Revolutionizing Quinoxaline Production: A Green Aqueous Phase Catalytic Approach for Industrial Scale
The pharmaceutical and fine chemical industries are currently undergoing a significant paradigm shift towards greener, more sustainable manufacturing processes, driven by both regulatory pressure and the economic necessity of efficiency. Patent CN102010376A introduces a groundbreaking methodology for the synthesis of quinoxaline compounds, a class of nitrogen-containing heterocycles critical to drug discovery and material science. This innovation replaces hazardous organic solvents with water and employs a novel, biodegradable acidic functional ionic liquid as a catalyst. By leveraging a quaternary ammonium cationic structure, this technology achieves exceptional reaction rates under mild conditions, addressing the longstanding challenges of toxicity and waste generation associated with traditional heterocyclic synthesis. For R&D directors and procurement strategists, this patent represents a viable pathway to modernize production lines, ensuring compliance with stringent environmental standards while simultaneously optimizing yield and purity profiles for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied heavily on the condensation of substituted o-phenylenediamines with 1,2-dicarbonyl compounds in organic media. Traditional protocols often necessitate the use of volatile and toxic solvents such as glacial acetic acid, methanol, or dimethyl sulfoxide (DMSO), which pose significant health risks to operators and create substantial burdens for waste management teams. Furthermore, these conventional reactions typically require extended heating periods ranging from 2 to 12 hours to reach acceptable conversion levels, leading to high energy consumption and reduced throughput in batch reactors. The reliance on non-biodegradable catalysts or harsh acidic conditions in these legacy methods frequently results in equipment corrosion and complex downstream purification processes, where removing trace solvent residues becomes a costly and time-consuming bottleneck. Additionally, the inability to easily recover and recycle catalysts in these organic systems contributes to inflated operational expenditures and a larger overall carbon footprint for the manufacturing facility.
The Novel Approach
In stark contrast, the technology disclosed in CN102010376A utilizes a biodegradable ionic liquid with a specific quaternary ammonium structure to catalyze the reaction in an aqueous environment. This approach eliminates the need for volatile organic compounds entirely, utilizing water as a safe, inexpensive, and non-flammable reaction medium that simplifies safety protocols and reduces fire hazards. The reaction proceeds rapidly at ambient temperatures between 20°C and 40°C, completing within a mere 10 to 60 minutes, which dramatically accelerates production cycles compared to thermal organic methods. 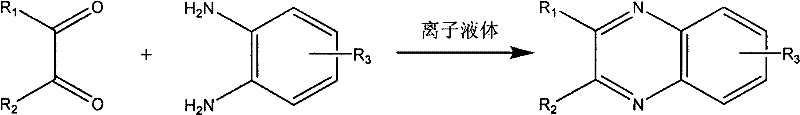 The process yields a solid-liquid two-phase system upon completion, allowing for straightforward filtration to isolate the crude product, thereby streamlining the workup procedure significantly. This novel methodology not only achieves superior yields ranging from 86% to 95% but also ensures that the catalytic system remains stable and active in the aqueous filtrate, enabling direct reuse for subsequent batches without loss of efficiency.
The process yields a solid-liquid two-phase system upon completion, allowing for straightforward filtration to isolate the crude product, thereby streamlining the workup procedure significantly. This novel methodology not only achieves superior yields ranging from 86% to 95% but also ensures that the catalytic system remains stable and active in the aqueous filtrate, enabling direct reuse for subsequent batches without loss of efficiency.
Mechanistic Insights into Biodegradable Ionic Liquid Catalysis
The core of this technological advancement lies in the unique structural properties of the acidic functional ionic liquid catalyst, which features a sulfonic acid group attached to a quaternary ammonium backbone. 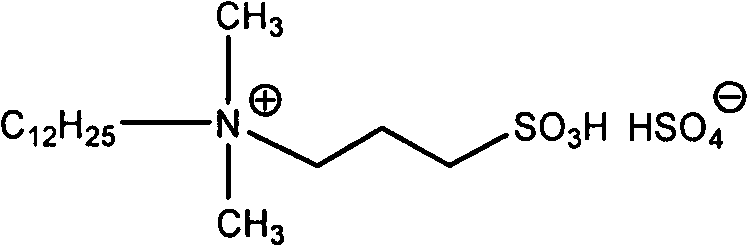 This dual-functionality design allows the molecule to act as both a Brønsted acid catalyst and a phase-transfer agent, facilitating the protonation of the carbonyl oxygen in the 1,2-dicarbonyl substrate to enhance its electrophilicity. The long alkyl chain (C12H25) present in the cationic structure imparts amphiphilic characteristics, which improves the solubility of organic reactants within the aqueous phase, effectively creating a micro-emulsion environment that maximizes molecular collisions. The bisulfate (HSO4-) counter-ion further contributes to the acidity of the medium, driving the condensation equilibrium forward without the need for external mineral acids that could degrade sensitive functional groups on the substrate. This precise molecular engineering ensures that the reaction proceeds with high chemoselectivity, minimizing the formation of polymeric by-products or over-oxidized impurities that often plague traditional acid-catalyzed condensations.
