Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, addressing significant bottlenecks in current manufacturing processes. These nitrogen-containing five-membered heterocycles are ubiquitous in medicinal chemistry, forming the core structure of renowned drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity of bioactive molecules below. The introduction of a trifluoromethyl group into these scaffolds is particularly valuable, as it enhances metabolic stability, lipophilicity, and bioavailability, making this synthetic methodology highly relevant for modern drug discovery and development pipelines.
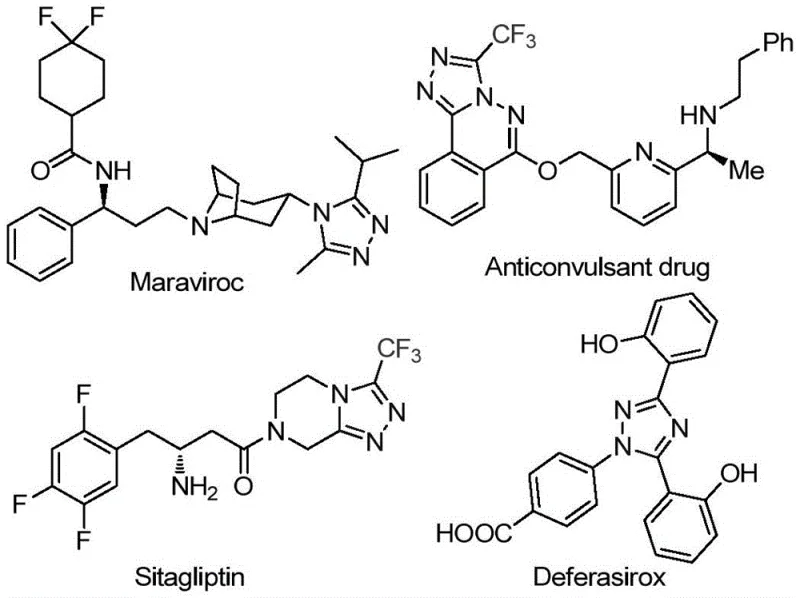
This novel approach leverages a non-metal promoted strategy that circumvents the complexities associated with traditional transition metal catalysis. By utilizing inexpensive and commercially available starting materials, specifically aryl ethyl ketones and trifluoroethylimide hydrazides, the process achieves high efficiency without demanding stringent anhydrous or oxygen-free conditions. For R&D directors and process chemists, this represents a paradigm shift towards greener, more accessible chemistry that maintains high purity standards while drastically simplifying the operational workflow. The ability to generate complex trifluoromethylated triazoles through a tandem cyclization sequence offers a versatile platform for generating diverse libraries of potential drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those bearing both trifluoromethyl and acyl groups, has been fraught with challenges that hinder industrial scalability. Conventional methodologies often rely heavily on transition metal catalysts, which introduce significant complications regarding residual metal removal, a critical quality attribute for pharmaceutical intermediates. These metal-catalyzed routes frequently necessitate rigorous anhydrous and inert atmosphere conditions, requiring specialized equipment and increasing the overall cost of goods sold (COGS). Furthermore, many existing protocols suffer from limited substrate scope, failing to tolerate a wide range of functional groups, which restricts the chemical space available for medicinal chemists to explore. The reliance on expensive reagents and complex purification steps to remove metal traces creates a bottleneck in the supply chain, leading to longer lead times and higher procurement costs for high-purity intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN113105402B employs a metal-free, iodine-promoted system that streamlines the synthesis of 3,4,5-trisubstituted 1,2,4-triazoles. This innovative route utilizes a tandem reaction sequence involving iodination and Kornblum oxidation followed by cyclization, all mediated by elemental iodine in dimethyl sulfoxide (DMSO). The versatility of this approach is evidenced by its compatibility with various substituted aryl groups, including those with methyl, methoxy, chloro, and trifluoromethyl substituents, as demonstrated by the successful synthesis of compounds I-1 through I-5 shown below. This broad functional group tolerance allows for the rapid generation of diverse analogues, accelerating the hit-to-lead optimization process. By eliminating the need for heavy metals and harsh conditions, this method not only reduces environmental impact but also simplifies the downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
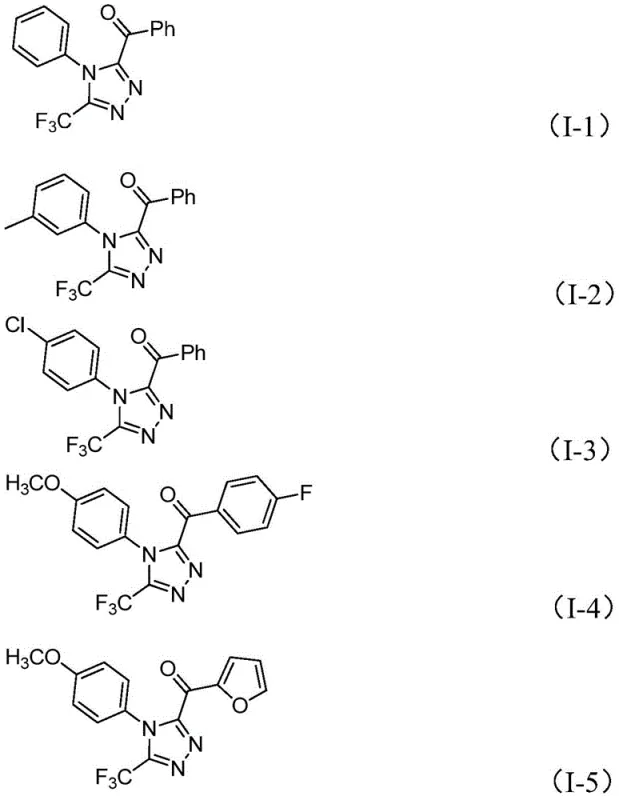
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions driven by elemental iodine and the solvent system. The reaction initiates with the iodination of the aryl ethyl ketone in DMSO, which acts as both the solvent and an oxidant in the Kornblum oxidation process to generate an aryl diketone intermediate in situ. This reactive species then undergoes a dehydration condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine serves to buffer the reaction medium and facilitate the subsequent intramolecular cyclization. Under the thermal conditions of 110-130°C, the hydrazone undergoes ring closure promoted by iodine to yield the final 3,4,5-trisubstituted 1,2,4-triazole scaffold. This cascade reaction design minimizes the number of isolation steps, thereby improving overall atom economy and process mass intensity (PMI).
