Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Strategy for Commercial Scale-up
Revolutionizing Ortho-Cyanation: A Green Transition Metal Catalysis Strategy for Commercial Scale-up
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. A pivotal advancement in this domain is detailed in Chinese Patent CN108690007B, which discloses a novel transition metal-catalyzed C-H coupling reaction for the efficient preparation of ortho-cyanated aromatic ring or unsaturated aliphatic ring compounds. This technology represents a paradigm shift from traditional stoichiometric methods to a catalytic, room-temperature process utilizing N1,N3-disubstituted imidazole-type ionic liquids as the reaction medium. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent offers a compelling value proposition by addressing critical pain points such as toxic waste generation, harsh reaction conditions, and high operational costs associated with conventional cyanation techniques.
The core innovation lies in the synergistic combination of a robust transition metal catalyst system, specifically rhodium, ruthenium, iridium, or cobalt complexes, with a specialized ionic liquid solvent. Unlike volatile organic solvents that pose significant environmental and safety hazards, these ionic liquids provide a stable, non-flammable environment that facilitates the activation of inert C-H bonds. The use of N-cyano-N-phenyl-p-toluenesulfonamide as a safe and stable cyano source further distinguishes this method from hazardous alternatives like copper cyanide. By enabling direct functionalization without the need for substrate pre-activation, this process not only streamlines the synthetic route but also significantly enhances the atom economy, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group into an aromatic framework has relied heavily on classical reactions such as the Rosenmund-von Braun reaction and the Sandmeyer reaction. These traditional pathways are fraught with substantial operational drawbacks that hinder their efficiency in modern large-scale manufacturing. Primarily, they necessitate the use of stoichiometric amounts of highly toxic copper cyanide (CuCN), which generates massive quantities of heavy metal waste that requires expensive and rigorous disposal protocols. Furthermore, these reactions often demand extreme conditions, including high temperatures and strictly anhydrous environments, which escalate energy consumption and increase the risk of thermal runaway incidents. Another critical limitation is the requirement for pre-functionalized substrates, such as aryl halides, which adds extra synthetic steps, reduces overall yield, and increases the cost of raw materials. The reliance on polar, toxic organic solvents exacerbates the environmental footprint, creating a significant barrier for companies aiming to meet stringent global sustainability standards.
The Novel Approach
In stark contrast, the methodology described in patent CN108690007B introduces a streamlined, one-step C-H coupling strategy that operates under remarkably mild conditions. By leveraging the unique solvation properties of ionic liquids, the reaction proceeds efficiently at room temperature, eliminating the need for energy-intensive heating. The use of N-cyano-N-phenyl-p-toluenesulfonamide as the cyanating agent provides a safer, solid alternative to gaseous or highly toxic liquid cyanide sources, significantly improving workplace safety. Moreover, the direct activation of the C-H bond bypasses the need for halogenated precursors, effectively shortening the synthetic timeline and reducing the accumulation of halogenated byproducts. This approach not only simplifies the purification process but also aligns perfectly with the principles of green chemistry, offering a sustainable pathway for cost reduction in pharmaceutical intermediates manufacturing.

The versatility of this new approach is further evidenced by its broad substrate scope, accommodating various aromatic heterocycles, oxime ethers, and azo compounds. As illustrated in the reaction schemes, the system tolerates a wide range of functional groups, allowing for the late-stage modification of complex drug molecules. This flexibility is crucial for medicinal chemists who need to rapidly generate molecular libraries for structure-activity relationship (SAR) studies without being constrained by incompatible protecting groups or sensitive functionalities. The ability to perform these transformations in a single pot with high regioselectivity underscores the robustness of the catalytic system and its potential for widespread adoption in the fine chemical industry.
Mechanistic Insights into Transition Metal-Catalyzed C-H Cyanation
At the heart of this transformative technology is a sophisticated catalytic cycle driven by transition metals, typically rhodium(III) or similar congeners. The mechanism initiates with the coordination of the directing group on the substrate to the metal center, facilitating the selective cleavage of the proximal C-H bond through a concerted metalation-deprotonation (CMD) pathway or electrophilic substitution. This step forms a stable metallacycle intermediate, which is the key determinant of the reaction's regioselectivity. The presence of silver additives, such as silver bistrifluoromethylsulfonylimide or silver acetate, plays a dual role: they act as halide scavengers to generate the active cationic metal species and assist in the transmetallation or ligand exchange processes necessary for the subsequent insertion of the cyano group. The ionic liquid solvent is not merely a passive medium; it actively stabilizes the charged intermediates and transition states through electrostatic interactions, lowering the activation energy barrier and enabling the reaction to proceed at ambient temperatures.
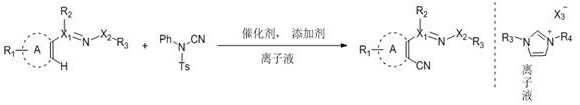
Impurity control is another critical aspect where this mechanistic understanding translates into practical benefits. Traditional methods often suffer from poor selectivity, leading to poly-cyanated byproducts or homocoupling impurities that are difficult to separate. In this ionic liquid system, the steric and electronic environment provided by the solvent-catalyst complex enforces strict mono-cyanation at the ortho-position relative to the directing group. The mild reaction conditions prevent the decomposition of sensitive functional groups, thereby minimizing the formation of degradation products. Furthermore, the recyclability of the ionic liquid phase allows for the retention of the catalyst, ensuring consistent performance over multiple batches and reducing the variability in impurity profiles that often plagues batch-to-batch production in conventional solvent systems.
