Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Manufacturing with Safe Metal-Free Catalysis
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Manufacturing with Safe Metal-Free Catalysis
In the rapidly evolving landscape of pharmaceutical and agrochemical intermediate manufacturing, the demand for efficient, safe, and scalable synthetic routes is paramount. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, offering a robust platform for the production of high-value heterocyclic scaffolds. For R&D directors and procurement strategists, understanding the nuances of this base-promoted, metal-free cyclization is critical for optimizing supply chains and reducing the total cost of ownership for complex fluorinated intermediates. The patent highlights a process that not only eliminates the need for explosive azides but also removes the burden of heavy metal contamination, thereby streamlining downstream purification and ensuring higher product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloadditions or organocatalytic reactions involving toxic azides. As illustrated in the prior art comparison, traditional pathway (a) typically involves the reaction of organic azides with alkynes in the presence of copper catalysts, followed by trifluoromethylation. This approach presents severe safety hazards due to the inherent instability and explosivity of organic azides, posing significant risks during scale-up. Furthermore, the use of transition metal catalysts introduces the challenge of residual metal removal, which is a costly and time-consuming step required to meet stringent pharmaceutical purity standards. Alternative methods involving 1,3-dipolar cycloadditions often suffer from harsh reaction conditions, limited substrate scope, or the requirement for expensive and specialized trifluoromethylating reagents that drive up raw material costs.
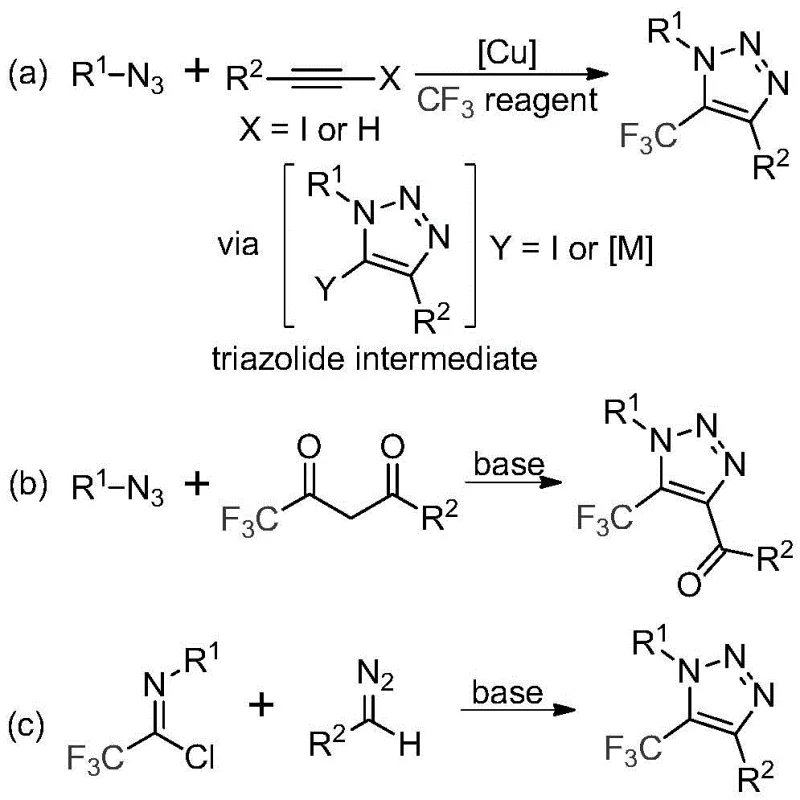
The Novel Approach
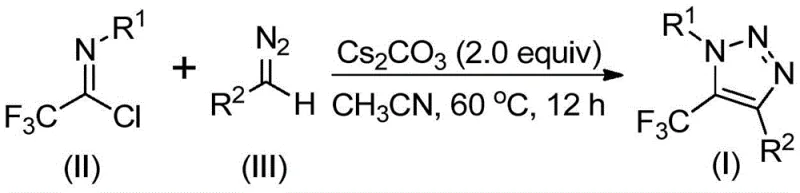
In stark contrast to these legacy techniques, the methodology described in CN113121462B utilizes readily available trifluoroethylimidoyl chloride and diazo compounds as starting materials. This innovative route operates under mild, base-promoted conditions without the necessity for transition metals or dangerous azide reagents. The reaction proceeds efficiently in common aprotic solvents such as acetonitrile at moderate temperatures ranging from 50°C to 70°C. By bypassing the use of copper catalysts, this method inherently produces a cleaner crude reaction profile, significantly simplifying the workup procedure. The elimination of explosive precursors enhances operational safety, making this process particularly attractive for large-scale commercial manufacturing where risk mitigation is a top priority for supply chain heads. The versatility of this approach allows for the introduction of diverse functional groups at the 4-position of the triazole ring, enabling the rapid generation of chemical libraries for drug discovery.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation offers fascinating insights into its efficiency and selectivity. The reaction is believed to initiate with a base-promoted intermolecular nucleophilic addition-elimination process where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This key step facilitates the formation of a critical carbon-carbon bond, setting the stage for ring closure. Following this initial addition, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is energetically favorable under the described conditions and leads directly to the formation of the 1,2,3-triazole core. The use of cesium carbonate as the base is particularly effective, likely due to its solubility profile and basicity which optimally activates the diazo species without causing excessive decomposition. This mechanistic understanding assures R&D teams that the process is robust and less prone to the formation of complex byproducts often seen in radical-based trifluoromethylation strategies.
