Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
Introduction to Patent CN113105402B
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in blockbuster drugs such as Sitagliptin, Maraviroc, and Deferasirox. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses significant limitations in current manufacturing technologies. This innovation introduces a metal-free, iodine-promoted cyclization strategy that enables the simultaneous incorporation of trifluoromethyl and acyl groups into the triazole core. For R&D directors and process chemists, this represents a paradigm shift away from complex transition-metal catalysis toward simpler, more sustainable organic transformations. The technology leverages a tandem iodination and Kornblum oxidation sequence, allowing for the direct conversion of inexpensive aryl ethyl ketones into high-value heterocyclic intermediates. By eliminating the requirement for stringent anhydrous or oxygen-free conditions, this method significantly lowers the barrier to entry for commercial scale-up, offering a reliable pathway for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing polysubstituted 1,2,4-triazoles often rely heavily on transition metal catalysts, such as copper or palladium complexes, which introduce substantial cost and purity challenges. These conventional methods typically necessitate rigorous exclusion of moisture and oxygen, requiring specialized equipment like Schlenk lines or gloveboxes that increase capital expenditure and operational complexity. Furthermore, the removal of trace heavy metal residues from the final active pharmaceutical ingredient (API) is a critical regulatory hurdle, often demanding additional purification steps like scavenging or recrystallization that reduce overall yield. Many existing protocols also struggle to efficiently introduce both electron-withdrawing trifluoromethyl groups and acyl functionalities simultaneously, limiting the structural diversity accessible to medicinal chemists. The reliance on exotic reagents and harsh reaction conditions frequently results in poor atom economy and generates significant hazardous waste, conflicting with modern green chemistry principles and increasing the environmental burden of chemical manufacturing processes.
The Novel Approach
The methodology described in patent CN113105402B overcomes these barriers by utilizing elemental iodine and dimethyl sulfoxide (DMSO) as the primary promoters for the cyclization reaction. This novel approach operates under atmospheric conditions, removing the need for expensive inert gas protection and simplifying the reactor setup significantly. The process initiates with the iodination and subsequent Kornblum oxidation of aryl ethyl ketones to generate reactive alpha-diketone intermediates in situ, which then condense with trifluoroethylimide hydrazides to form the target triazole ring. 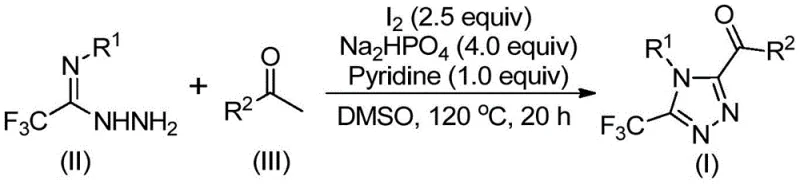 This cascade transformation is highly efficient, tolerating a wide range of functional groups on both the ketone and hydrazide substrates, including halogens, alkoxy groups, and alkyl chains. By avoiding heavy metal catalysts entirely, the resulting product stream is inherently cleaner, drastically reducing the burden on downstream purification units and ensuring compliance with strict ICH guidelines for residual metals in drug substances.
This cascade transformation is highly efficient, tolerating a wide range of functional groups on both the ketone and hydrazide substrates, including halogens, alkoxy groups, and alkyl chains. By avoiding heavy metal catalysts entirely, the resulting product stream is inherently cleaner, drastically reducing the burden on downstream purification units and ensuring compliance with strict ICH guidelines for residual metals in drug substances.
Mechanistic Insights into Iodine-Promoted Cyclization
The reaction mechanism involves a sophisticated interplay between iodine-mediated oxidation and nucleophilic cyclization. Initially, the aryl ethyl ketone undergoes alpha-iodination followed by oxidation by DMSO to form an alpha-dicarbonyl species. This electrophilic intermediate is then trapped by the nucleophilic nitrogen of the trifluoroethylimide hydrazide, leading to the formation of a hydrazone intermediate. Under the continued influence of iodine and the basic environment provided by pyridine and sodium dihydrogen phosphate, an intramolecular cyclization occurs to close the 1,2,4-triazole ring. The presence of the trifluoromethyl group is crucial, as it enhances the electrophilicity of the imine carbon and stabilizes the final aromatic system through strong electron-withdrawing effects. This mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric byproducts that are common in less controlled cyclocondensation reactions. The use of sodium dihydrogen phosphate acts as a mild buffer, maintaining the optimal pH for cyclization without promoting hydrolysis of sensitive functional groups, thereby preserving the integrity of complex substrate molecules throughout the synthesis.
