Advanced Cobalt-Catalyzed Hydrocarbon Amination for Scalable Carbazole Production
Advanced Cobalt-Catalyzed Hydrocarbon Amination for Scalable Carbazole Production
The landscape of fine chemical synthesis is undergoing a transformative shift towards sustainable and cost-effective catalytic systems, particularly for high-value nitrogen-containing heterocycles. A groundbreaking development in this sector is detailed in patent CN113004248A, which discloses a novel method for synthesizing carbazole compounds via a cobalt-catalyzed hydrocarbon amination reaction. This technology addresses critical bottlenecks in the production of carbazole derivatives, which are indispensable scaffolds in the pharmaceutical and optoelectronic industries. As illustrated in the structural diversity of bioactive molecules and functional materials, carbazoles serve as the core backbone for anticancer agents, anti-HIV drugs, and advanced OLED materials. The ability to construct these complex architectures efficiently is paramount for meeting the growing global demand for high-purity pharmaceutical intermediates.
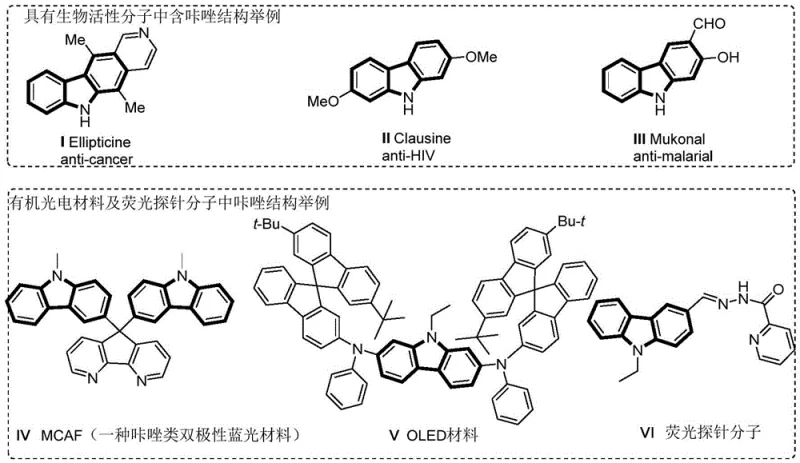
For R&D directors and process chemists, the significance of this patent lies in its departure from traditional reliance on scarce precious metals. By leveraging earth-abundant cobalt, the methodology not only aligns with green chemistry principles but also offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The reaction utilizes pyridine amide compounds as precursors, undergoing an intramolecular cyclization facilitated by a cobalt catalyst, an oxidant, and a base in an organic solvent. This approach eliminates the need for expensive ligands often required in noble metal catalysis, thereby streamlining the synthetic route and reducing the overall impurity profile of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbazole derivatives has been dominated by transition metal-catalyzed C-H activation strategies, predominantly utilizing rhodium and palladium. While effective, these conventional methods suffer from significant economic and operational drawbacks that hinder large-scale manufacturing. For instance, prior art reported by Brankica Jovanovic et al. in 2009 utilized rhodium catalysis for bisaryl azide synthesis via nitrogen carbene insertion. Although chemically feasible, this route necessitates the use of expensive rhodium catalysts and often requires additional ligands to control regioselectivity, drastically inflating the production cost. Similarly, methods reported by So Won Youn in 2011 employed palladium-catalyzed intramolecular cyclization of N-p-toluenesulfonyl o-aminobiphenyl. These palladium-based routes, while versatile, introduce challenges related to residual metal contamination, which is a critical concern for pharmaceutical grade intermediates.
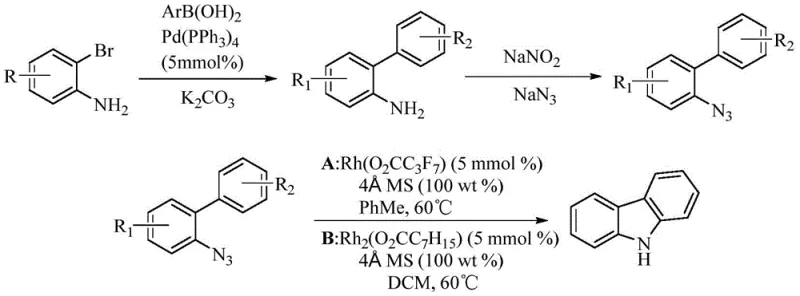
Furthermore, alternative approaches such as the palladium-catalyzed dearomatization-re-aromatization strategy reported by Huiying Zeng in 2020 face limitations regarding substrate availability. The requirement for difficult-to-synthesize diphenol compounds as starting materials restricts the practical utility of such methods for broad industrial application. The cumulative effect of these limitations—high catalyst cost, ligand dependency, difficult substrate synthesis, and potential heavy metal residues—creates a pressing need for a more economical and operationally simple synthetic strategy. The reliance on precious metals also exposes supply chains to geopolitical risks and price volatility, making the search for base metal alternatives a strategic priority for procurement managers.
