Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which significantly enhance metabolic stability and bioavailability. A groundbreaking development in this sector is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are ubiquitous in modern medicinal chemistry, serving as critical scaffolds in blockbuster drugs such as Sitagliptin and various antifungal agents. The ability to introduce a trifluoromethyl group directly onto the triazole ring via a streamlined catalytic process represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines. This novel approach not only simplifies the synthetic route but also ensures high purity profiles essential for downstream API manufacturing.
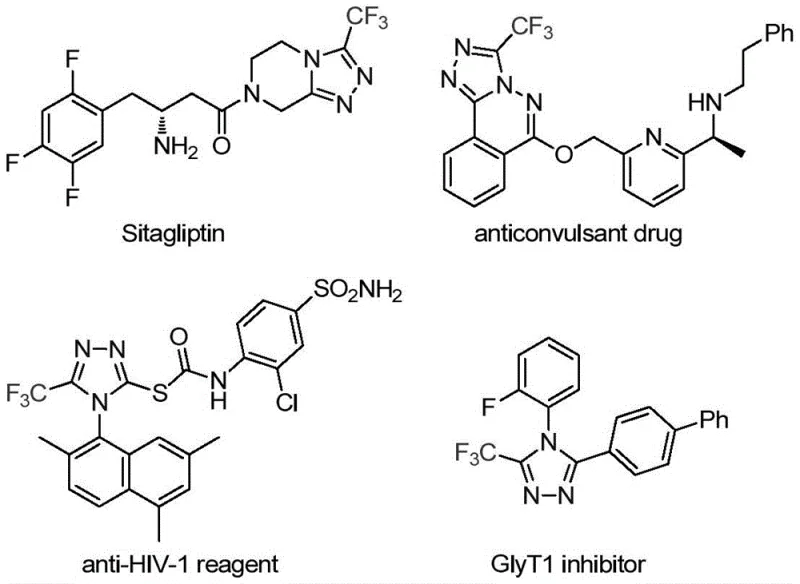
The strategic importance of this technology cannot be overstated, as the 1,2,4-triazole core is an extremely important five-membered nitrogen-containing heterocyclic ring found in countless molecular frameworks with biological activities. By leveraging a dual-metal catalytic system involving molybdenum and copper, this method overcomes many of the thermodynamic and kinetic barriers associated with traditional triazole synthesis. For R&D directors and process chemists, this translates to a versatile platform capable of generating diverse libraries of trifluoromethylated heterocycles with predictable outcomes. Furthermore, the use of readily available starting materials positions this technology as a cornerstone for cost reduction in pharmaceutical intermediate manufacturing, allowing companies to produce high-value building blocks without relying on exotic or prohibitively expensive reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant challenges that hinder efficient commercial production. Traditional literature reports primarily rely on the cyclization reaction of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone. These classical routes often suffer from harsh reaction conditions, requiring extreme temperatures or pressures that can degrade sensitive functional groups and lead to complex impurity profiles. Additionally, alternative methods involving copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane present severe safety hazards due to the explosive nature of diazo compounds, making them unsuitable for large-scale operations. The reliance on such dangerous reagents necessitates specialized equipment and rigorous safety protocols, drastically inflating the operational expenditure and extending the lead time for high-purity intermediates.
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN113307778A introduces a paradigm shift by employing a molybdenum and copper co-catalyzed cycloaddition reaction. This innovative strategy utilizes trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, reacting under remarkably mild conditions of 70~90℃. The elimination of hazardous diazonium salts and the avoidance of extreme thermal stress significantly enhance the safety profile of the process, making it inherently safer for operators and facilities. Moreover, the reaction efficiency is exceptionally high, with reported yields reaching up to 99% for certain substrates, which minimizes waste generation and maximizes atom economy. This streamlined approach allows for the direct synthesis of 3,4-disubstituted 1,2,4-triazoles, a structural class that has been historically difficult to access efficiently, thereby opening new avenues for drug discovery and process optimization.
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
The success of this synthetic methodology lies in the synergistic interaction between the molybdenum hexacarbonyl activator and the cuprous acetate catalyst. Mechanistically, the reaction initiates with the coordination of molybdenum hexacarbonyl to the functionalized isonitrile, forming a reactive metal-isocyanide complex that lowers the activation energy for the subsequent cycloaddition step. Simultaneously, the copper species promotes a [3+2] cycloaddition between the activated isonitrile and the trifluoroethylimidoyl chloride, facilitating the formation of the five-membered triazole ring intermediate. This dual-catalyst system ensures that the reaction proceeds smoothly even with sterically hindered or electronically diverse substrates, providing the broad substrate scope observed in the experimental data. The final step involves the removal of the triphenylphosphine oxide moiety under the action of water present in the system, yielding the final 3-trifluoromethyl-substituted 1,2,4-triazole compound with high fidelity.
