Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Production
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Production
The pharmaceutical industry is constantly seeking robust, scalable, and stereoselective methods to construct complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN112209947A, which discloses a novel synthesis method for chiral indoxazinone compounds. These compounds, characterized by their fused indole-oxazinone core, have demonstrated promising cytotoxic activity against PC-3 human prostate cancer cells, marking them as valuable candidates for anticancer drug development. The innovation lies not just in the biological potential of the final molecule, but in the elegance of the synthetic route itself. By leveraging a dual catalytic system comprising chiral phosphoric acid and hexafluoroisopropanol, the inventors have achieved a transformation that operates under remarkably mild conditions, bypassing the harsh reagents and extreme temperatures often required in traditional heterocycle synthesis. This report analyzes the technical merits of this protocol and its implications for cost-effective, high-purity manufacturing of pharmaceutical intermediates.
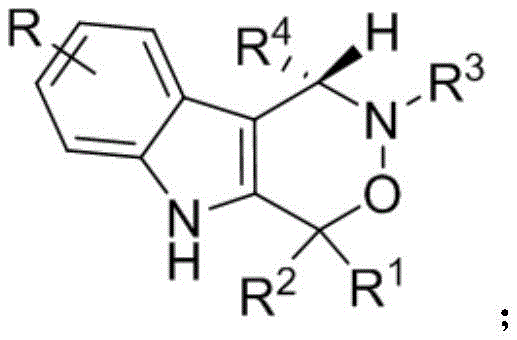
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole-fused ring systems has been fraught with significant technical and economic challenges that hinder efficient commercialization. Traditional synthetic pathways often rely heavily on transition metal catalysis, which introduces severe complications regarding residual metal contamination—a critical quality attribute for any Active Pharmaceutical Ingredient (API). The removal of trace metals such as palladium, rhodium, or copper requires additional downstream processing steps, including specialized scavenging resins or repeated recrystallizations, which inevitably drive up manufacturing costs and extend lead times. Furthermore, many conventional methods necessitate aggressive reaction conditions, including strong acids, high pressures, or cryogenic temperatures, to achieve acceptable levels of stereocontrol. These harsh environments not only pose safety risks in a plant setting but also limit the functional group tolerance, preventing the use of diverse substrates and restricting the chemical space available for medicinal chemistry optimization. Consequently, the industry has long suffered from low yields and poor enantioselectivity when attempting to access these complex architectures on a meaningful scale.
The Novel Approach
In stark contrast to these legacy issues, the methodology described in CN112209947A represents a paradigm shift towards sustainable and efficient organocatalysis. The core innovation involves the direct cycloaddition of 2-indolemethanol derivatives with nitrones, facilitated by a sophisticated chiral phosphoric acid catalyst system. This approach eliminates the need for toxic heavy metals entirely, replacing them with organic molecules that are easier to handle and dispose of. The reaction proceeds smoothly in toluene, a standard industrial solvent, at near-ambient temperatures ranging from 20°C to 30°C. This mild thermal profile drastically reduces energy consumption compared to refluxing conditions and minimizes the risk of thermal degradation of sensitive intermediates. Moreover, the inclusion of hexafluoroisopropanol (HFIP) as a co-catalyst or additive plays a pivotal role in enhancing the reactivity and selectivity of the transformation through hydrogen-bonding interactions. The result is a versatile platform capable of accommodating a wide array of substituents on both the indole and nitrone components, delivering products with high structural diversity and exceptional optical purity.
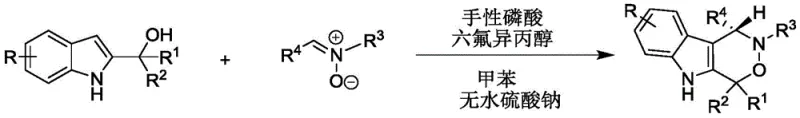
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
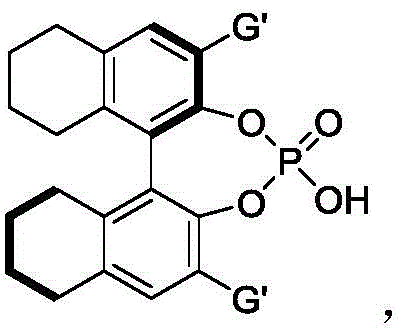
To fully appreciate the value of this technology for R&D teams, one must delve into the mechanistic nuances that enable such high fidelity in stereocontrol. The reaction is believed to proceed via a concerted or stepwise [3+2] cycloaddition mechanism, where the chiral phosphoric acid acts as a bifunctional catalyst. The acidic proton of the phosphate moiety activates the electrophilic nitrone species through hydrogen bonding, while the basic phosphoryl oxygen simultaneously interacts with the nucleophilic 2-indolemethanol. This dual activation brings the reactants into close proximity within a well-defined chiral pocket created by the bulky substituents on the catalyst backbone, typically binaphthyl or spiro-based skeletons. The presence of hexafluoroisopropanol further amplifies this effect; its strong hydrogen-bond donating ability helps to organize the transition state and stabilize charged intermediates, effectively lowering the activation energy barrier. This synergistic catalysis ensures that the cyclization occurs with precise facial selectivity, leading to the formation of the desired enantiomer with minimal formation of the opposite isomer.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction is highly selective and operates under mild conditions, the formation of side products such as polymerization byproducts or decomposition fragments is significantly suppressed. The use of anhydrous sodium sulfate as an additive likely serves to sequester water generated during the reaction or present in the reagents, preventing hydrolysis of the sensitive nitrone or the final oxazinone product. This inherent cleanliness of the reaction profile simplifies the purification process, often allowing for high-purity isolation via standard silica gel chromatography without the need for extensive preparative HPLC. For process chemists, this translates to a more robust and predictable manufacturing process where the critical quality attributes of the intermediate are consistently met batch after batch.
