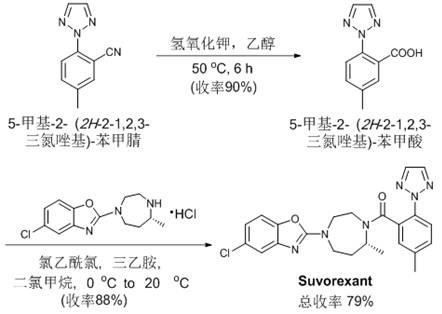
Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Platform for Complex Pharmaceutical Intermediates
Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Platform for Complex Pharmaceutical Intermediates
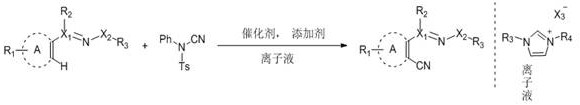
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more atom-economical methodologies, a trend vividly exemplified by the technological breakthroughs detailed in patent CN108690007B. This intellectual property introduces a sophisticated transition metal-catalyzed C-H coupling reaction that utilizes N1,N3-disubstituted imidazole-type ionic liquids as a sustainable solvent system. By employing N-cyano-N-phenyl-p-toluenesulfonamide as a safe and efficient cyano source, this method enables the direct ortho-cyanation of aromatic rings and unsaturated aliphatic rings under remarkably mild conditions. For R&D directors and process chemists, this represents a significant leap forward, offering a pathway to synthesize high-value nitrile intermediates without the need for harsh pre-activation steps or toxic reagents. The technology not only addresses the growing regulatory pressure for environmentally benign manufacturing but also provides a robust platform for generating diverse molecular libraries essential for modern drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group into an aromatic framework has relied on classical transformations such as the Rosenmund-von Braun reaction or the Sandmeyer reaction, both of which suffer from severe operational and environmental drawbacks. These traditional protocols typically necessitate the use of stoichiometric quantities of highly toxic copper(I) cyanide, posing significant health risks to operators and creating complex waste disposal challenges for environmental compliance teams. Furthermore, alternative cross-coupling strategies involving aromatic halides require a preliminary halogenation step to activate the substrate, adding unnecessary synthetic length and generating stoichiometric amounts of halogenated byproducts that complicate downstream purification. The reliance on high temperatures, oxygen-free environments, and expensive specialized ligands in many existing C-H activation methods further exacerbates production costs and limits the feasibility of large-scale commercial manufacturing for cost-sensitive pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent leverages a chelation-assisted C-H activation strategy that proceeds efficiently at room temperature, eliminating the energy-intensive heating requirements of conventional synthesis. By utilizing N-cyano-N-phenyl-p-toluenesulfonamide as the cyanating agent, the reaction avoids the handling of volatile and lethal hydrogen cyanide or unstable metal cyanides, thereby drastically improving workplace safety profiles. The integration of ionic liquids as the reaction medium not only enhances the solubility of polar intermediates but also enables the seamless recycling of the catalytic system, a feature that is virtually absent in traditional organic solvent-based processes. This approach allows for the direct functionalization of unactivated C-H bonds on pyridines, oxime ethers, and azo compounds, streamlining the synthetic route and significantly reducing the overall carbon footprint associated with the production of ortho-cyanated derivatives.

Mechanistic Insights into Transition Metal-Catalyzed C-H Cyanation
The core of this technological advancement lies in the intricate interplay between the transition metal catalyst, the directing group on the substrate, and the unique properties of the ionic liquid solvent. The reaction mechanism typically initiates with the coordination of the transition metal center, such as a Rhodium(III) or Ruthenium(II) species, to a nitrogen-containing directing group present on the substrate, such as a pyridyl or oxime moiety. This coordination facilitates the selective cleavage of the proximal ortho-C-H bond through a concerted metalation-deprotonation (CMD) pathway or a similar electrophilic substitution mechanism, forming a stable metallacycle intermediate. The presence of silver additives, such as silver acetate or silver bis(trifluoromethanesulfonyl)imide, plays a critical role in abstracting halide ligands from the metal precursor to generate the active cationic catalytic species, while also potentially assisting in the transmetallation or oxidative addition steps required for cyano group transfer.
Once the metallacycle is formed, the N-cyano-N-phenyl-p-toluenesulfonamide acts as an electrophilic cyanating source, transferring the cyano group to the metal center or directly to the activated carbon position. The ionic liquid environment stabilizes the charged intermediates and transition states throughout the catalytic cycle, preventing catalyst deactivation and aggregation which are common pitfalls in homogeneous catalysis. Following the reductive elimination step that releases the ortho-cyanated product, the catalyst is regenerated and remains dissolved in the ionic liquid phase, ready for subsequent turnover. This mechanistic robustness ensures high functional group tolerance, allowing sensitive moieties like halogens, esters, and aldehydes to remain intact during the transformation, which is crucial for the late-stage functionalization of complex drug candidates where preserving structural integrity is paramount.
