Advanced Axial Chiral Oxindole-Derived Styrene Phosphine Catalysts for Industrial Asymmetric Synthesis
The landscape of asymmetric organocatalysis is undergoing a significant transformation with the introduction of novel scaffolds that challenge the dominance of traditional biaryl systems. Patent CN111777637B discloses a groundbreaking class of axial chiral oxindole-derived styrene phosphine catalysts, represented generally by formula 6. These catalysts represent a strategic evolution in chiral ligand design, moving beyond the conventional binaphthyl frameworks that have long saturated the literature. The core innovation lies in the integration of an oxindole moiety with a styrene backbone, creating a rigid, sterically demanding environment that exerts precise control over stereochemical outcomes. This structural novelty addresses a critical gap in the field, where research on non-binaphthyl axial chiral phosphine catalysts has been notably sparse. By leveraging this unique architecture, the technology enables highly efficient catalytic asymmetric [4+2] and [3+2] cyclization reactions, delivering products with excellent yields and substantial enantiomeric excess.
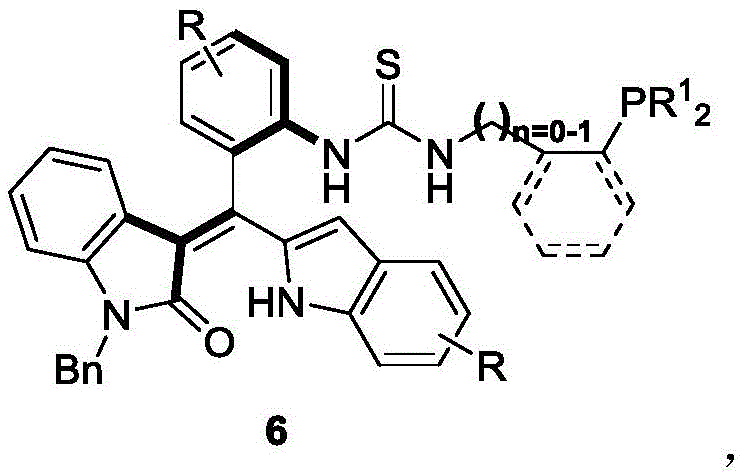
For R&D directors seeking reliable pharmaceutical intermediate supplier partnerships, understanding the mechanistic advantages of such catalysts is paramount. The structural rigidity provided by the oxindole-styrene axis prevents free rotation, locking the catalyst into a specific chiral conformation that effectively differentiates between prochiral faces of substrates. This level of stereocontrol is essential for the synthesis of complex drug candidates where even minor impurities can compromise safety profiles. Furthermore, the modular nature of the synthesis allows for the fine-tuning of electronic and steric properties through variations in the R and R1 groups, providing a versatile platform for optimizing specific transformations. This adaptability makes the technology not just a single solution, but a robust methodology applicable to a wide array of challenging synthetic problems in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the domain of axial chiral phosphine catalysts has been overwhelmingly dominated by binaphthyl-based derivatives, such as BINAP and its analogues. While these catalysts have served the industry well, they possess inherent limitations regarding substrate scope and steric bulk. The reliance on a single structural motif often leads to diminishing returns when attempting to catalyze sterically hindered or electronically distinct substrates. Moreover, the synthesis of modified binaphthyl ligands can be arduous, often requiring resolution steps that halve the theoretical yield or expensive chiral pool starting materials. In many cases, the rigid geometry of traditional biaryl systems fails to provide the necessary secondary interactions, such as hydrogen bonding or pi-stacking, required to stabilize transition states in complex cyclization reactions. This lack of versatility forces process chemists to screen extensive libraries of similar catalysts, consuming valuable time and resources during method development.