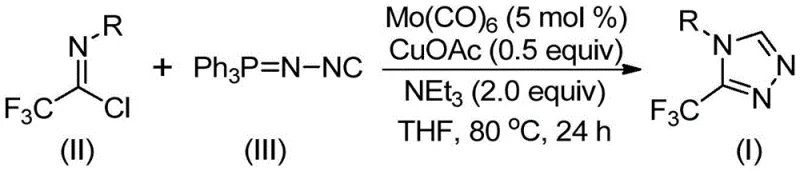
From an impurity control perspective, this mechanism offers distinct advantages over acid-mediated or high-temperature cyclizations. The mild basic conditions provided by triethylamine prevent the hydrolysis of sensitive groups such as esters or nitriles that might be present on the aromatic rings of the substrates. Furthermore, the specific stoichiometry of the catalysts—typically 5 mol % for Mo(CO)6 and 0.5 equivalents for CuOAc—ensures that side reactions like polymerization of the isonitrile are minimized. The use of molecular sieves in the reaction mixture further aids in controlling the water content, striking a balance between promoting the final deprotection step and preventing premature degradation of the imidoyl chloride. For quality control teams, this means a cleaner crude reaction mixture that requires less aggressive purification, ultimately preserving the integrity of the final product and ensuring it meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized parameters outlined in the patent to achieve maximum yield and reproducibility. The process is designed to be operationally simple, involving the mixing of solid and liquid reagents in a standard organic solvent followed by heating, which facilitates easy technology transfer from R&D to production scales. The detailed standardized synthesis steps below provide a clear roadmap for chemists to replicate the high efficiencies reported in the intellectual property, ensuring that the critical balance of catalyst loading and reaction time is maintained for optimal results.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing trifluoroethylimidoyl chloride and functionalized isonitriles, manufacturers can source starting materials that are not only commercially available but also significantly cheaper than the specialized hydrazines or diazo compounds required by older methods. This shift in raw material strategy leads to substantial cost savings in the overall bill of materials, allowing companies to improve their margins or offer more competitive pricing to their downstream clients. Additionally, the robustness of the reaction conditions reduces the risk of batch failures, ensuring a more consistent and reliable supply of critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts often used in cross-coupling alternatives, combined with the use of relatively inexpensive copper and molybdenum salts, drives down the direct cost of goods sold. The high atom economy of the cycloaddition reaction means that less raw material is wasted as byproduct, further enhancing the economic efficiency of the process. Furthermore, the mild reaction temperatures reduce energy consumption compared to high-pressure or high-temperature alternatives, contributing to lower utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Because the starting materials are stable and widely available commodity chemicals, the supply chain is less vulnerable to disruptions caused by the scarcity of exotic reagents. The scalability of the process, which has been demonstrated from milligram to gram levels in the patent data, suggests a smooth path to multi-kilogram and ton-scale production without the need for specialized reactor configurations. This reliability allows supply chain planners to forecast demand more accurately and maintain leaner inventory levels while still meeting customer delivery timelines.
- Scalability and Environmental Compliance: The process operates in common organic solvents like THF, which are easily recovered and recycled in modern solvent recovery units, minimizing environmental impact and waste disposal costs. The absence of toxic heavy metals or explosive precursors simplifies the regulatory compliance landscape, reducing the administrative burden and permitting time required for new production lines. This environmental friendliness aligns with the growing global demand for green chemistry practices, enhancing the corporate sustainability profile of manufacturers who adopt this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl-triazole synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making regarding process adoption and integration into existing manufacturing workflows.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents with yields ranging from moderate to excellent.
Q: Is this process suitable for large-scale manufacturing?
A: The invention explicitly states the method can be expanded to gram-level reactions and beyond, utilizing cheap and commercially available starting materials suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics and agrochemicals. Our team of expert process chemists has thoroughly analyzed the Mo/Cu co-catalyzed pathway described in CN113307778A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet the most rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific solvent and catalyst requirements of this chemistry, ensuring that every batch delivered to our partners reflects the highest levels of consistency and purity.
We invite global pharmaceutical and fine chemical companies to collaborate with us to leverage this cutting-edge synthesis technology for their specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can optimize your budget. We encourage you to contact us today to discuss your specific COA data requirements and to receive detailed route feasibility assessments for your target molecules, ensuring a seamless transition from laboratory concept to commercial reality.