How to Synthesize Chiral Indoxazinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific stoichiometry and catalyst loading defined in the patent to ensure optimal results. The general protocol involves dissolving the 2-indolemethanol and nitrone substrates in toluene, followed by the addition of the chiral phosphoric acid catalyst and hexafluoroisopropanol. The mixture is then stirred at a controlled temperature, typically around 30°C, until thin-layer chromatography (TLC) indicates complete consumption of the starting materials. The workup procedure is straightforward, involving filtration to remove the drying agent and concentration under reduced pressure, followed by purification. While the general principles are clear, the exact execution depends on the specific substrate combination and scale. For detailed operational parameters and specific examples of substrate scope, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone in toluene with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol as co-catalysts to the mixture.
- Stir the reaction at 20-30°C until completion, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from precious metal catalysts to organic phosphoric acids represents a significant reduction in raw material costs, as chiral phosphoric acids can often be recovered or are less expensive than their metallic counterparts on a per-kilogram basis. Furthermore, the elimination of metal scavenging steps simplifies the supply chain by removing the need for specialized filtration media and reducing the volume of hazardous waste generated. This streamlining of the process flow enhances overall throughput and reduces the turnaround time for producing critical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by its operational simplicity and high atom economy. By conducting the reaction at ambient temperatures (20-30°C), the facility saves substantially on heating and cooling utilities compared to processes requiring cryogenic conditions or high-temperature reflux. Additionally, the high yields reported across a broad range of substrates mean that less raw material is wasted, maximizing the output per batch. The absence of heavy metals also lowers the cost of waste disposal and regulatory compliance testing, as there is no need for rigorous ICP-MS analysis to verify metal limits. These factors combine to create a leaner, more cost-efficient manufacturing model that allows for competitive pricing of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. 2-indolemethanols and nitrones are commercially accessible or easily synthesized from common precursors, reducing the risk of supply disruptions associated with exotic reagents. The robustness of the reaction conditions—tolerating a variety of functional groups and operating in standard solvents like toluene—means that the process is less susceptible to variations in raw material quality or minor fluctuations in plant conditions. This reliability ensures consistent delivery schedules and reduces the likelihood of batch failures that could halt downstream API production. Moreover, the scalability of the method from gram to kilogram scales has been demonstrated, providing confidence that supply can be ramped up quickly to meet market demand without extensive process re-engineering.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry principles. The use of toluene, while requiring proper handling, is a well-understood solvent in the industry with established recovery protocols. More importantly, the avoidance of toxic heavy metals and harsh reagents significantly reduces the environmental footprint of the synthesis. The mild reaction conditions minimize the risk of runaway reactions or thermal hazards, enhancing plant safety. As regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing emissions and waste, adopting such a clean and efficient process future-proofs the supply chain against increasingly stringent environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral indoxazinone compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines.
Q: What are the key advantages of this synthesis method over traditional metal-catalyzed routes?
A: This method utilizes metal-free organocatalysis, eliminating the need for expensive transition metals and complex removal steps, thereby reducing production costs and environmental impact while maintaining high enantioselectivity.
Q: What is the typical enantiomeric excess (ee) achieved in this process?
A: The process demonstrates extremely high enantioselectivity, with specific examples achieving up to 96% ee, ensuring the production of high-purity chiral intermediates essential for pharmaceutical applications.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates under mild conditions (20-30°C) in common solvents like toluene, making it highly scalable, safe, and economically viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
The technological advancements presented in patent CN112209947A highlight the immense potential of chiral indoxazinones as key building blocks for novel anticancer therapies. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent research into reliable commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the bench to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral intermediate we deliver meets the highest standards of quality and enantiomeric excess required for global pharmaceutical registration.
We invite you to leverage our expertise to optimize your supply chain and accelerate your drug development timeline. Whether you require custom synthesis of specific indoxazinone derivatives or scale-up of the organocatalytic process described herein, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us be your strategic partner in bringing life-saving medicines to patients faster and more efficiently.