The Novel Approach
The invention disclosed in CN113004248A presents a paradigm shift by introducing a cobalt-catalyzed system that circumvents the aforementioned limitations. This novel approach utilizes inexpensive cobalt salts, such as cobalt chloride, cobalt acetate, or basic cobalt carbonate, to catalyze the hydrocarbon amination of pyridine amide compounds. Unlike the rhodium and palladium counterparts, this system operates efficiently without the need for specialized, costly ligands. The reaction mechanism involves the activation of the C-H bond adjacent to the nitrogen atom, followed by cyclization to form the carbazole core. This direct functionalization strategy simplifies the synthetic sequence, reducing the number of steps and reagents required.

Moreover, the novel approach demonstrates superior reaction yields and broader substrate tolerance. Experimental data indicates that the cobalt catalyst effectively promotes the cyclization of various substituted pyridine amides, accommodating electron-donating and electron-withdrawing groups alike. The use of common oxidants like silver carbonate and bases like sodium carbonate further enhances the practicality of the method. By replacing precious metals with base metals, this technology offers a viable solution for cost reduction in fine chemical manufacturing, enabling the production of high-purity carbazole intermediates at a fraction of the traditional cost. This advancement is particularly relevant for the commercial scale-up of complex polymer additives and electronic chemicals where margin pressure is high.
Mechanistic Insights into Cobalt-Catalyzed Hydrocarbon Amination
From a mechanistic perspective, the cobalt-catalyzed hydrocarbon amination represents a sophisticated interplay between the metal center, the oxidant, and the substrate. The catalytic cycle likely initiates with the coordination of the cobalt species to the nitrogen atom of the pyridine amide directing group. This coordination facilitates the activation of the proximal aromatic C-H bond through a concerted metalation-deprotonation (CMD) pathway or a radical mechanism, depending on the specific oxidation state of the cobalt. The presence of the oxidant, typically silver carbonate, plays a dual role: it regenerates the active cobalt species from lower oxidation states formed during the cycle and may also participate in the oxidative C-N bond formation step. The base, such as sodium carbonate, assists in the deprotonation steps necessary for the restoration of aromaticity in the final carbazole product.
Impurity control is a critical aspect of this mechanism, especially for pharmaceutical applications. The mild reaction conditions (110-130°C) and the specific selectivity of the cobalt catalyst minimize side reactions such as over-oxidation or non-selective C-H activation at remote positions. The use of a pyridine amide directing group ensures high regioselectivity, guiding the cyclization to occur exclusively at the ortho-position relative to the amide linkage. This intrinsic selectivity reduces the formation of regioisomers, simplifying downstream purification processes. Furthermore, the absence of phosphine ligands, which are prone to oxidation and can generate difficult-to-remove phosphine oxide impurities, contributes to a cleaner reaction profile. This mechanistic robustness ensures that the resulting carbazole compounds meet stringent purity specifications required for API intermediates and high-performance electronic materials.
How to Synthesize Carbazole Compounds Efficiently
The practical implementation of this cobalt-catalyzed methodology is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves charging a sealed reaction vessel with the pyridine amide substrate, a catalytic amount of cobalt salt (typically 5-10 mol%), a stoichiometric amount of oxidant (e.g., Ag2CO3), and a base. The mixture is suspended in a suitable organic solvent such as toluene, dichloroethane, or fluorobenzene. The reaction is then heated to approximately 120°C and stirred for 10 to 20 hours. Upon completion, the solvent is removed under reduced pressure, and the crude product is purified via column chromatography. This standardized protocol allows for the rapid screening of substrates and optimization of conditions for specific derivatives.
- Combine N-(2-phenylphenyl)pyridinecarboxamide substrate with a cobalt catalyst (e.g., basic cobalt carbonate), an oxidant (e.g., silver carbonate), and a base (e.g., sodium carbonate) in an organic solvent like toluene.
- Seal the reaction vessel and heat the mixture to a temperature between 110°C and 130°C, maintaining stirring for a duration of 10 to 20 hours to ensure complete cyclization.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using column chromatography to isolate the high-purity carbazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By substituting rhodium or palladium, which are subject to extreme price volatility and supply constraints, with cobalt, manufacturers can achieve significant cost savings in the production of carbazole intermediates. This cost efficiency is compounded by the elimination of expensive ligands, which are often proprietary and add substantial overhead to the bill of materials. The simplified reagent profile also reduces the complexity of inventory management and sourcing.
