Advanced Pd-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Introduction to Next-Generation Imidazole Synthesis
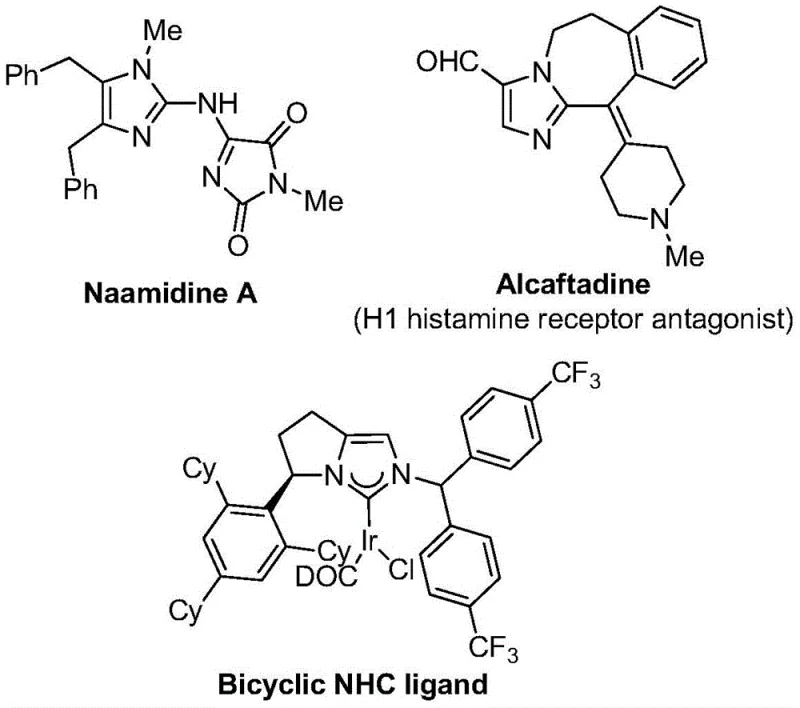
The strategic incorporation of trifluoromethyl groups into nitrogen-containing heterocycles represents a cornerstone of modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As illustrated by bioactive scaffolds such as Alcaftadine and various NHC ligands shown in the reference data, the 2-trifluoromethyl imidazole motif is a high-value structural element sought after by R&D teams globally. Patent CN111423381A discloses a groundbreaking preparation method that addresses the longstanding challenges associated with synthesizing these complex molecules. This technology leverages a transition metal palladium-catalyzed carbonylation cascade, utilizing cheap and readily available starting materials to achieve high reaction efficiency under remarkably mild conditions. For procurement and supply chain leaders, this innovation signals a shift towards more sustainable and cost-effective manufacturing pathways for critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on the use of highly reactive and often hazardous synthons such as trifluorodiazoethane. These traditional approaches frequently necessitate stringent safety protocols due to the explosive nature of diazo compounds, creating significant bottlenecks in large-scale production environments. Furthermore, many existing methods require harsh reaction conditions, including elevated temperatures or the use of high-pressure carbon monoxide gas, which complicates reactor design and increases capital expenditure. The limited substrate compatibility of older methodologies often restricts the diversity of accessible derivatives, forcing chemists to employ lengthy protection-deprotection sequences that erode overall yield. Consequently, the supply chain for these valuable intermediates has been plagued by volatility, high costs, and inconsistent quality, posing risks to the continuity of downstream API manufacturing processes.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a sophisticated palladium-catalyzed system that operates at a mild 30°C, drastically reducing energy consumption and thermal stress on sensitive functional groups. By employing trifluoroethylimidoyl chloride and propargylamine as stable, commercially available building blocks, the process eliminates the need for dangerous diazo reagents entirely. The reaction cleverly utilizes a formic acid and acetic anhydride mixture as a safe carbon monoxide surrogate, thereby removing the logistical and safety hazards associated with handling high-pressure CO gas cylinders. As demonstrated in the general reaction scheme below, this cascade transformation efficiently constructs the imidazole core with excellent atom economy and broad functional group tolerance.
