Advanced Manufacturing of Alkyne Intermediates for Cardiovascular Drug Synthesis
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, particularly those serving as precursors for cardiovascular medications. Patent CN1296342C discloses a groundbreaking production method for a specific alkyne compound, designated as formula (3), which serves as a vital intermediate in the synthesis of anti-atrial fibrillation and antihypertensive agents. This technology represents a significant paradigm shift from traditional methodologies by utilizing 4-nitrofluorobenzene as a key starting material. Unlike conventional approaches that rely on unstable halides or expensive coupling reagents, this novel process leverages the nucleophilic aromatic substitution capabilities of alkoxides derived from 2-methyl-3-butyn-2-ol. The innovation lies not just in the chemical transformation but in the precise control of reaction parameters, specifically maintaining temperatures between -20°C and 10°C. This strict thermal regulation is crucial for maximizing yield while suppressing the formation of persistent impurities that often plague downstream processing. For R&D directors and process chemists, this patent offers a blueprint for a cleaner, more efficient synthesis that aligns with modern green chemistry principles by eliminating heavy metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific alkyne framework has been fraught with chemical and operational challenges that hinder industrial scalability. Traditional methods often involve the reaction of 4-nitrophenol with 2-methyl-3-butyne-2-chloride in the presence of a base. As illustrated in the structural comparisons below, the reliance on the chloro-derivative (Compound 6) introduces significant instability issues, as this reagent is prone to decomposition and requires careful handling. Furthermore, alternative routes utilizing copper catalysts, while offering moderate yield improvements, introduce the severe complication of heavy metal contamination. Removing trace copper to meet pharmaceutical grade specifications requires additional, costly purification steps such as chelation or specialized filtration. Other methods employing Mitsunobu conditions with reagents like DEAD are economically prohibitive due to the high cost of azodicarboxylates and the generation of stoichiometric phosphine oxide waste. These legacy processes suffer from low atom economy, extended reaction times spanning several days, and yields that frequently struggle to exceed 50%, creating a bottleneck for reliable supply chains.
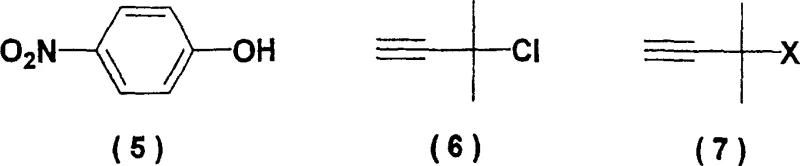
The Novel Approach
The methodology described in CN1296342C fundamentally re-engineers the synthetic pathway by substituting the problematic phenol and chloride partners with 4-nitrofluorobenzene and a pre-formed alkoxide. This strategic change capitalizes on the high reactivity of the fluorine leaving group in electron-deficient aromatic systems, facilitating a smooth nucleophilic attack without the need for transition metal catalysis. The reaction proceeds efficiently in polar aprotic solvents such as N,N-dimethylacetamide (DMAc) or N,N'-dimethylimidazolidinone (DMI). By generating the sodium alkoxide of 2-methyl-3-butyn-2-ol in situ and reacting it with the fluorobenzene derivative at controlled low temperatures, the process achieves exceptional conversion rates. The visual representation of this core transformation highlights the direct displacement of the fluorine atom, resulting in the desired carbon-oxygen bond formation with high fidelity. This approach not only simplifies the reaction setup by removing the need for inert atmosphere conditions typically required for sensitive organometallic reagents but also drastically reduces the environmental footprint by avoiding toxic metal waste streams.
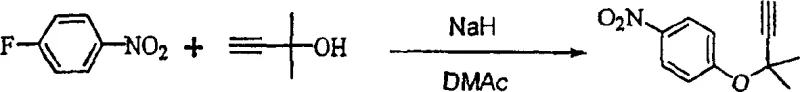
Mechanistic Insights into Nucleophilic Aromatic Substitution
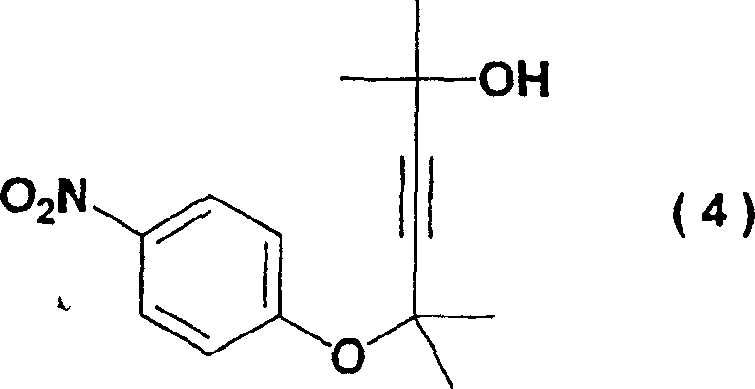
The success of this synthesis hinges on the mechanistic nuances of nucleophilic aromatic substitution (SnAr) involving activated nitro-aromatics. In this system, the nitro group at the para-position strongly withdraws electron density from the aromatic ring, thereby activating the carbon-fluorine bond towards nucleophilic attack by the alkoxide anion. However, the reaction landscape is complicated by the potential for competitive side reactions. One major challenge identified in prior art is the formation of byproduct (4), an ether derivative resulting from O-alkylation or rearrangement pathways that are thermodynamically favored at higher temperatures. The patent data explicitly demonstrates that maintaining the reaction temperature within the narrow window of -20°C to 10°C is critical. At these cryogenic to near-freezing conditions, the kinetic pathway leading to the desired C-O bond formation is favored, while the activation energy required for the formation of the stubborn byproduct (4) is not readily overcome. This temperature control effectively acts as a chemical gatekeeper, ensuring high selectivity.
Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition state and solvating the ionic intermediates. Polar aprotic solvents like DMAc enhance the nucleophilicity of the alkoxide anion by solvating the cation without hydrogen bonding to the nucleophile itself. The patent emphasizes that the ratio of solvent to substrate is another critical variable; using at least two times the mass of solvent relative to the fluorobenzene substrate helps dissipate the heat of reaction and maintains homogeneity, further preventing localized hot spots that could trigger byproduct formation. This deep understanding of the interplay between temperature, solvent polarity, and stoichiometry allows for the reproducible manufacture of high-purity intermediates. For quality control teams, this means a much cleaner impurity profile, reducing the burden on analytical laboratories to detect and quantify trace contaminants that could affect the safety profile of the final drug substance.
How to Synthesize Alkyne Compound Efficiently
The practical execution of this synthesis requires precise adherence to the optimized conditions outlined in the patent examples to ensure maximum efficiency and safety. The process begins with the careful generation of the alkoxide species, followed by the controlled addition of the electrophile. Detailed operational parameters regarding addition rates, cooling capacities, and workup procedures are essential for translating this laboratory-scale success to pilot and commercial production environments. The following guide summarizes the critical operational steps derived from the patent's exemplary embodiments.
- Preparation of Alkoxide: React 2-methyl-3-butyn-2-ol with sodium hydride in an amide solvent like DMAc at ice-cooled temperatures to form the stable sodium alkoxide.
- Nucleophilic Substitution: Slowly add 4-nitrofluorobenzene to the alkoxide solution while maintaining the temperature between -20°C and 10°C to ensure high conversion and minimize byproducts.
- Workup and Purification: Quench the reaction with water, extract with an organic solvent such as toluene, and purify the crude product via column chromatography or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their sourcing of pharmaceutical intermediates. The shift away from unstable and hazardous reagents like 2-methyl-3-butyne-2-chloride significantly enhances supply chain reliability. This chloro-compound is known for its sensitivity and limited shelf-life, often necessitating specialized storage and expedited shipping, which drives up logistics costs. By replacing it with stable, commodity-grade 4-nitrofluorobenzene and 2-methyl-3-butyn-2-ol, manufacturers can secure long-term contracts with multiple suppliers, mitigating the risk of raw material shortages. Additionally, the elimination of expensive catalysts such as copper iodide or specialized ligands like DBU removes a significant cost driver from the bill of materials. These reagents not only carry high price tags but also require rigorous recovery or disposal protocols to meet environmental regulations.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the downstream processing workflow. In traditional copper-catalyzed methods, the removal of residual heavy metals is a mandatory and costly step, often requiring scavenger resins or multiple recrystallizations to meet strict ICH guidelines for elemental impurities. By operating under metal-free conditions, this novel method completely bypasses the need for metal scavenging, thereby reducing both material costs and processing time. Furthermore, the high yield achieved (often exceeding 90% in optimized examples) means less raw material is wasted per kilogram of product produced. The use of recoverable amide solvents also contributes to a lower overall cost of goods sold, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 4-Nitrofluorobenzene is a bulk chemical produced on a multi-ton scale for various agrochemical and pharmaceutical applications, ensuring a steady and predictable supply. Unlike specialized reagents that may have single-source bottlenecks, the commoditization of the inputs allows for flexible sourcing strategies. Moreover, the robustness of the reaction conditions—specifically the tolerance for standard industrial equipment without the need for exotic high-pressure or ultra-low-temperature reactors—means that production can be easily transferred between different manufacturing sites. This flexibility is crucial for maintaining continuity of supply in the face of regional disruptions or capacity constraints at specific facilities.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of exothermic runaway risks associated with unstable chlorides. The controlled addition of reagents at low temperatures is a standard unit operation in modern chemical plants, posing minimal engineering challenges. From an environmental standpoint, the process generates significantly less hazardous waste. The avoidance of phosphine oxides (from Mitsunobu reactions) and heavy metal sludge simplifies wastewater treatment and solid waste disposal. This alignment with green chemistry principles not only reduces disposal costs but also enhances the sustainability profile of the supply chain, a factor increasingly weighted in vendor selection criteria by major pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and operational feasibility. Understanding these details is essential for technical teams evaluating the viability of this route for their specific project timelines and quality requirements.
Q: Why is 4-nitrofluorobenzene preferred over 4-nitrophenol for this synthesis?
A: Using 4-nitrofluorobenzene avoids the formation of difficult-to-remove byproducts associated with phenol-based routes and eliminates the need for unstable chloro-alkyne reagents, significantly improving process stability and yield.
Q: How does the new method control the formation of byproduct (4)?
A: The process strictly controls the reaction temperature between -20°C and 10°C and optimizes solvent ratios, which kinetically suppresses the side reactions that lead to the formation of the ether byproduct (4).
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes stable, commercially available raw materials and avoids expensive heavy metal catalysts or complex purification steps, making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyne Compound Supplier
The technological advancements detailed in CN1296342C underscore the potential for producing high-value pharmaceutical intermediates with superior efficiency and purity. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative synthetic routes for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from process development to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for API synthesis. Our commitment to quality ensures that the impurity profiles, particularly regarding the suppression of byproduct (4), are tightly controlled through precise process parameter monitoring.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for cardiovascular drug intermediates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of advanced nucleophilic substitution chemistry can deliver reliable, cost-effective, and high-purity solutions for your critical drug development programs.