- Cost Reduction in Manufacturing: The transition from precious metal catalysis to base metal catalysis fundamentally alters the cost structure of carbazole synthesis. Cobalt is significantly more abundant and less expensive than rhodium or palladium, leading to a direct decrease in catalyst expenditure. Additionally, the ligand-free nature of the reaction removes the need for purchasing specialized organic ligands, further lowering the variable costs per kilogram of product. The high yields reported in the patent examples suggest that material throughput is maximized, reducing waste and improving overall process economy. These factors collectively contribute to a more competitive pricing model for the final carbazole derivatives.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like cobalt mitigates the supply chain risks associated with precious metals, which are often concentrated in geopolitically unstable regions. Cobalt supply chains are well-established due to its widespread use in batteries and alloys, ensuring a stable and continuous supply of the catalyst. Furthermore, the reagents used in this process, such as silver carbonate and sodium carbonate, are commodity chemicals available from multiple global suppliers. This diversification of the supply base enhances resilience against disruptions, ensuring consistent delivery schedules for downstream customers in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The operational simplicity of the cobalt-catalyzed process facilitates easy scale-up from gram to ton quantities. The reaction conditions are moderate, avoiding the need for extreme pressures or cryogenic temperatures, which simplifies reactor design and safety protocols. From an environmental perspective, the use of a base metal aligns with green chemistry initiatives, potentially reducing the regulatory burden associated with heavy metal discharge limits. The simplified workup procedure, involving standard solvent removal and chromatography, minimizes the generation of complex waste streams, supporting sustainable manufacturing practices and compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the cobalt-catalyzed synthesis of carbazoles. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of using cobalt over rhodium for carbazole synthesis?
A: The primary advantage is cost efficiency. Rhodium is a precious metal with high market volatility and cost, often requiring expensive ligands. Cobalt is an earth-abundant base metal that significantly lowers raw material costs while maintaining high catalytic activity and yield.
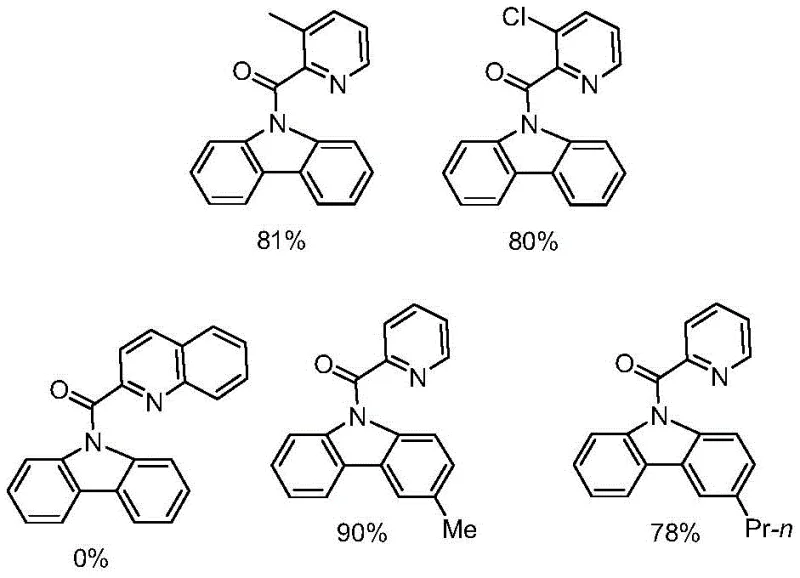
Q: What is the substrate scope for this cobalt-catalyzed reaction?
A: The method demonstrates broad substrate tolerance. It effectively processes pyridine amide compounds with various substituents, including methyl, chlorine, and propyl groups on the aromatic rings, yielding diverse carbazole derivatives suitable for different pharmaceutical and material applications.
Q: Does this method require specialized ligands or harsh conditions?
A: No, one of the key innovations is the ligand-free nature of the catalytic system. The reaction proceeds efficiently under relatively mild thermal conditions (120°C) using standard oxidants and bases, simplifying the operational complexity compared to traditional noble metal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cobalt-catalyzed hydrocarbon amination technology for the production of high-value carbazole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of carbazole derivative we produce. We are committed to delivering high-purity OLED material and pharmaceutical intermediates that meet the exacting standards of the global market.
We invite R&D directors and procurement specialists to collaborate with us to leverage this cost-effective synthesis route for their specific projects. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your target carbazole compounds. Let us help you optimize your supply chain and reduce manufacturing costs through the adoption of this advanced cobalt-catalyzed technology.