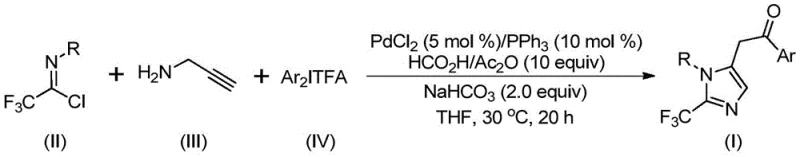
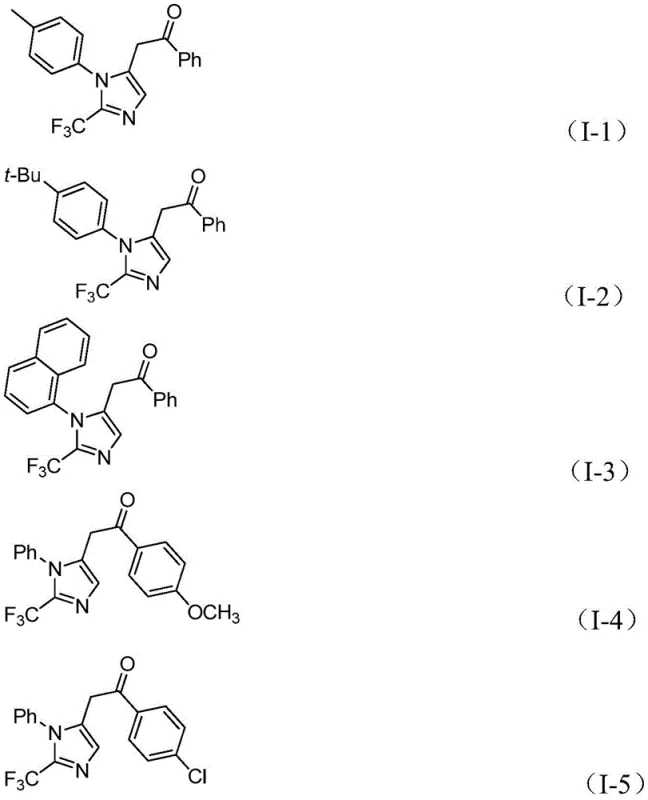
The versatility of this approach is further evidenced by the successful synthesis of diverse derivatives, including those bearing electron-donating and electron-withdrawing groups, as seen in the specific examples provided. This robustness ensures that the method can be adapted for the rapid generation of compound libraries for drug discovery as well as the reliable production of key intermediates for commercial APIs.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The elegance of this synthesis lies in its intricate catalytic cycle, which orchestrates multiple bond-forming events in a single pot. The reaction is initiated by a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. Following isomerization, the palladium catalyst facilitates an aminopalladation of the alkyne moiety, yielding an alkenyl palladium species that subsequently isomerizes to a more stable alkyl palladium intermediate. This critical organometallic species then undergoes carbonylation, driven by the carbon monoxide released in situ from the formic acid and acetic anhydride mixture, to form an acyl palladium complex. The cycle culminates in an oxidative addition with the diaryliodonium salt to generate a high-valent tetravalent palladium intermediate, which finally undergoes reductive elimination to release the desired 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From a quality control perspective, this mechanism offers distinct advantages regarding impurity profiles. The use of diaryliodonium salts as oxidants and aryl sources is particularly beneficial, as the byproducts are typically benign iodobenzene derivatives that are easily separated during workup. The mild temperature of 30°C suppresses side reactions such as polymerization or decomposition of the trifluoromethyl group, which are common pitfalls in high-temperature heterocycle synthesis. Furthermore, the specific stoichiometry of the catalyst system, utilizing palladium chloride and triphenylphosphine, ensures high turnover numbers while minimizing residual metal contamination in the final product. This level of mechanistic control is essential for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical ingredients.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized reaction parameters to ensure maximum yield and reproducibility. The process involves combining the palladium catalyst, ligand, base, and CO surrogate in an aprotic solvent such as tetrahydrofuran before introducing the substrates. Detailed standard operating procedures regarding mixing rates, addition sequences, and purification protocols are critical for scaling this chemistry from gram to kilogram quantities. For a comprehensive, step-by-step guide tailored to your specific production needs, please refer to the standardized synthesis instructions provided below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The shift from hazardous, specialized reagents to commodity chemicals fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the need for custom-synthesized diazo compounds and high-pressure gas infrastructure, manufacturers can achieve significant cost reductions in API manufacturing while simultaneously enhancing operational safety. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the incidence of costly failed batches and rework. Furthermore, the scalability of the process from small-scale discovery to multi-ton commercial production provides a seamless pathway for drug development, mitigating the risks associated with technology transfer.
