Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrroles for Commercial Pharma Applications
Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrroles for Commercial Pharma Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for more sustainable and efficient pathways to access bioactive scaffolds. A significant breakthrough in this domain is documented in patent CN113636968B, which outlines a novel synthetic method for 3-acylpyrrole compounds. This technology represents a paradigm shift from traditional multi-step sequences to a streamlined, one-pot cascade reaction. By leveraging a ruthenium-catalyzed transfer hydrogenation strategy, this process converts readily available alpha-amino alcohols and alpha,beta-unsaturated alkynones directly into highly substituted pyrrole derivatives. For R&D directors and process chemists, this innovation offers a compelling solution to the longstanding challenges of constructing the pyrrole core, particularly the difficult installation of the acyl group at the 3-position without resorting to hazardous reagents or complex protection-deprotection strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-acylpyrrole skeletons has been fraught with synthetic inefficiencies that hinder large-scale manufacturing. Conventional routes often rely on the use of stoichiometric oxidants, such as IBX (2-iodoxybenzoic acid), to mediate intramolecular oxidative cyclization of N-hydroxyalkylenamines. While effective on a milligram scale, these oxidative protocols generate substantial amounts of iodine-containing waste, creating severe environmental burdens and complicating downstream purification. Furthermore, alternative palladium-catalyzed methods frequently require expensive phosphine ligands and strictly anhydrous conditions, increasing the operational complexity and cost of goods. Many existing processes also suffer from poor atom economy, where significant portions of the starting material mass end up as byproducts rather than incorporated into the final API intermediate, thereby inflating raw material costs and reducing overall process sustainability.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113636968B introduces a highly atom-economical borrowing hydrogen methodology. This approach utilizes ethanol not merely as a solvent but as a sacrificial hydrogen donor, facilitating a redox-neutral cyclization that eliminates the need for external oxidants or high-pressure hydrogen gas. The reaction proceeds through a tandem sequence where the alpha-amino alcohol is transiently oxidized to an amino aldehyde, which then condenses with the ynone substrate before undergoing cyclization and rearomatization. This elegant design simplifies the workflow into a single operational step, drastically reducing labor hours and equipment occupancy time. The use of a robust ruthenium carbonyl cluster catalyst ensures high turnover numbers even under the relatively forcing thermal conditions required for the cyclization step.
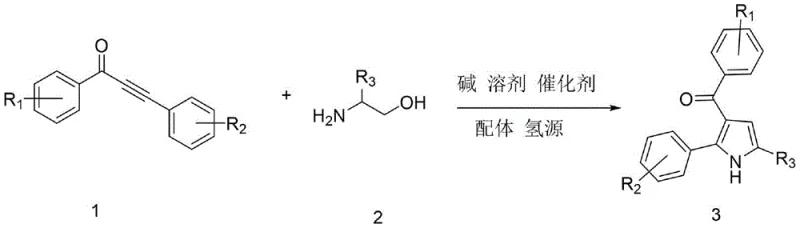
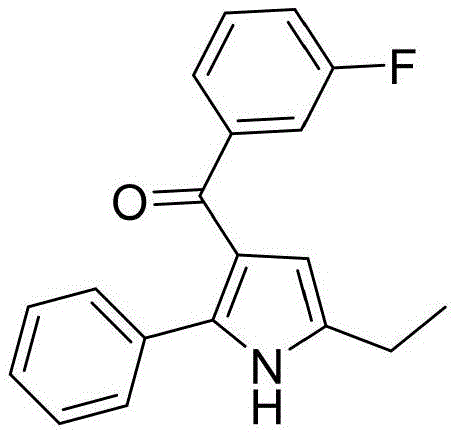
The versatility of this new synthetic route is further evidenced by its broad substrate scope, accommodating a wide array of electronic and steric environments on the aromatic rings. As illustrated in the specific examples provided within the patent data, the system tolerates electron-withdrawing groups such as trifluoromethyl and fluoro substituents, which are critical motifs in modern medicinal chemistry for modulating metabolic stability and lipophilicity. Simultaneously, electron-donating groups like methyl and methoxy are compatible, allowing for the rapid generation of diverse chemical libraries. This flexibility is paramount for process development teams aiming to optimize the physicochemical properties of lead compounds without being constrained by synthetic limitations. The ability to access these complex heterocycles from simple, commercially available building blocks positions this technology as a cornerstone for next-generation pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ru-Catalyzed Transfer Hydrogenation Cyclization
To fully appreciate the technical sophistication of this process, one must delve into the mechanistic underpinnings of the ruthenium-catalyzed cycle. The reaction initiates with the activation of the alpha-amino alcohol by the dodecacarbonyl triruthenium catalyst, likely facilitated by the 4-methyl-1,10-phenanthroline ligand which stabilizes the metal center against aggregation. This initial step involves the dehydrogenation of the alcohol moiety to generate a reactive amino aldehyde intermediate in situ, releasing molecular hydrogen which is temporarily stored on the metal complex. This "borrowed hydrogen" is then utilized in the final aromatization step, effectively shuttling hydrogen atoms from the alcohol substrate to the dihydropyrrole intermediate. This internal redox balance is the key to the reaction's high atom economy, as it avoids the accumulation of reduced or oxidized waste streams that typically plague stepwise syntheses.
