Advanced Electrochemical Manufacturing of High-Purity 4-Selenopyrazole Derivatives for Global Pharma
Introduction to Next-Generation Electroorganic Synthesis
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards greener, more sustainable manufacturing technologies, driven by the urgent need to reduce environmental footprints and eliminate toxic impurities. A pivotal development in this arena is documented in Chinese patent CN113046772B, which discloses a novel electrochemical synthesis method for 4-selenopyrazole derivatives. These compounds are critical scaffolds in medicinal chemistry, known for their broad biological activities and potential applications in drug discovery. Unlike traditional methods that rely heavily on stoichiometric oxidants and transition metal catalysts, this innovative approach utilizes electrons as a trace-free oxidant, enabling the cyclization-coupling domino reaction of aryl hydrazines, 1,3-propanediones, and diselenides under remarkably mild conditions. This technology represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediates supplier, as it directly addresses the stringent purity requirements of modern drug development while simultaneously offering a pathway for cost reduction in pharmaceutical intermediates manufacturing through simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
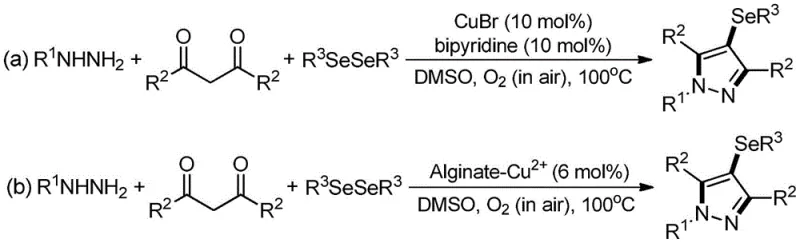
Prior to this electrochemical breakthrough, the synthesis of 4-selenopyrazole derivatives was predominantly achieved through transition metal-catalyzed oxidative cyclization. As illustrated in earlier literature, such as the work by Raquel G. Jacob et al., these conventional protocols typically employed cuprous bromide (CuBr) as a catalyst and bipyridine as a ligand, requiring high temperatures around 100°C and molecular oxygen from air as the terminal oxidant. While chemically feasible, these conditions present severe drawbacks for industrial scale-up, particularly in the pharmaceutical sector where metal contamination is strictly regulated. The use of copper salts inevitably leads to metal residue issues, necessitating expensive and time-consuming purification steps such as chromatography or specialized scavenging to meet regulatory standards. Furthermore, the combination of high temperatures, organic solvents like DMSO, and oxidants creates significant safety hazards, including flammability and explosion risks, which complicate the commercial scale-up of complex pharmaceutical intermediates and increase the overall operational expenditure.
The Novel Approach
In stark contrast, the electrochemical method described in patent CN113046772B offers a transformative solution by replacing chemical oxidants with electricity. This metal-free protocol operates at room temperature in an undivided cell, utilizing a simple electrolyte system composed of lithium tetrafluoroborate (LiBF4) in acetonitrile. The reaction proceeds via a one-pot cyclization-coupling domino mechanism that achieves high selectivity and yields often exceeding 90% without the need for external oxidants or transition metals. By eliminating the copper catalyst entirely, this method inherently solves the metal residue problem, drastically simplifying the purification workflow and enhancing the safety profile of the manufacturing process. The ability to conduct this transformation under ambient conditions not only reduces energy consumption but also minimizes the formation of thermal byproducts, resulting in a cleaner crude reaction mixture that is far easier to isolate. This advancement positions the technology as a premier choice for reducing lead time for high-purity pharmaceutical intermediates, as the streamlined workup allows for faster turnover from synthesis to final quality control.
