel.
- Stir the reaction mixture at 80°C for approximately 48 hours, monitoring progress via thin-layer chromatography until completion.
- Cool the mixture, filter to recover the heterogeneous catalyst, wash the organic phase with base, acidify the aqueous layer, and extract to isolate the pure phenylpropionic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Pd@POL catalytic method offers tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of homogeneous palladium catalysts removes the necessity for purchasing expensive metal scavengers and reduces the consumption of solvents associated with extensive purification workflows, leading to a significant reduction in raw material costs. Moreover, the simplified workup procedure, which relies on basic filtration and liquid-liquid extraction rather than complex chromatographic separations, drastically shortens the batch cycle time, allowing for increased throughput and better utilization of manufacturing assets. This efficiency gain is particularly valuable in a high-demand market where speed to market for new drug candidates is a critical competitive differentiator.
- Cost Reduction in Manufacturing: The economic benefits of this heterogeneous catalytic system are multifaceted, stemming primarily from the drastic simplification of the downstream processing requirements. By avoiding the use of homogeneous catalysts, manufacturers eliminate the costly and technically challenging step of removing trace heavy metals to parts-per-million levels, a requirement that often dictates the choice of purification technology and drives up processing expenses. The ability to recycle the Pd@POL catalyst for multiple runs without significant loss of activity further amortizes the initial catalyst cost over a larger volume of product, effectively lowering the catalyst cost per kilogram of API intermediate produced. Additionally, the use of formic acid as a safe, liquid carbon monoxide surrogate avoids the infrastructure costs associated with storing and handling high-pressure gas cylinders, reducing capital expenditure and insurance premiums related to hazardous material storage.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route enhances reliability by reducing dependency on specialized reagents and complex equipment. The reagents involved, such as styrenes, formic acid, and toluene, are commodity chemicals with stable global supply chains, minimizing the risk of raw material shortages that can disrupt production schedules. The mild reaction conditions (80°C, atmospheric pressure) allow the process to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, increasing the number of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This flexibility in manufacturing location mitigates geopolitical risks and logistical bottlenecks, ensuring a continuous supply of high-purity phenylpropionic acid intermediates even during periods of global disruption.
- Scalability and Environmental Compliance: The scalability of the Pd@POL method is supported by its inherent safety and environmental profile, which aligns with modern green chemistry principles and regulatory expectations. The heterogeneous nature of the catalyst minimizes the generation of heavy metal-containing wastewater, simplifying effluent treatment and reducing the environmental compliance burden on manufacturing sites. The process operates at moderate temperatures and pressures, reducing energy consumption compared to high-pressure carbonylation methods, which contributes to a lower carbon footprint for the final product. As regulatory agencies worldwide tighten restrictions on residual solvents and heavy metals in pharmaceuticals, adopting this cleaner technology future-proofs the supply chain against potential compliance issues, avoiding costly reformulations or process changes down the line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heterogeneous catalytic technology, derived from the specific experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this synthesis route for their specific applications.
Q: What are the advantages of using Pd@POL over homogeneous palladium catalysts?
A: The Pd@POL heterogeneous catalyst allows for simple filtration and recovery, eliminating the need for complex heavy metal removal steps often required with homogeneous catalysts, thereby reducing production costs and environmental impact.
Q: Can this synthesis method be applied to substituted styrenes?
A: Yes, the patent demonstrates high versatility, successfully synthesizing various derivatives including 3-(4-fluorophenyl)propionic acid, 3-(4-trifluoromethylphenyl)propionic acid, and 3-(3,5-dimethoxyphenyl)propionic acid with good yields.
Q: Is the catalyst reusable for industrial scale-up?
A: Recycling experiments indicate that the Pd@POL catalyst maintains high activity over at least 10 cycles, making it highly suitable for continuous commercial manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylpropionic Acid Supplier
The technological potential of the Pd@POL catalyzed synthesis of phenylpropionic acids represents a significant opportunity for optimizing the production of key pharmaceutical intermediates, and NINGBO INNO PHARMCHEM is uniquely positioned to leverage this innovation for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, guaranteeing that every batch of phenylpropionic acid delivered meets the highest quality standards required by the international pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to evaluate the feasibility of integrating this green catalytic route into your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, as well as obtain specific COA data and route feasibility assessments for your target molecules. Let us partner with you to drive innovation, reduce costs, and secure a sustainable supply of high-quality phenylpropionic acid derivatives for your next-generation therapeutic programs.
