Advanced Synthesis of Polysubstituted Pyrrole Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Polysubstituted Pyrrole Derivatives for Commercial Pharmaceutical Applications
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and material science industries, particularly for heterocyclic compounds that serve as critical building blocks. A significant breakthrough in this domain is detailed in patent CN113200902A, which discloses a highly efficient preparation method for polysubstituted pyrrole derivatives. This technology leverages a sophisticated cascade reaction involving carbon insertion, cyclization, and [1,5] migration to construct the pyrrole core with exceptional precision. For R&D directors and procurement specialists seeking reliable sources for complex heterocycles, this methodology represents a paradigm shift from traditional, labor-intensive routes to a streamlined, high-yield process. The ability to synthesize these valuable scaffolds from simple, commercially available precursors under mild conditions addresses long-standing challenges in supply chain stability and cost efficiency for high-purity pharmaceutical intermediates.
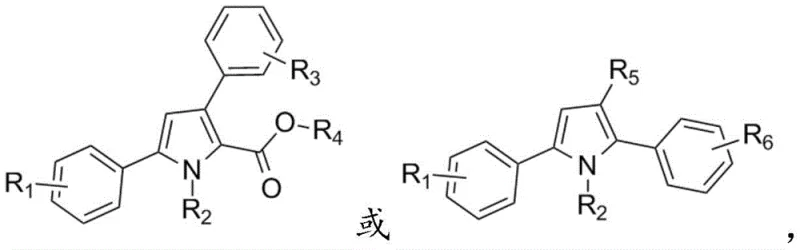
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole rings has relied on classical methodologies such as the Knorr, Paal-Knorr, and Hantzsch syntheses, which, while foundational, often suffer from significant limitations in a modern commercial context. These traditional routes frequently necessitate harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can degrade sensitive functional groups and lead to complex impurity profiles that are difficult to separate. Furthermore, many modern transition-metal catalyzed approaches, although effective, often depend on expensive catalysts, specialized ligands, and substrates that are themselves difficult and costly to prepare. For a procurement manager, these factors translate into volatile pricing, extended lead times for raw materials, and increased waste disposal costs, creating bottlenecks in the manufacturing of active pharmaceutical ingredients (APIs) and advanced materials.
The Novel Approach
The methodology described in the patent offers a robust alternative by utilizing a cascade reaction between amino-substituted unsaturated carbonyl compounds and either diazo compounds or phenylhydrazone compounds. This approach successfully constructs the pyrrole ring through a concerted mechanism that avoids the pitfalls of stepwise synthesis. By employing readily accessible starting materials like acetophenone derivatives and anilines, the process drastically simplifies the supply chain logistics. The reaction conditions are notably mild, typically ranging from 0°C to 130°C depending on the specific pathway chosen, which enhances safety and reduces energy consumption. This novel strategy not only achieves high yields but also ensures that the resulting polysubstituted pyrrole derivatives possess the structural diversity required for next-generation drug candidates, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
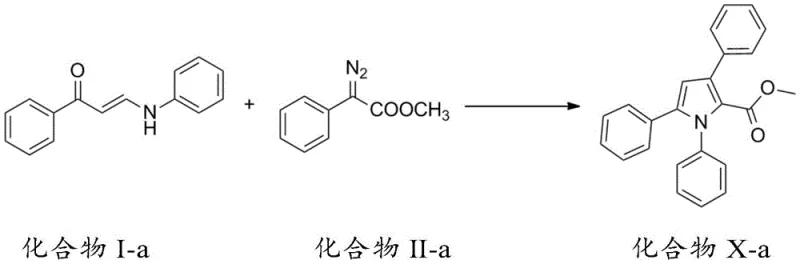
Mechanistic Insights into Silver-Catalyzed Cascade Cyclization
The core of this synthetic innovation lies in the intricate mechanistic pathway that transforms linear precursors into the cyclic pyrrole architecture. The reaction initiates with the interaction between the amino-substituted unsaturated carbonyl compound (Compound I) and the diazo species (Compound II) or phenylhydrazone (Compound III) under the influence of a metal catalyst, such as silver trifluoromethanesulfonate or rhodium acetate. This catalytic system facilitates a carbon-carbon insertion followed by a rapid cyclization event. Crucially, the process involves a [1,5] migration cascade that rearranges the molecular framework to stabilize the five-membered nitrogen-containing ring. For R&D teams, understanding this mechanism is vital as it explains the high regioselectivity observed in the products, ensuring that the substituents are positioned exactly where needed for biological activity without generating significant amounts of regioisomers that would complicate downstream purification.
Impurity control is inherently built into this mechanism due to the mild thermal conditions and the specificity of the metal catalyst. Unlike free-radical processes that can generate a myriad of side products, this coordinated insertion and migration pathway channels the reactants efficiently toward the desired pyrrole product. The use of solvents like dichloromethane or toluene further aids in maintaining a homogeneous reaction environment, minimizing localized hot spots that could trigger decomposition. This level of control is paramount for producing high-purity OLED materials or pharmaceutical intermediates where trace impurities can have detrimental effects on performance or safety. The ability to tune the electronic properties of the catalyst and the steric environment of the substrates allows chemists to optimize the reaction for specific derivatives, ensuring consistent quality across different batches.