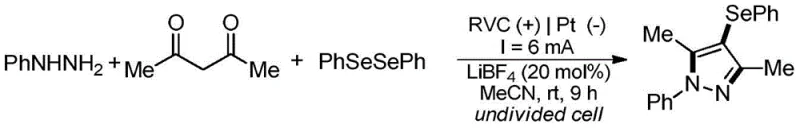
Mechanistic Insights into Electrochemical Cyclization-Coupling
Understanding the mechanistic underpinnings of this electrochemical transformation is crucial for R&D directors evaluating its robustness and scalability. The reaction initiates at the anode surface, where the diselenide compound undergoes a single-electron oxidation to generate a diselenide radical cation intermediate. This unstable species rapidly dissociates into a phenylselenyl cation and a phenylselenyl radical. Concurrently, the aryl hydrazine and 1,3-propanedione undergo a condensation and cyclization sequence to form a pyrazole intermediate. The phenylselenyl radical then attacks this pyrazole core to generate a radical intermediate, which is subsequently oxidized at the anode to form a cationic species. Finally, deprotonation yields the target 4-selenopyrazole product. At the cathode, the phenylselenyl cation can be reduced back to the diselenide, effectively recycling some of the selenium source, while protons are reduced to hydrogen gas. This elegant interplay of anodic oxidation and cathodic reduction ensures high atom efficiency and minimizes waste generation.
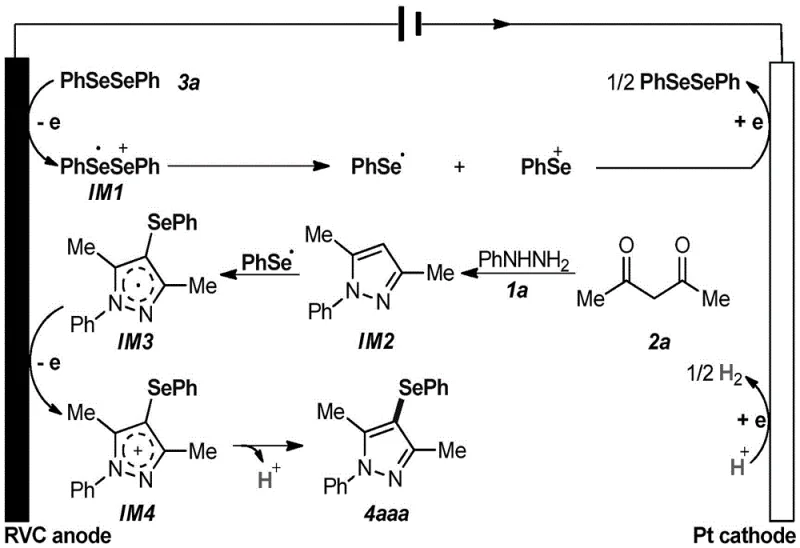
From an impurity control perspective, the absence of transition metals eliminates a major class of hard-to-remove contaminants that often plague batch chemical synthesis. The use of electrons as the reagent means that the "oxidant" leaves no chemical footprint, avoiding the salt waste associated with chemical oxidants like peroxides or hypervalent iodine. Furthermore, the mild room temperature conditions suppress thermal decomposition pathways that typically lead to tar formation or polymerization in high-temperature processes. The patent data indicates that the reaction is highly tolerant to various substituents on the aryl rings, including electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and methyl. This broad substrate scope suggests that the electrochemical potential is well-tuned to activate the specific bonds required for cyclization without affecting sensitive functional groups elsewhere in the molecule, ensuring a consistent and predictable impurity profile across different derivatives.
How to Synthesize 4-Selenopyrazole Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize yield and reproducibility. The standard procedure involves dissolving the aryl hydrazine, 1,3-propanedione, and diselenide in acetonitrile with 20 mol% LiBF4 as the supporting electrolyte. The reaction is conducted in an undivided cell equipped with a reticulated vitreous carbon (RVC) anode and a platinum sheet cathode, applying a constant current of 6 mA for 9 hours at room temperature. Optimization studies within the patent reveal that the choice of electrode material is critical; while graphite and nickel can function, the RVC/Pt pair delivers superior performance due to its high surface area and stability. Similarly, the electrolyte concentration and solvent choice are pivotal, with acetonitrile proving far superior to polar aprotic solvents like DMF or DMSO. For detailed operational parameters and troubleshooting tips, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the electrolyte solution by dissolving aryl hydrazine, 1,3-propanedione, and diselenide compounds in acetonitrile with LiBF4 as the supporting electrolyte.
- Insert a reticulated vitreous carbon (RVC) anode and a platinum sheet cathode into the undivided cell containing the reaction mixture.
