Scalable Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Global Pharma Supply Chains
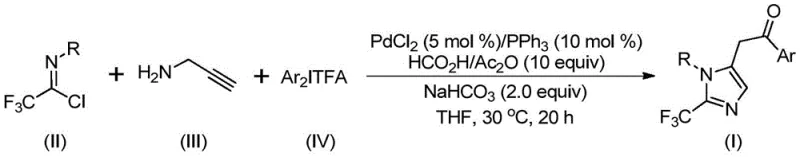
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are critical motifs in modern drug discovery due to their ability to enhance metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing the long-standing challenges associated with introducing trifluoromethyl groups into nitrogen-containing five-membered rings. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. By operating under mild conditions at 30°C, this process not only simplifies the operational complexity but also significantly broadens the utility of trifluoroethylimide acid halides, which have historically been underutilized compared to other trifluoromethyl synthons. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of highly reactive and often unstable synthons like trifluorodiazoethane. As noted in literature such as Angew. Chem. Int. Ed. 2018, while these methods can directly yield trifluoromethyl-substituted heterocycles, they pose significant safety hazards and handling difficulties on a commercial scale. Furthermore, alternative approaches using trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in the availability of safe, scalable routes for these valuable scaffolds. The reliance on harsh conditions or expensive, specialized reagents in conventional pathways often leads to increased production costs and complex waste management issues, creating bottlenecks for supply chain heads who require consistent, high-volume delivery of API intermediates without compromising on safety or purity standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated yet operationally simple palladium-catalyzed system that transforms stable precursors into complex imidazole derivatives with high efficiency. The core innovation lies in the synergistic combination of trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts within a THF solvent system, driven by a PdCl2/PPh3 catalyst and a formic acid/acetic anhydride mixture as a carbon monoxide source. This methodology eliminates the need for dangerous diazo compounds and allows for the precise construction of the imidazole ring with a trifluoromethyl group at the 2-position. The reaction proceeds smoothly at a low temperature of 30°C over 16 to 24 hours, demonstrating exceptional substrate compatibility and functional group tolerance. This温和 (mild) nature of the reaction ensures that sensitive functional groups remain intact, thereby reducing the need for extensive protection-deprotection sequences and streamlining the overall synthetic route for commercial production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
For the technical team, understanding the mechanistic underpinnings of this transformation is crucial for process optimization and troubleshooting. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the palladium catalytic cycle. The palladium catalyst then facilitates the aminopalladation of the alkyne moiety, forming a vinyl-palladium intermediate which further isomerizes to an alkyl-palladium species. A critical step follows where carbon monoxide, released in situ from the decomposition of the formic acid and acetic anhydride mixture, inserts into the palladium-carbon bond to generate an acyl-palladium intermediate. This acyl species is then subjected to oxidative addition by the diaryliodonium salt, forming a high-valent tetravalent palladium complex. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole compound and regenerates the active palladium catalyst, ensuring the continuity and efficiency of the catalytic turnover.
From an impurity control perspective, this mechanism offers distinct advantages. The use of diaryliodonium salts as arylating agents is known for its high chemoselectivity, minimizing the formation of homocoupling byproducts that often plague cross-coupling reactions. Furthermore, the mild reaction temperature of 30°C suppresses thermal degradation pathways and side reactions that typically occur at elevated temperatures, leading to a cleaner crude reaction profile. The specific stoichiometry recommended, such as a molar ratio of trifluoroethylimidoyl chloride to propargylamine to diaryliodonium salt of roughly 1.5:1:1.5, ensures that the limiting reagent is fully consumed while maintaining an excess of the coupling partners to drive the equilibrium forward. This precise control over reaction parameters allows manufacturers to achieve high-purity specifications with minimal downstream purification burden, a key metric for quality assurance in pharmaceutical manufacturing.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, operators should adhere to the standardized protocol outlined in the patent examples, which emphasizes the importance of reagent quality and mixing efficiency. The process begins with the preparation of the catalytic system in an anhydrous environment to prevent catalyst deactivation, followed by the sequential addition of substrates to manage exotherms and ensure homogeneity. While the patent provides specific molar ratios and solvent volumes, scaling this reaction requires careful attention to heat transfer and mass transfer rates, particularly during the carbon monoxide generation phase. Detailed standard operating procedures (SOPs) regarding the handling of diaryliodonium salts and the workup involving silica gel chromatography are essential for reproducibility. For a comprehensive guide on the exact step-by-step execution, please refer to the standardized synthesis instructions provided below.
- Prepare the reaction mixture by adding palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture into an organic solvent such as THF.
- Introduce the key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific diaryliodonium salt into the catalytic system under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, followed by filtration and purification via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The primary benefit stems from the utilization of starting materials that are described as cheap and readily available in nature, such as aromatic amines used to prepare the trifluoroethylimidoyl chloride. This abundance of raw materials mitigates the risk of supply shortages and price volatility that often affects specialty chemical markets. Additionally, the elimination of hazardous reagents like trifluorodiazoethane reduces the regulatory burden and safety infrastructure costs associated with manufacturing, leading to substantial cost savings in facility operations and compliance. The ability to operate at near-ambient temperatures (30°C) further contributes to energy efficiency, lowering the utility costs per kilogram of product produced compared to high-temperature processes.