The Novel Approach
The approach detailed in CN111777637B circumvents these bottlenecks by introducing a fundamentally different chiral scaffold. The oxindole-derived styrene backbone offers a distinct spatial arrangement that complements, rather than replicates, the steric environment of binaphthyl catalysts. This novelty translates directly into practical benefits: the ability to access chemical space that was previously difficult to navigate with high enantioselectivity. The synthesis of these catalysts is streamlined, utilizing a three-step sequence that avoids the harsh conditions often associated with traditional ligand preparation. By operating at mild temperatures ranging from 0°C to 25°C and employing common solvents like dichloromethane, the process minimizes thermal degradation risks and simplifies purification protocols. This shift towards milder, more efficient synthetic routes represents a significant advancement in cost reduction in pharmaceutical intermediate manufacturing, as it reduces energy consumption and waste generation while maintaining high product quality.
Mechanistic Insights into Asymmetric Cyclization Catalysis
The efficacy of the formula 6 catalysts stems from their ability to activate substrates through nucleophilic phosphine attack while simultaneously directing stereochemistry via the bulky axial chiral backbone. In the context of asymmetric [4+2] cyclizations, the phosphine moiety attacks the allenoate substrate to generate a zwitterionic intermediate. The chiral environment surrounding the phosphorus center, dictated by the oxindole-styrene axis, shields one face of this intermediate, forcing the incoming electrophile (such as an isatin-derived olefin) to approach from the less hindered trajectory. This precise spatial control is critical for achieving high diastereo- and enantioselectivity. The rigidity of the catalyst ensures that this chiral information is transmitted faithfully throughout the catalytic cycle, preventing racemization pathways that often plague flexible ligand systems.
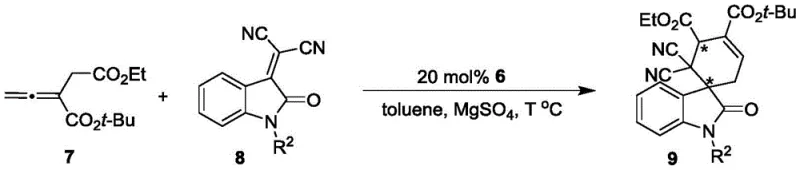
Furthermore, the catalyst design incorporates potential hydrogen-bonding donors within the thiourea linkage (in specific embodiments), which can further stabilize transition states through non-covalent interactions. This dual-activation mode—combining nucleophilic activation with hydrogen bonding—enhances the reaction rate and selectivity. For the asymmetric [3+2] cyclizations involving MBH esters and ninhydrin-derived olefins, the catalyst similarly facilitates the formation of key dipolar intermediates. The steric bulk of the phenyl or cyclohexyl groups on the phosphorus atom (R1) plays a crucial role in discriminating between competing transition states. This mechanistic robustness ensures that the catalyst performs consistently across a range of substrates, providing the reliability needed for commercial scale-up of complex pharmaceutical intermediates. The ability to maintain high enantiomeric excess values, as demonstrated in the patent examples, underscores the precision of this catalytic system.
How to Synthesize Axial Chiral Phosphine Catalyst Efficiently
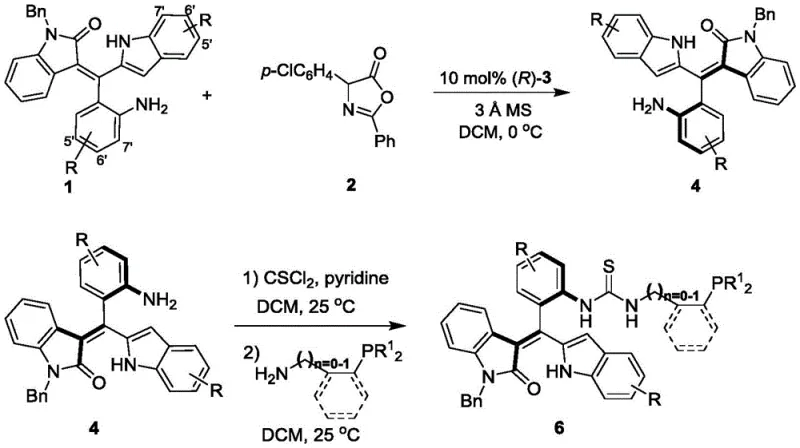
The preparation of these high-value catalysts follows a logical and scalable three-step sequence that prioritizes yield and purity. The process begins with the construction of the axially chiral styrene backbone, followed by functionalization to introduce the phosphine moiety. Each step is optimized to minimize side reactions and simplify workup procedures, ensuring that the final catalyst meets the stringent purity specifications required for sensitive asymmetric transformations. The use of molecular sieves in the initial condensation step effectively drives the equilibrium towards product formation by removing water, a critical detail for maximizing yield in the formation of the chiral scaffold.
