Advanced Copper-Catalyzed Synthesis of Polysubstituted Isoquinolinone Derivatives for Commercial Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of Polysubstituted Isoquinolinone Derivatives for Commercial Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN111410656B, which discloses a robust preparation method for isoquinolinone derivatives. These compounds are not merely academic curiosities; they represent a critical class of structural units found extensively in natural products and plant alkaloids, possessing profound biological activities ranging from anti-inflammatory and antitumor properties to vasodilation effects. For R&D directors and procurement specialists alike, the ability to access these high-value scaffolds through a streamlined, catalytic process represents a substantial strategic advantage. The patent outlines a methodology that transforms readily available 8-alkynyl-1-aroyl naphthylamine compounds into polysubstituted isoquinolinones using a transition metal catalyst, thereby bypassing the limitations of classical stoichiometric approaches.
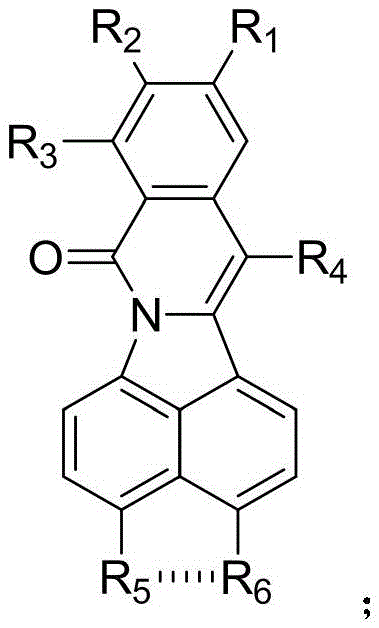
The structural versatility of the resulting isoquinolinone derivatives is a key factor in their utility across medicinal chemistry programs. As illustrated in the general structure, the core scaffold allows for extensive functionalization at multiple positions (R1 through R6), enabling the fine-tuning of physicochemical properties and biological potency. This flexibility is paramount for drug discovery teams aiming to optimize lead compounds. Furthermore, the patent highlights that these derivatives exhibit excellent fluorescence properties, opening additional avenues for their application in material science and diagnostic imaging. By leveraging this patented technology, manufacturers can produce high-purity intermediates that serve as foundational building blocks for next-generation therapeutics and advanced functional materials, ensuring a reliable supply chain for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinolinone skeleton has been fraught with significant technical challenges and safety hazards that impede efficient commercial scale-up. Traditional methodologies often rely on the amination of isocoumarins, a process that necessitates the pre-preparation of specific substrates and the use of refluxing acetic acid, leading to harsh reaction conditions and potential corrosion issues. Another prevalent route involves dilithiation reactions utilizing sec-butyllithium, a pyrophoric reagent that demands stringent cryogenic conditions and specialized handling equipment, thereby inflating operational costs and posing severe safety risks to personnel. Additionally, methods employing strong bases like triethylamine with 2-bromomethylbenzonitrile are frequently limited to electron-deficient substrates, restricting the chemical space accessible to chemists. These conventional pathways often suffer from poor atom economy, generate substantial waste streams, and lack the chemoselectivity required for complex molecule synthesis, making them ill-suited for modern green chemistry standards.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach detailed in the patent utilizes a transition metal-catalyzed cyclization strategy that fundamentally reshapes the synthesis paradigm. By employing 8-alkynyl-1-aroyl naphthylamine compounds as starting materials, the reaction proceeds through an elegant intramolecular cyclization facilitated by a catalytic amount of copper or silver salts. This method operates under remarkably mild conditions, typically between 80°C and 150°C, eliminating the need for cryogenic temperatures or super-stoichiometric amounts of hazardous organometallic reagents. The process is characterized by its "one-pot" nature, which drastically simplifies the workflow by removing the necessity for isolating unstable intermediates. This streamlined operation not only enhances the overall yield but also significantly reduces the solvent consumption and waste generation associated with multiple purification steps, aligning perfectly with the principles of sustainable manufacturing.
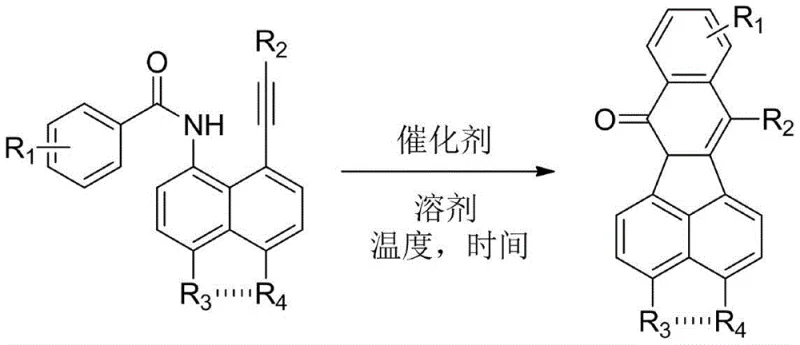
Mechanistic Insights into CuCN-Catalyzed Cyclization
The core of this technological advancement lies in the precise mechanistic pathway enabled by the copper catalyst, specifically copper(I) cyanide (CuCN), which acts as a potent Lewis acid and coordination center. The mechanism likely involves the activation of the alkyne moiety by the copper species, rendering it susceptible to nucleophilic attack by the adjacent amide carbonyl oxygen or nitrogen, depending on the specific tautomeric state. This activation lowers the energy barrier for the cyclization step, allowing the reaction to proceed efficiently at moderate temperatures without the need for external oxidants that are often required in C-H activation strategies. The use of aprotic polar solvents like N,N-dimethylformamide (DMF) further stabilizes the charged intermediates and ensures complete dissolution of the reactants, facilitating homogeneous catalysis. This controlled environment minimizes side reactions such as polymerization or decomposition, which are common pitfalls in alkyne chemistry, thereby ensuring high fidelity in the formation of the desired isoquinolinone core.