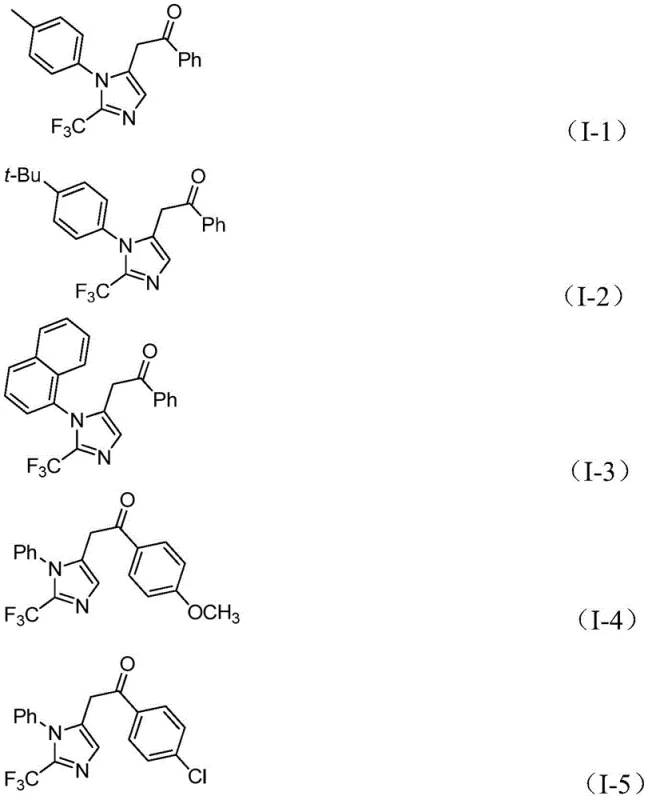
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of palladium chloride, which is noted in the patent as being relatively inexpensive compared to other palladium catalysts, combined with the high reaction efficiency that minimizes catalyst loading requirements. By avoiding complex multi-step sequences and protecting group strategies, the overall material throughput is improved, resulting in a lower cost of goods sold (COGS). The high yields reported, reaching up to 96% for certain substrates, mean that less raw material is wasted, directly translating to better margin protection for the final API. Furthermore, the simplified post-processing, which involves basic filtration and standard column chromatography, reduces the labor and solvent consumption typically associated with difficult purifications.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a reliable supply of high-purity pharmaceutical intermediates, as the method demonstrates excellent compatibility with a wide range of functional groups including methyl, tert-butyl, chloro, bromo, and nitro substituents. This versatility allows manufacturers to produce a diverse library of imidazole derivatives from a single platform technology, reducing the need for multiple dedicated production lines. The scalability of the method, which has been proven effective from milligram to gram scales and is amenable to further expansion, provides supply chain heads with the confidence to plan for long-term commercial production without fearing technical bottlenecks. The use of common solvents like THF also ensures that solvent supply chains remain stable and cost-effective.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a prerequisite for vendor selection, and this process aligns well with green chemistry principles by utilizing formic acid as a safe carbon monoxide surrogate instead of toxic CO gas cylinders. The mild conditions reduce the energy footprint of the reaction, contributing to a lower overall carbon footprint for the manufacturing site. Waste generation is minimized through high atom economy in the coupling steps and the use of recyclable catalysts where possible. The straightforward workup procedure generates less hazardous waste compared to traditional methods involving heavy metal scavengers or aggressive quenching agents, simplifying waste disposal and ensuring compliance with stringent environmental regulations in global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios. We encourage technical teams to review these points carefully to appreciate the full scope of the technology's capabilities.
Q: What are the safety advantages of this method compared to traditional trifluoromethylation?
A: Unlike conventional methods that often rely on unstable and potentially hazardous trifluorodiazoethane, this patented process utilizes stable trifluoroethylimidoyl chloride and diaryliodonium salts, significantly enhancing operational safety and handling ease in industrial settings.
Q: Does this synthesis route support large-scale commercial production?
A: Yes, the patent explicitly demonstrates that the method is scalable to the gram level and beyond. The use of mild reaction conditions (30°C) and commercially available catalysts like PdCl2 facilitates straightforward scale-up for manufacturing high-purity intermediates.
Q: What kind of yields can be expected for diverse substrate scopes?
A: Experimental data within the patent indicates excellent efficiency, with isolated yields reaching up to 96% for specific substrates such as naphthyl-substituted derivatives, demonstrating robust compatibility with various functional groups including halogens and nitro groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111423381A for accelerating drug development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to meet the demanding requirements of global regulatory agencies. We are committed to delivering high-purity 2-trifluoromethyl imidazole intermediates that empower your R&D teams to focus on innovation rather than supply constraints.
We invite you to engage with our technical procurement team to discuss how this specific technology can be tailored to your project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this route for your specific target molecules. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your trusted partner in the synthesis of complex heterocyclic intermediates. Let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