Following the initial condensation between the in situ generated amino aldehyde and the alpha,beta-unsaturated alkynone, the system undergoes a Michael-type addition followed by cyclization. The presence of the base, specifically potassium carbonate in the optimized protocol, plays a dual role: it facilitates the initial deprotonation steps and assists in the final elimination of water to restore aromaticity. The specific choice of tert-amyl alcohol as the co-solvent is also mechanistically significant; its high boiling point allows the reaction to reach the necessary activation energy of 150°C without requiring pressurized vessels, while its steric bulk prevents it from acting as a competing nucleophile. This precise orchestration of catalyst, ligand, and reaction conditions ensures that the formation of the 3-acylpyrrole core proceeds with high regioselectivity, minimizing the formation of isomeric byproducts that would otherwise complicate purification and lower overall yield.
How to Synthesize 3-Acylpyrrole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful charging of the reactor under an inert atmosphere to prevent catalyst oxidation prior to reaction initiation. The sequential addition of reagents and the controlled temperature ramping are critical control points that dictate the success of the transformation. Operators must ensure that the nitrogen protection is maintained throughout the initial low-temperature phase to allow for proper catalyst activation before subjecting the mixture to the high-temperature cyclization conditions. Detailed standard operating procedures regarding the specific molar ratios and solvent volumes are essential for scaling this chemistry from gram to kilogram quantities.
- Charge reactor with alpha-amino alcohol, alpha,beta-unsaturated ynone, Ru3(CO)12 catalyst, ligand, ethanol, base, and solvent.
- Stir mixture at 80°C under nitrogen protection for 1.5 hours to initiate catalytic activation.
- Raise temperature to 150°C and maintain for 18 hours to complete cyclization, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ruthenium-catalyzed methodology offers profound advantages for supply chain resilience and cost management. By eliminating the need for stoichiometric oxidants like IBX or expensive palladium catalysts, the raw material cost profile is significantly improved. The reliance on ethanol as a hydrogen source replaces hazardous reagents with a commodity chemical that is inexpensive, readily available globally, and easy to handle. This substitution not only lowers the direct cost of goods sold but also reduces the regulatory burden associated with the storage and disposal of toxic oxidizing agents. Furthermore, the simplified one-pot nature of the reaction reduces the number of unit operations, leading to lower utility consumption and decreased labor costs per kilogram of produced intermediate.
- Cost Reduction in Manufacturing: The transition from multi-step oxidative protocols to a single-pot transfer hydrogenation process inherently drives down manufacturing costs by consolidating reaction steps. Eliminating intermediate isolation and purification stages reduces solvent usage and waste treatment expenses substantially. Additionally, the use of a base metal catalyst system like ruthenium, while precious, is employed at low loadings and offers high efficiency, providing a favorable balance between catalyst cost and productivity compared to palladium alternatives. The overall process intensification leads to a leaner manufacturing footprint, allowing facilities to produce higher volumes of high-purity pharmaceutical intermediates within existing infrastructure constraints.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain security by utilizing widely available starting materials such as alpha-amino alcohols and substituted chalcones. Unlike specialized reagents that may have single-source suppliers or long lead times, the key inputs for this process are commodity chemicals with established global supply chains. This diversification of sourcing options mitigates the risk of production stoppages due to raw material shortages. Moreover, the operational simplicity of the reaction, which does not require specialized high-pressure hydrogenation equipment, allows for greater flexibility in choosing contract manufacturing organizations, thereby expanding the potential supplier base and ensuring continuity of supply for critical drug substances.
- Scalability and Environmental Compliance: Scaling this process to commercial levels is facilitated by the use of standard heating protocols and ambient pressure conditions, avoiding the capital expenditure associated with high-pressure autoclaves. The environmental profile is markedly superior to conventional methods, as the primary byproduct is water, and the solvent system can be designed for efficient recovery and recycling. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory filings related to impurity profiles and residual solvents. The ability to run the reaction at 150°C in a high-boiling solvent ensures that the kinetics are favorable for large batch sizes, enabling the efficient production of multi-ton quantities required for late-stage clinical and commercial API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their current development pipelines. The answers reflect the consensus of the technical data regarding yield, purity, and operational parameters observed across multiple examples.
Q: What are the key advantages of this Ru-catalyzed method over traditional oxidative cyclization?
A: Unlike traditional methods requiring stoichiometric oxidants like IBX or harsh acidic conditions, this protocol utilizes ethanol as a benign hydrogen source via transfer hydrogenation, significantly reducing toxic waste and improving atom economy.
Q: Is the catalyst system scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of dodecacarbonyl triruthenium with a stable phenanthroline ligand allows for robust performance at elevated temperatures (150°C), making it suitable for commercial scale-up in standard pressure reactors.
Q: What is the functional group tolerance for the R1 and R2 substituents?
A: The method demonstrates excellent compatibility with electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methyl and methoxy, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113636968B for accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-acylpyrrole intermediate meets the exacting standards required by global regulatory agencies. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry, delivering high-quality materials that support your critical timelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, exploring how this ruthenium-catalyzed route can optimize your budget. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed decisions about your supply chain strategy. Let us be your partner in transforming complex chemical challenges into commercial successes.