- Condense formula 1 and formula 2 compounds in dichloromethane with molecular sieves and chiral phosphoric acid catalyst at 0°C to obtain axially chiral styrene compound 4.
- React compound 4 with thiophosgene and pyridine in dichloromethane at 25°C to form the isothiocyanate intermediate.
- Add the amine component (formula 5) to the intermediate in dichloromethane at 25°C, stir until completion, and purify to yield the final catalyst 6.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this catalyst technology offers compelling advantages rooted in process efficiency and supply chain resilience. The synthetic route relies on readily available starting materials, such as substituted anilines and commercially sourced phosphine amines, which mitigates the risk of raw material shortages. Unlike catalysts dependent on rare earth metals or precious transition metals, this organocatalytic system eliminates the need for expensive metal scavengers and the associated regulatory burden of heavy metal residue testing in final API products. This transition to metal-free catalysis aligns perfectly with modern green chemistry initiatives and simplifies the regulatory filing process for new drug applications.
- Cost Reduction in Manufacturing: The elimination of transition metals significantly lowers the cost of goods sold by removing the need for costly metal catalysts and the specialized equipment required for their removal. Furthermore, the mild reaction conditions (0°C to 25°C) reduce energy consumption compared to processes requiring cryogenic temperatures or high heat. The high atom economy of the cyclization reactions means less waste is generated per kilogram of product, leading to substantial savings in waste disposal and solvent recovery costs. These factors combine to create a leaner, more cost-effective manufacturing process that enhances overall profit margins without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the synthetic pathway ensures consistent supply availability. Because the catalyst synthesis does not depend on geopolitically sensitive rare metals, the supply chain is less vulnerable to market fluctuations and trade disruptions. The scalability of the process, demonstrated by the straightforward purification methods like silica gel column chromatography which can be adapted to preparative HPLC or crystallization on scale, guarantees that production volumes can be ramped up quickly to meet demand. This reliability is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of organic solvents that can be easily recycled. The absence of toxic heavy metals simplifies environmental compliance and reduces the ecological footprint of the manufacturing facility. The high yields and selectivity observed in the patent examples indicate that the process is mature and ready for transfer to pilot and production scales. This ease of scale-up reduces the time-to-market for new products, allowing companies to capitalize on market opportunities faster while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this novel catalyst system. Understanding these details is essential for integrating this technology into existing workflows and maximizing its potential benefits. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation.
Q: What distinguishes this axial chiral catalyst from traditional binaphthyl catalysts?
A: Unlike traditional biaryl axial catalysts limited to binaphthyl skeletons, this invention utilizes a novel oxindole-derived styrene backbone, offering unique steric environments and enhanced enantioselectivity for specific cyclization reactions.
Q: What are the primary applications of catalyst formula 6?
A: The catalyst is specifically designed to facilitate asymmetric [4+2] cyclization reactions between allenoates and isatin-derived olefins, as well as asymmetric [3+2] cyclizations involving MBH esters.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the synthesis operates under mild conditions (0°C to 25°C) using common solvents like dichloromethane and readily available reagents, making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity axial chiral phosphine catalysts that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in chiral synthesis allows us to optimize these routes further, ensuring cost-effectiveness and supply security for our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthesis needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our axial chiral oxindole-derived styrene phosphine catalysts can enhance your manufacturing efficiency and product quality.