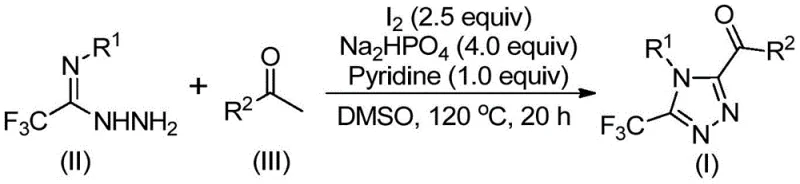
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal-catalyzed side reactions that often generate difficult-to-remove organometallic impurities. The use of DMSO as a polar aprotic solvent ensures high solubility of the polar intermediates, promoting homogeneous reaction kinetics and reducing the formation of oligomeric byproducts. Furthermore, the specific stoichiometry of reagents, particularly the excess of aryl ethyl ketone relative to the hydrazide, drives the equilibrium towards the desired product, suppressing the formation of unreacted starting materials. The post-treatment process involving simple filtration and silica gel column chromatography is sufficient to achieve high purity specifications, ensuring that the final API intermediate meets the rigorous quality standards required for clinical and commercial applications.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory-scale discovery and pilot-plant production. The protocol involves a two-stage heating process where the initial oxidation is conducted at 90-110°C, followed by the addition of the remaining reagents and a second heating stage at 110-130°C. This stepwise addition allows for better control over the exothermic nature of the iodination and prevents the degradation of sensitive intermediates. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the structured guide below to ensure reproducibility and safety during scale-up operations.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The primary driver for cost optimization lies in the elimination of expensive transition metal catalysts and the associated ligands, which often constitute a significant portion of the raw material budget in fine chemical synthesis. Additionally, the removal of heavy metal catalysts negates the need for specialized scavenging resins or complex extraction protocols designed to lower metal residues to ppm levels, thereby reducing both material costs and processing time. The use of commodity chemicals like elemental iodine, DMSO, and simple aryl ketones ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or proprietary reagents that may be subject to market volatility.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of cheap and readily available starting materials that are widely produced in the global chemical market. By avoiding the use of precious metals such as palladium, copper, or rhodium, manufacturers can achieve drastic reductions in raw material expenditures. Furthermore, the simplified workup procedure, which relies on standard filtration and chromatography rather than complex metal removal technologies, lowers the operational expenditure (OPEX) related to waste treatment and solvent recovery. This streamlined approach translates directly into a more competitive pricing structure for the final pharmaceutical intermediates, allowing companies to maintain healthy margins even in a price-sensitive market environment.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures a high degree of supply security, as these materials are produced by multiple vendors globally, reducing the risk of single-source dependency. The robustness of the reaction conditions, which do not require inert atmospheres or strictly anhydrous solvents, means that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized nitrogen blanketing or drying systems. This flexibility allows for greater agility in production scheduling and facilitates the rapid ramp-up of capacity to meet fluctuating demand from downstream API manufacturers. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly reduced, ensuring continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles by minimizing the use of toxic heavy metals and reducing the generation of hazardous waste streams. The scalability of the process has been validated from gram levels to larger batches, demonstrating its suitability for commercial scale-up of complex pharmaceutical intermediates. The simplified effluent profile, devoid of heavy metal contaminants, eases the burden on wastewater treatment facilities and ensures compliance with increasingly stringent environmental regulations. This sustainability advantage not only reduces regulatory risk but also enhances the corporate social responsibility (CSR) profile of the manufacturing entity, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metal promoter, eliminating the need for costly and toxic heavy metal catalysts often found in traditional triazole synthesis.
Q: What are the key advantages regarding reaction conditions?
A: The method operates under ambient atmospheric conditions without the need for strict anhydrous or oxygen-free environments, significantly simplifying operational requirements and reducing equipment costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol has been demonstrated to be easily scalable from gram levels to larger batches, utilizing cheap and readily available starting materials like aryl ethyl ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and ensuring supply chain stability. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate adheres to the highest industry standards. We are committed to leveraging advanced technologies like the iodine-promoted cyclization method to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs for your next-generation therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in metal-free synthesis can drive efficiency and innovation in your pharmaceutical projects.