- Apply a constant direct current of 6 mA at room temperature for approximately 9 hours, then isolate the product via solvent removal and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from thermal copper catalysis to electrochemical synthesis offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials and the reduction of downstream processing costs. By removing the requirement for expensive transition metal catalysts and ligands, the bill of materials is streamlined, and the dependency on volatile metal markets is reduced. Moreover, the elimination of metal scavengers and extensive chromatographic purification steps significantly shortens the production cycle time, allowing for faster fulfillment of orders and improved responsiveness to market demand. This efficiency gain is compounded by the enhanced safety profile, which lowers insurance premiums and reduces the regulatory burden associated with handling hazardous oxidants and high-temperature reactions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the complete removal of transition metal catalysts, which are not only costly to purchase but also expensive to remove to meet ppm-level specifications required by pharma clients. Without copper residues, the need for specialized metal scavenging resins or repeated recrystallizations is eliminated, leading to substantial cost savings in both materials and labor. Additionally, operating at room temperature rather than 100°C results in significantly lower energy consumption for heating and cooling, further driving down the utility costs per kilogram of product. The high atom efficiency of the electrochemical process also means less raw material is wasted in side reactions, maximizing the yield from every batch and improving the overall cost-effectiveness of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this electrochemical method contributes to a more resilient supply chain by reducing the complexity of the manufacturing process. The starting materials—aryl hydrazines, 1,3-propanediones, and diselenides—are commercially available commodity chemicals, ensuring a stable and continuous supply of feedstock without reliance on specialized catalysts that may have long lead times. The mild reaction conditions also reduce the wear and tear on reactor equipment, minimizing unplanned downtime for maintenance and repair. This reliability ensures consistent delivery schedules for clients, mitigating the risk of production delays that can disrupt downstream drug development timelines. Furthermore, the scalability of electrochemical flow or batch processes allows for flexible production volumes, adapting easily to fluctuations in demand without compromising product quality.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this metal-free synthesis aligns perfectly with green chemistry principles and increasingly strict global environmental regulations. The absence of heavy metal waste simplifies wastewater treatment and disposal, reducing the environmental compliance costs and the carbon footprint of the manufacturing facility. The use of electrons as a clean reagent eliminates the generation of stoichiometric oxidant byproducts, resulting in a cleaner E-factor (mass of waste per mass of product). This sustainability advantage is increasingly valued by multinational corporations seeking to green their supply chains, making suppliers who adopt such technologies preferred partners. The simplicity of the workup, often requiring only solvent removal and basic chromatography, facilitates easier scale-up from laboratory to pilot and commercial production scales without the need for complex engineering modifications.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical feasibility and commercial viability of this electrochemical synthesis, we have compiled answers based on the specific data points and experimental results detailed in patent CN113046772B. These questions cover critical aspects such as electrode selection, substrate compatibility, and reaction optimization, providing clarity for technical teams evaluating this route for their own pipelines. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: Why is the electrochemical method superior to copper-catalyzed synthesis for pharmaceutical intermediates?
A: The electrochemical method eliminates the need for transition metal catalysts like copper bromide, thereby removing the risk of toxic metal residues in the final API intermediate. It also operates at room temperature rather than 100°C, significantly reducing energy consumption and safety risks associated with high-temperature oxidation.
Q: What are the optimal electrode materials for this 4-selenopyrazole synthesis?
A: According to patent CN113046772B, the optimal electrode pair consists of a reticulated vitreous carbon (RVC) mesh anode and a platinum (Pt) sheet cathode. This combination provides the highest current efficiency and yield compared to graphite or nickel alternatives.
Q: Does this process support a wide range of substrate functional groups?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully accommodates aryl hydrazines and diselenides with electron-donating groups (like methyl, methoxy) and electron-withdrawing groups (like fluoro, chloro, trifluoromethyl, nitro), maintaining high yields across diverse substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Selenopyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-quality chemical building blocks for the global pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113046772B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-selenopyrazole derivative we supply is free from metal contaminants and ready for immediate use in drug discovery programs. We are committed to leveraging cutting-edge technologies to provide our clients with a competitive edge in terms of both quality and speed to market.
We invite you to collaborate with us to explore how this metal-free electrochemical route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this greener methodology can impact your bottom line. Please contact us today to request specific COA data for our available 4-selenopyrazole inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a sustainable and efficient supply chain for your critical pharmaceutical intermediates.