How to Synthesize O-Cyanated Aromatic Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and industrial production. The process involves a straightforward charging sequence where the substrate, cyano source, catalyst, additive, and ionic liquid are combined in a reactor. The mixture is then stirred at room temperature for a defined period, typically around 24 hours, allowing the transformation to reach completion without the need for specialized heating equipment. Following the reaction, a simple extraction protocol using cyclohexane separates the organic product from the ionic liquid phase. The detailed standardized synthesis steps for this procedure are outlined below to ensure reproducibility and quality control.
- Charge the reactor with the substrate (aromatic heterocycle, oxime ether, or azo compound), N-cyano-N-phenyl-p-toluenesulfonamide, transition metal catalyst (e.g., Rhodium dimer), silver additive, and the ionic liquid solvent.
- Stir the reaction mixture at room temperature for approximately 24 hours to allow the C-H coupling cyanation to proceed efficiently without external heating.
- Upon completion, extract the product using cyclohexane, separate the organic layer, remove solvent under reduced pressure, and purify via silica gel chromatography while recycling the ionic liquid layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-based cyanation technology offers profound strategic advantages that extend beyond mere technical feasibility. The most significant benefit is the drastic simplification of the supply chain regarding raw material sourcing and waste management. By eliminating the need for pre-activated halogenated starting materials, manufacturers can source cheaper, more abundant bulk chemicals, thereby insulating the production process from the volatility of specialized reagent markets. Additionally, the replacement of volatile organic solvents with non-flammable ionic liquids significantly reduces the regulatory burden associated with solvent storage, handling, and emission controls, leading to lower compliance costs and improved facility safety ratings.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the recyclability of the reaction medium. Since the ionic liquid and the catalyst remain in the bottom layer after product extraction, they can be reused for multiple cycles without the need for fresh additions. This closed-loop system dramatically lowers the cost of goods sold (COGS) by minimizing solvent purchase and disposal expenses. Furthermore, the operation at room temperature eliminates the energy costs associated with heating and cooling large reactors, contributing to substantial long-term savings. The high yields and selectivity reported in the patent also mean less raw material is wasted on side products, maximizing the output per unit of input.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent production schedules, free from the delays often caused by the sensitivity of traditional cyanation methods to moisture or oxygen. The use of stable, solid cyano sources like N-cyano-N-phenyl-p-toluenesulfonamide mitigates the risks associated with transporting and storing hazardous gases or unstable liquids. This stability translates into a more resilient supply chain capable of meeting tight deadlines and scaling up rapidly to meet market demand without the bottlenecks typical of complex multi-step syntheses.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the inherent safety of the ionic liquid system. The lack of vapor pressure reduces the risk of explosions and fire, making it easier to obtain permits for larger reactors. From an environmental perspective, the reduction in heavy metal waste and volatile organic compound (VOC) emissions aligns with increasingly strict global environmental regulations. This proactive approach to sustainability not only future-proofs the manufacturing process but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
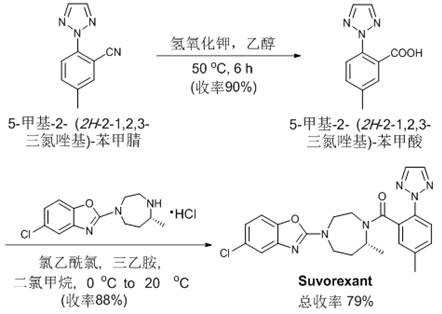
The commercial viability of this method is further validated by its successful application in the synthesis of high-value drug candidates, such as the anti-insomnia medication Suvorexant. The ability to efficiently construct the core cyanated scaffold required for such molecules demonstrates the technology's readiness for GMP manufacturing. By integrating this green chemistry approach, suppliers can offer clients a competitive edge through faster time-to-market and lower development costs, solidifying their position as a preferred partner for complex intermediate synthesis.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and operational parameters of this cyanation technology, we have compiled a set of frequently asked questions based on the patent data. These insights are intended to clarify the capabilities of the process and its suitability for various pharmaceutical applications. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of using ionic liquids over traditional organic solvents in this cyanation process?
A: Ionic liquids offer superior thermal and chemical stability, negligible vapor pressure which enhances safety, and the unique ability to be recycled multiple times without significant loss in catalytic activity, drastically reducing waste and solvent costs compared to volatile organic compounds.
Q: Does this method require pre-activation of the aromatic substrate?
A: No, one of the key innovations of this patent is the direct C-H functionalization which eliminates the need for pre-halogenation or other activation steps typically required in traditional cross-coupling reactions, thereby shortening the synthetic route.
Q: Can the catalyst and solvent system be reused for subsequent batches?
A: Yes, the patent explicitly demonstrates that the ionic liquid layer containing the catalyst can be recycled for at least five cycles with no obvious reduction in product yield, providing a sustainable and economically viable model for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Cyanated Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid-catalyzed C-H cyanation technology described in patent CN108690007B. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instruments to guarantee stringent purity specifications for every batch of high-purity pharmaceutical intermediates we deliver. We are committed to leveraging our technical expertise to optimize this catalytic system for your specific molecule, ensuring maximum yield and minimal impurity levels.
We invite you to collaborate with us to unlock the full potential of this efficient synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your supply chain.