How to Synthesize Ortho-Cyanated Derivatives Efficiently
Executing this synthesis requires precise control over reagent stoichiometry and mixing protocols to maximize yield and purity while maintaining the integrity of the recyclable solvent system. The standard operating procedure involves charging a clean reactor with the specific aromatic or alkenyl substrate, the solid cyano source, the transition metal catalyst precursor, and the necessary silver additive in the designated ionic liquid. The mixture is then stirred at ambient temperature for a defined period, typically around 24 hours, allowing the C-H activation and cyanation to reach completion without external heating. Detailed standardized synthesis steps for specific substrates, including workup and purification protocols, are outlined below to ensure reproducibility and quality control.
- Charge a clean reactor with the aromatic substrate (heterocycle, oxime ether, or azo compound), N-cyano-N-phenyl-p-toluenesulfonamide, transition metal catalyst, silver additive, and ionic liquid solvent.
- Stir the reaction mixture at room temperature for approximately 24 hours to allow the C-H coupling cyanation to proceed efficiently without external heating.
- Upon completion, extract the product using cyclohexane, separate the organic layer, remove solvent under reduced pressure, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-based C-H cyanation technology offers transformative benefits that extend far beyond simple yield improvements. The ability to recycle the solvent and catalyst system multiple times without significant degradation in performance translates directly into substantial cost savings on raw materials, effectively decoupling production volume from linear reagent consumption. By eliminating the need for pre-activation steps like halogenation, the process reduces the number of unit operations required, shortening the overall manufacturing cycle time and minimizing the inventory of hazardous intermediates that must be stored on-site. This streamlined workflow enhances supply chain reliability by reducing dependency on multiple specialized reagent suppliers and mitigating the risks associated with the transport and storage of toxic cyanide sources.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the eliminaton of expensive pre-functionalization steps and the reuse of the catalytic system. Since the ionic liquid and catalyst can be recovered and reused for at least five cycles without obvious reduction in yield, the effective cost per kilogram of the final API intermediate is significantly lowered. Furthermore, the use of a solid, stable cyano source avoids the premium pricing and special handling fees associated with gaseous or liquid cyanide reagents, leading to a more predictable and optimized cost structure for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Operational simplicity is a key driver for supply continuity, and this method excels by utilizing readily available starting materials and avoiding air-sensitive or moisture-sensitive conditions that often cause batch failures. The mild room temperature conditions reduce the strain on utility infrastructure, such as cooling water and steam, ensuring that production can proceed even during periods of limited utility availability. Additionally, the high functional group tolerance minimizes the formation of difficult-to-remove impurities, reducing the burden on purification resources and ensuring consistent delivery of high-purity materials to downstream formulation teams.
- Scalability and Environmental Compliance: From a regulatory perspective, the replacement of volatile organic compounds (VOCs) with non-flammable, low-vapor-pressure ionic liquids significantly simplifies environmental permitting and waste management. The process generates less hazardous waste due to the absence of stoichiometric metal byproducts and halogenated salts, aligning perfectly with corporate sustainability goals and increasingly stringent global environmental regulations. This green chemistry profile not only future-proofs the manufacturing asset against regulatory tightening but also enhances the marketability of the final pharmaceutical products to eco-conscious consumers and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H cyanation technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the primary advantages of using ionic liquids over traditional organic solvents in this cyanation process?
A: Ionic liquids offer superior thermal and chemical stability, negligible vapor pressure, and non-flammability, significantly enhancing operational safety. Furthermore, they facilitate catalyst recycling, allowing the reaction system to be reused multiple times without significant loss in yield, which drastically lowers long-term production costs.
Q: How does this method improve upon traditional Rosenmund-von Braun or Sandmeyer reactions?
A: Traditional methods often require harsh conditions, stoichiometric amounts of toxic copper cyanide, and pre-activation of substrates (e.g., halogenation). This novel C-H activation approach operates at room temperature, eliminates the need for substrate pre-functionalization, and uses a safer, solid cyano source, thereby simplifying the workflow and reducing hazardous waste generation.
Q: Is the catalyst system scalable for industrial production of API intermediates?
A: Yes, the protocol is designed for scalability. The use of robust transition metal catalysts (such as Rhodium or Ruthenium complexes) combined with a recyclable ionic liquid medium supports commercial scale-up. The mild reaction conditions reduce energy consumption, and the high functional group tolerance ensures applicability across diverse molecular libraries required for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Cyanated Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating cutting-edge academic research into robust, commercial-grade manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory patents like CN108690007B can be reliably reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of ortho-cyanated intermediate meets the exacting standards required for GMP pharmaceutical synthesis. Our commitment to quality and consistency makes us the preferred partner for global innovators seeking to secure their supply chains for complex small molecules.
We invite you to leverage our technical expertise to evaluate the feasibility of integrating this green cyanation technology into your specific project pipelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are prepared to provide specific COA data for analogous structures and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timelines while optimizing your overall cost of goods sold.