How to Synthesize Polysubstituted Pyrrole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the preparation of the key intermediates and the precise control of the cyclization step. The process begins with the formation of the enaminone precursor, followed by the generation of the reactive diazo or hydrazone species. Once these components are ready, the cyclization is performed under inert atmosphere to prevent oxidation of sensitive intermediates. The patent provides detailed examples demonstrating that slow addition of the diazo compound can further enhance yield and selectivity by managing the concentration of reactive species in the pot. For those looking to replicate this high-efficiency protocol, the detailed standardized synthesis steps are outlined in the guide below.
- Preparation of Compound I: React acetophenone derivatives with N,N-dimethylformamide methylal to form an enaminone intermediate, then react with an amine to obtain the amino-substituted unsaturated carbonyl compound.
- Preparation of Reagents: Synthesize diazo compounds (Compound II) via diazotization of esters or prepare phenylhydrazone compounds (Compound III) from ketones and sulfonyl hydrazides.
- Cyclization Reaction: Mix Compound I with Compound II or III in the presence of a silver or rhodium catalyst (3-10 mol%) in solvents like dichloromethane or toluene at 0-130°C to induce cyclization and [1,5] migration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly impact the bottom line and operational reliability for chemical manufacturers. The primary benefit stems from the use of commodity chemicals as starting materials; acetophenones, anilines, and simple esters are produced on a massive global scale, ensuring a stable supply and competitive pricing compared to specialized heterocyclic building blocks. This accessibility eliminates the risk of supply chain disruptions often associated with niche reagents. Furthermore, the elimination of complex multi-step sequences reduces the overall processing time and labor costs, allowing for faster turnaround from order to delivery. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and a more resilient production schedule that can adapt to fluctuating market demands without significant retooling.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the avoidance of expensive transition metal catalysts in large quantities; typically, only 3 mol% to 10 mol% of silver or rhodium catalyst is required, which can often be recovered or used in minimal amounts relative to the product value. Additionally, the mild reaction conditions mean that standard glass-lined or stainless steel reactors can be used without the need for specialized high-pressure or cryogenic equipment, leading to substantial capital expenditure savings. The high yields reported, often exceeding 85-90% in examples, minimize raw material waste and maximize output per batch, significantly lowering the cost of goods sold (COGS) for the final polysubstituted pyrrole products.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with pharmaceutical clients. Since the raw materials are stable and widely available, procurement teams can secure inventory easily, avoiding the bottlenecks that plague supply chains dependent on custom-synthesized precursors. The simplicity of the work-up procedure, which often involves standard extraction and column chromatography or recrystallization, means that production throughput is not limited by complex purification bottlenecks. This reliability fosters trust with downstream customers who require guaranteed continuity of supply for their own drug development pipelines.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or ton scale is straightforward due to the absence of hazardous reagents or extreme conditions that pose safety risks in large vessels. The solvents used, such as toluene and dichloromethane, are well-understood in industrial hygiene and can be effectively recycled, aligning with green chemistry principles and reducing environmental compliance costs. The reduction in waste generation, coupled with the high efficiency of the cascade reaction, supports sustainability goals which are increasingly important for corporate social responsibility reporting. This scalability ensures that the technology can grow with the demand, supporting commercial scale-up of complex polymer additives or drug intermediates seamlessly.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope and practicality of this synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industrial implementation. Understanding these nuances helps in assessing the feasibility of adopting this route for specific target molecules within your portfolio.
Q: What are the key advantages of this pyrrole synthesis method over traditional Knorr synthesis?
A: Unlike traditional methods that often require harsh conditions or complex multi-step preparations, this patented cascade reaction utilizes readily available starting materials and operates under mild temperatures (0-130°C), significantly reducing energy consumption and simplifying purification processes.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly highlights the method's suitability for large-scale production due to its environment-friendly nature, high yields (up to 94%), and the use of common solvents like toluene and dichloromethane which are easily managed in standard chemical reactors.
Q: What types of substituents are tolerated in this pyrrole ring construction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (fluoro, chloro, bromo), nitro groups, alkoxy groups, and alkyl chains on the aromatic rings, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic chemistry plays in accelerating drug discovery and material innovation. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory patents can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polysubstituted pyrrole derivatives meets the exacting standards required by global regulatory bodies. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of high-value intermediates.
We invite you to discuss how our manufacturing capabilities can support your specific project requirements. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, along with specific COA data and route feasibility assessments. Whether you are in the early stages of process development or looking to optimize an existing supply chain, we are ready to provide the technical expertise and production capacity needed to bring your projects to market efficiently and cost-effectively.