From an impurity control perspective, this catalytic system offers superior selectivity compared to non-catalytic thermal cyclizations. The patent data indicates that the reaction avoids the formation of complex mixtures often seen when using o-alkynyl benzamides under non-optimized conditions. The specific coordination geometry imposed by the catalyst directs the regioselectivity of the ring closure, ensuring that the lactam ring forms exclusively at the desired position. This high degree of selectivity is crucial for pharmaceutical applications where regulatory agencies mandate strict limits on unidentified impurities. By minimizing the generation of regioisomers and byproducts, the downstream purification burden is significantly alleviated, allowing for simpler crystallization or chromatography protocols. This results in a final product with a cleaner impurity profile, reducing the risk of batch rejection and ensuring consistent quality for clinical trial materials.
How to Synthesize Polysubstituted Isoquinolinone Derivatives Efficiently
Implementing this synthesis route in a practical setting requires careful attention to the stoichiometric ratios and thermal parameters defined in the patent to maximize efficiency. The standard protocol involves weighing the 8-alkynyl-1-aroyl naphthylamine substrate and the CuCN catalyst according to a molar ratio of 1.0:0.2, although ratios ranging from 1:0.1 to 1:1.5 are permissible depending on the specific substrate reactivity. These components are placed into a standard reaction vessel, and an aprotic solvent is added in sufficient quantity to ensure complete dissolution of the solid reactants. The reaction mixture is then heated to a temperature between 80°C and 150°C, with 120°C identified as the optimal setpoint for balancing reaction rate and energy consumption. Following the reaction period, which can range up to 36 hours depending on conversion, the mixture is cooled and subjected to a standard aqueous workup involving extraction with ethyl acetate.
- Weigh 8-alkynyl-1-aroyl naphthylamine compounds and CuCN catalyst at a molar ratio of 1.0: 0.2 and place them into a reaction vessel.
- Add an aprotic solvent such as N,N-dimethylformamide (DMF) until the starting materials are completely dissolved.
- Heat the reaction mixture to 120°C and stir for the required duration, followed by aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible economic and logistical benefits that directly impact the bottom line. The shift from stoichiometric, hazardous reagents to catalytic systems fundamentally alters the cost structure of the manufacturing process. By eliminating the need for expensive and dangerous reagents like sec-butyllithium, the direct material costs are significantly reduced, and the overhead associated with specialized safety infrastructure is minimized. Furthermore, the use of commodity chemicals such as CuCN and DMF ensures that the supply chain remains robust and resilient against market fluctuations, as these materials are widely available from multiple global vendors. This accessibility mitigates the risk of supply disruptions that often plague processes relying on niche or custom-synthesized reagents, ensuring continuous production schedules.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in reagent costs and waste disposal fees. Traditional methods often require excess amounts of strong bases or organometallics, which generate large volumes of saline or metallic waste that is costly to treat. In contrast, the catalytic nature of this new method means that only a small fraction of the metal salt is required per batch, leading to substantial savings in raw material expenditure. Additionally, the simplified one-pot procedure reduces the number of unit operations, lowering labor costs and energy consumption associated with heating, cooling, and transferring materials between vessels. These cumulative efficiencies result in a lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliability of the supply chain is bolstered by the simplicity and stability of the starting materials. The 8-alkynyl-1-aroyl naphthylamine precursors are synthesized from readily available building blocks, ensuring that the upstream supply is secure and scalable. Unlike processes that depend on unstable intermediates requiring immediate use or cold storage, the reagents in this protocol are stable solids that can be stocked in bulk without degradation. This stability allows for better inventory management and just-in-time manufacturing capabilities. Moreover, the mild reaction conditions reduce the wear and tear on reactor equipment, extending the lifespan of capital assets and reducing maintenance downtime, which further contributes to a consistent and reliable delivery schedule for customers.
- Scalability and Environmental Compliance: Scaling this process from the laboratory to industrial production is straightforward due to the absence of extreme conditions. The reaction does not require high-pressure autoclaves or cryogenic cooling loops, meaning it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This ease of scale-up accelerates the time-to-market for new drug candidates utilizing this scaffold. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding hazardous waste. By avoiding halogenated solvents in the reaction phase (using DMF or toluene instead) and minimizing heavy metal load through catalytic usage, the environmental footprint is kept low. This compliance reduces the regulatory burden and facilitates smoother audits, making the manufacturing site more attractive to international partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and strategic value of the method. Understanding these nuances is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the advantages of this copper-catalyzed method over traditional dilithiation routes?
A: Unlike traditional dilithiation methods which require hazardous sec-butyllithium reagents and cryogenic conditions, this novel approach utilizes stable copper catalysts under mild thermal conditions (80-150°C), significantly enhancing operational safety and reducing equipment costs.
Q: Can this synthesis method accommodate diverse substrate scopes for drug discovery?
A: Yes, the method demonstrates excellent substrate universality, successfully tolerating various substituents including halogens, alkyl groups, and heterocycles on the aromatic rings, making it ideal for generating diverse libraries of bioactive isoquinolinone analogs.
Q: Is the process suitable for large-scale industrial production?
A: Absolutely. The one-pot procedure eliminates the need for intermediate isolation, uses commercially available solvents like DMF, and operates at atmospheric pressure, ensuring seamless scalability from laboratory grams to multi-ton commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory method into a robust commercial reality. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isoquinolinone derivatives meets the highest international standards for quality and safety, ready for immediate use in drug development pipelines.
We invite you to collaborate with us to leverage this advanced technology for your specific projects. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, cost-effective, and scalable supply of complex heterocyclic intermediates, empowering your R&D efforts and accelerating your path to market success.