From an impurity control perspective, the absence of transition metals means there is no risk of metal-catalyzed side reactions such as homocoupling of alkynes or azides, which are common pitfalls in copper-catalyzed variants. The primary impurities are likely to be unreacted starting materials or hydrolysis products of the imidoyl chloride, both of which are generally easier to separate from the final triazole product via standard silica gel chromatography or crystallization. The high functional group tolerance observed in the patent examples, including esters, ketones, and phosphonates, suggests that the reaction conditions are sufficiently mild to preserve sensitive moieties. This level of chemoselectivity is vital for synthesizing complex pharmaceutical intermediates where protecting group strategies can add significant cost and time to the overall synthesis.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it highly accessible for laboratory and pilot plant settings. The protocol involves combining the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in an organic solvent, typically acetonitrile. The mixture is then heated to facilitate the cyclization. Detailed procedural specifics regarding stoichiometry, solvent volumes, and purification techniques are critical for reproducibility and yield optimization. For a comprehensive guide on executing this transformation with maximum efficiency, please refer to the standardized synthesis steps outlined below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the final triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The shift towards this metal-free methodology addresses several critical pain points in the sourcing of fluorinated heterocycles. By removing the dependency on copper catalysts and explosive azides, manufacturers can significantly reduce the regulatory burden and insurance costs associated with handling hazardous materials. The use of commodity chemicals like cesium carbonate and acetonitrile ensures a stable and predictable supply of reagents, mitigating the risk of raw material shortages that often plague specialty reagent-dependent processes. Furthermore, the simplified purification workflow translates directly into reduced processing time and lower utility consumption, contributing to a more sustainable and cost-effective manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive scavenging resins or complex extraction protocols required to meet heavy metal limits in APIs. This simplification of the downstream processing significantly lowers the operational expenditure per kilogram of product. Additionally, the starting materials, specifically the diazo compounds and imidoyl chlorides, are derived from abundant precursors, ensuring that raw material costs remain competitive even at large scales. The high atom economy of the cyclization step further contributes to waste reduction, aligning with green chemistry principles that are increasingly valued by global pharmaceutical partners.
- Enhanced Supply Chain Reliability: Relying on stable, non-explosive reagents drastically improves the logistical flexibility of the supply chain. Unlike organic azides which require special shipping classifications and storage conditions, the reagents used in this patent can be transported and stored using standard industrial protocols. This reduces lead times and simplifies inventory management. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by sensitivity to moisture or oxygen, ensuring consistent on-time delivery of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: The patent data indicates that the reaction performs well at gram scales with yields reaching up to 91% for certain substrates, suggesting excellent potential for ton-scale production. The mild temperature range of 50°C to 70°C is energy-efficient and easily manageable in standard glass-lined or stainless steel reactors. From an environmental standpoint, the avoidance of toxic heavy metals and explosive waste streams simplifies effluent treatment and disposal, helping manufacturing sites maintain compliance with increasingly strict environmental regulations without incurring excessive waste management fees.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this method in an industrial setting.
Q: Why is this metal-free method safer than traditional copper-catalyzed routes?
A: Traditional methods often require explosive organic azides and toxic copper catalysts. This novel route utilizes stable diazo compounds and avoids heavy metals entirely, significantly reducing safety risks and purification costs.
Q: What are the scalability prospects for this trifluoromethyl-triazole synthesis?
A: The patent explicitly demonstrates that the method can be easily extended to gram-level scales using mild conditions (60°C) and common solvents like acetonitrile, indicating strong potential for industrial kilogram-to-ton production.
Q: How does the substrate scope impact commercial availability?
A: The reaction tolerates a wide range of functional groups including esters, ketones, and phosphonates on the diazo component, allowing for the flexible production of diverse intermediates for drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN113121462B for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the exacting standards required for drug substance development. We are committed to leveraging this advanced metal-free technology to provide our clients with superior quality intermediates at competitive market rates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient route. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to request detailed route feasibility assessments tailored to your unique molecular targets. Let us collaborate to accelerate your development timelines while optimizing your manufacturing costs.