Impurity control is inherently built into this design due to the mild nature of the reagents and the thermodynamic stability of the final triazole product. Unlike radical-based metal catalysis which can generate diverse side products through uncontrolled radical propagation, this ionic mechanism proceeds through well-defined intermediates. The patent data demonstrates excellent functional group tolerance, successfully synthesizing derivatives with chloro, methoxy, and fluoro substituents without significant degradation. 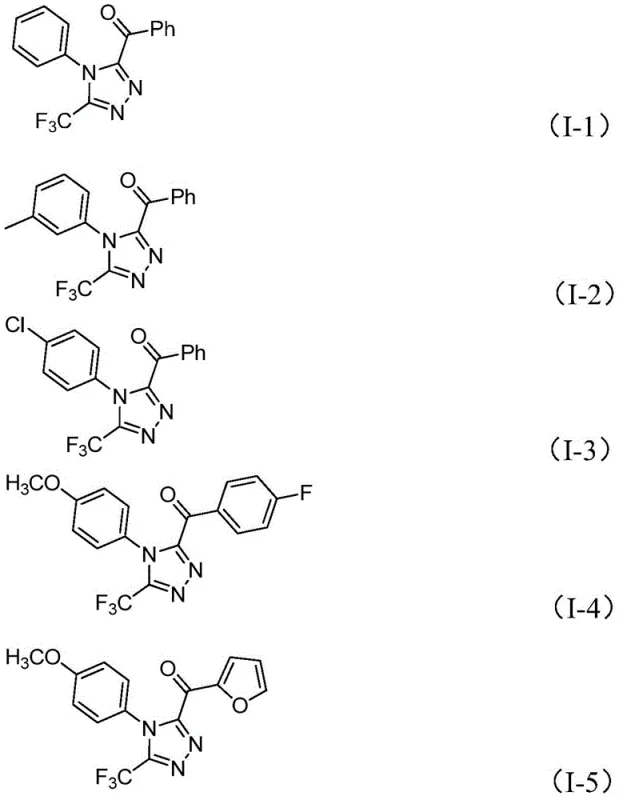 For instance, substrates containing electron-rich methoxy groups or electron-deficient chloro groups both proceed to high conversion, indicating that the electronic nature of the starting material has a minimal negative impact on the reaction efficiency. This robustness is essential for process chemistry, where batch-to-batch consistency and impurity profiles are critical quality attributes. The ability to tune the R1 and R2 groups allows for the rapid generation of analog libraries for structure-activity relationship (SAR) studies, accelerating the drug discovery timeline for potential new therapeutic agents targeting various biological pathways.
For instance, substrates containing electron-rich methoxy groups or electron-deficient chloro groups both proceed to high conversion, indicating that the electronic nature of the starting material has a minimal negative impact on the reaction efficiency. This robustness is essential for process chemistry, where batch-to-batch consistency and impurity profiles are critical quality attributes. The ability to tune the R1 and R2 groups allows for the rapid generation of analog libraries for structure-activity relationship (SAR) studies, accelerating the drug discovery timeline for potential new therapeutic agents targeting various biological pathways.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and pilot plant operations. The procedure involves a two-stage heating process in a single pot, starting with the activation of the ketone followed by the addition of the hydrazide component. This telescoped approach minimizes solvent usage and handling time, contributing to a lower overall cost of goods sold (COGS). The detailed standardized synthesis steps, including precise molar ratios and temperature ramps, are outlined below to ensure reproducibility and safety during implementation.
- Combine aryl ethyl ketone and elemental iodine in DMSO solvent and heat to 90-110°C for 4-6 hours to initiate iodination and oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic advantages by decoupling production from the volatile supply chains of precious metals. The primary reagents, aryl ethyl ketones and elemental iodine, are commodity chemicals available in bulk quantities from multiple global suppliers, ensuring supply continuity even during market disruptions. The elimination of expensive ligands and transition metal catalysts results in significant cost reduction in pharmaceutical intermediate manufacturing, as the raw material bill of materials is drastically simplified. Furthermore, the absence of heavy metals removes the need for specialized metal scavenger resins or complex extraction protocols, which are often cost-prohibitive at large scales. This streamlined workflow translates directly into improved margin potential for contract development and manufacturing organizations (CDMOs) and generic drug manufacturers seeking to optimize their production economics.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with inexpensive elemental iodine creates a fundamental shift in the cost structure of triazole synthesis. By removing the dependency on palladium or copper, companies can avoid the high price volatility associated with these metals and eliminate the capital costs of metal recovery systems. Additionally, the use of DMSO as a solvent, which is cheap and effective for this transformation, further lowers operating expenses compared to specialized dry solvents required for organometallic chemistry. The high atom economy of the reaction ensures that a larger proportion of the input mass is converted into valuable product, reducing waste disposal costs and maximizing yield per batch.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified because the key starting materials are widely produced fine chemicals rather than specialized reagents with long lead times. Aryl ethyl ketones and trifluoroethylimide hydrazides can be stockpiled easily, allowing manufacturers to maintain robust inventory levels and respond quickly to surges in demand. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert atmosphere maintenance. This reliability is critical for securing long-term supply agreements with major pharmaceutical clients who prioritize consistent delivery schedules and risk mitigation in their vendor selection criteria.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, having been demonstrated effectively from gram to multi-gram levels without loss of efficiency. The lack of pyrophoric reagents or high-pressure hydrogenation steps reduces the safety risks associated with scaling exothermic reactions in large reactors. From an environmental standpoint, the avoidance of toxic heavy metals aligns with increasingly stringent global regulations regarding wastewater discharge and solid waste management. This green chemistry profile facilitates faster regulatory approvals and enhances the corporate sustainability metrics of manufacturers adopting this technology, making it a preferred choice for eco-conscious supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted synthesis method. These answers are derived directly from the experimental data and specifications provided in the patent documentation to assist technical teams in evaluating feasibility.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metal promoter, completely eliminating the need for costly palladium or copper catalysts and simplifying downstream purification.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) at temperatures between 110°C and 130°C for 12 to 20 hours, under atmospheric conditions without the need for strict anhydrous or oxygen-free environments.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily scalable from gram-level laboratory synthesis to industrial production due to the use of cheap, readily available raw materials and simple operational procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this DMSO-based process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency is key in API manufacturing, and our dedicated process chemistry team is ready to optimize this route specifically for your target molecule.
We invite you to collaborate with us to leverage this cost-effective technology for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative method can enhance your supply chain resilience and profitability. Let us help you secure a competitive advantage in the global market with our reliable supply of high-quality triazole intermediates.