- Cost Reduction in Manufacturing: The utilization of inexpensive and widely available starting materials, such as aromatic amines and propargylamine, drastically lowers the raw material cost basis compared to traditional methods relying on exotic fluorinated synthons. The elimination of high-pressure equipment requirements and the use of mild heating conditions result in substantially lower energy consumption and capital depreciation costs. Additionally, the simplified workup procedure, which avoids complex quenching steps associated with reactive diazo species, reduces labor hours and waste disposal expenses. These cumulative efficiencies allow for a more competitive pricing structure for the final pharmaceutical intermediate without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key reagents, including trifluoroethylimidoyl chloride and diaryliodonium salts, are derived from abundant feedstocks with established global supply networks. The reduced dependency on niche suppliers of hazardous materials minimizes the risk of supply disruptions caused by regulatory crackdowns or transportation restrictions on dangerous goods. The high functional group tolerance of the reaction means that a single robust platform can produce a wide variety of derivatives, allowing for greater flexibility in responding to changing market demands. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates, keeping your development pipelines moving forward without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, having been validated from milligram to gram scales with the potential for multi-kilogram production, aligning with the needs of commercial scale-up of complex heterocyclic intermediates. The use of a liquid CO surrogate instead of gaseous carbon monoxide simplifies reactor engineering and enhances safety compliance, making it easier to obtain necessary environmental and safety permits. The generation of minimal hazardous waste and the use of recyclable solvents like THF contribute to a greener manufacturing footprint, supporting corporate sustainability goals. This alignment with green chemistry principles future-proofs the supply chain against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation to assist decision-makers in evaluating feasibility. Understanding these details is crucial for assessing the fit of this technology within your existing manufacturing infrastructure and quality systems.
Q: What are the key advantages of this Pd-catalyzed method over traditional diazo-based routes?
A: This method avoids the use of hazardous trifluorodiazoethane, utilizing safer trifluoroethylimidoyl chloride instead. It operates at mild temperatures (30°C) without high-pressure CO gas, significantly improving operational safety and substrate compatibility.
Q: What is the role of the formic acid and acetic anhydride mixture in this reaction?
A: The mixture serves as a safe and effective carbon monoxide (CO) surrogate. It releases CO in situ under the reaction conditions, facilitating the carbonylation step without requiring dangerous high-pressure gas cylinders.
Q: Can this synthesis method accommodate diverse functional groups on the aryl rings?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substituents such as methyl, tert-butyl, methoxy, chloro, bromo, trifluoromethyl, and nitro groups on both the imidoyl chloride and the iodonium salt components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced intermediates like 2-trifluoromethyl imidazoles for your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in pre-clinical trials or full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical applications. Our commitment to technical excellence allows us to navigate the complexities of fluorinated chemistry with precision and reliability.
We invite you to collaborate with us to leverage this innovative Pd-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-value chemical solutions.