This dual-functionality design allows the molecule to act as both a Brønsted acid catalyst and a phase-transfer agent, facilitating the protonation of the carbonyl oxygen in the 1,2-dicarbonyl substrate to enhance its electrophilicity. The long alkyl chain (C12H25) present in the cationic structure imparts amphiphilic characteristics, which improves the solubility of organic reactants within the aqueous phase, effectively creating a micro-emulsion environment that maximizes molecular collisions. The bisulfate (HSO4-) counter-ion further contributes to the acidity of the medium, driving the condensation equilibrium forward without the need for external mineral acids that could degrade sensitive functional groups on the substrate. This precise molecular engineering ensures that the reaction proceeds with high chemoselectivity, minimizing the formation of polymeric by-products or over-oxidized impurities that often plague traditional acid-catalyzed condensations.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining the integrity of the final product profile. Because the reaction occurs at near-room temperature, thermal degradation pathways that typically generate tarry residues or decomposition products in heated organic solvents are effectively suppressed. The aqueous environment also serves as a heat sink, preventing localized hot spots that could lead to runaway reactions or uneven product quality. Furthermore, the ease of separating the solid product from the liquid catalytic phase via simple filtration means that the product is not exposed to prolonged contact with the catalyst after formation, reducing the risk of secondary reactions or hydrolysis. This inherent selectivity translates to a cleaner crude product that requires less aggressive purification steps, ultimately preserving the yield and ensuring that the final quinoxaline derivatives meet the rigorous purity specifications demanded by pharmaceutical applications.
How to Synthesize Quinoxaline Derivatives Efficiently
The implementation of this synthesis route is designed for operational simplicity, requiring standard glass-lined or stainless steel reactors without the need for specialized high-pressure or high-temperature equipment. The process begins with the precise metering of substituted o-phenylenediamine and the 1,2-dicarbonyl compound in a 1:1 molar ratio, followed by the addition of the ionic liquid catalyst at a loading of 5 to 20 mol% relative to the diamine. Water is introduced as the bulk solvent, constituting 50 to 80% of the total mass, creating a homogeneous or semi-homogeneous mixture that is stirred vigorously to ensure adequate mass transfer.
- Prepare the reaction mixture by combining substituted o-phenylenediamine and 1,2-dicarbonyl compound in a 1: 1 molar ratio with the biodegradable ionic liquid catalyst (5-20 mol%) and water (50-80% of total mass).
- Stir the mixture at ambient temperature (20-40°C) for a duration of 10 to 60 minutes to allow the condensation reaction to proceed to completion.
- Filter the resulting solid-liquid two-phase system to isolate the crude product, wash with cold water, and recrystallize from ethanol to obtain pure quinoxaline compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous ionic liquid technology offers profound strategic benefits that extend far beyond simple chemical efficiency. The elimination of expensive and regulated organic solvents like DMSO and glacial acetic acid results in a drastic reduction in raw material procurement costs and lowers the complexity of solvent recovery infrastructure. Since water is the primary medium, the costs associated with solvent purchase, storage, and hazardous waste disposal are significantly diminished, directly improving the gross margin of the manufactured intermediates. The ability to recycle the catalyst directly from the filtrate without complex regeneration steps means that the effective consumption of the catalytic material is minimal, further driving down the variable cost per kilogram of product. These factors combine to create a highly cost-competitive manufacturing process that is resilient against fluctuations in the petrochemical markets that typically drive organic solvent prices.
- Cost Reduction in Manufacturing: The transition to a water-based system fundamentally alters the cost structure of quinoxaline production by removing the dependency on volatile organic compounds. By avoiding the use of solvents that require expensive distillation columns for recovery or incineration for disposal, facilities can realize substantial operational savings. The mild reaction conditions also translate to lower energy utility costs, as there is no need for prolonged heating or refluxing, allowing reactors to turn over much faster. Additionally, the high atom economy and excellent yields reported in the patent minimize the loss of valuable starting materials, ensuring that every dollar spent on raw o-phenylenediamines and dicarbonyls is efficiently converted into saleable product.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent mitigates many of the logistical risks associated with the transport and storage of flammable or toxic organic liquids. This simplifies regulatory compliance and reduces the likelihood of supply disruptions caused by hazardous material transport restrictions. The robustness of the catalyst, which remains stable and active in the aqueous phase, ensures consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that could delay shipments to downstream customers. Furthermore, the simplicity of the workup procedure—filtration followed by recrystallization—allows for faster turnaround times, enabling manufacturers to respond more agilely to sudden increases in market demand.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process make it ideally suited for large-scale industrial expansion without incurring prohibitive environmental remediation costs. The biodegradability of the ionic liquid catalyst ensures that wastewater treatment is straightforward and compliant with increasingly strict global discharge regulations. Scaling up from laboratory to commercial production is facilitated by the absence of exothermic risks associated with strong mineral acids or high-temperature organic reactions, making the process safer for operators and easier to manage in large multipurpose reactors. This environmental compatibility enhances the brand reputation of the supplier as a sustainable partner, aligning with the ESG (Environmental, Social, and Governance) goals of major multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process feasibility. Understanding these details is crucial for technical teams evaluating the integration of this green chemistry approach into existing manufacturing workflows.
Q: What are the primary environmental advantages of this ionic liquid catalyzed method?
A: The method utilizes water as a benign solvent instead of toxic organic solvents like glacial acetic acid or DMSO. Furthermore, the quaternary ammonium-based ionic liquid catalyst is biodegradable, significantly reducing the environmental footprint of the wastewater compared to traditional non-degradable catalysts.
Q: Can the ionic liquid catalyst be recovered and reused?
A: Yes, the process generates a solid-liquid two-phase system upon completion. The filtrate contains the ionic liquid catalyst and can be directly reused for subsequent batches without additional treatment, maintaining catalytic activity and reducing raw material costs.
Q: How does the reaction time compare to conventional organic solvent methods?
A: Conventional methods typically require 2 to 12 hours of heating in organic solvents. In contrast, this aqueous ionic liquid protocol achieves high yields (86-95%) within just 10 to 60 minutes at room temperature, drastically improving throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biodegradable ionic liquid catalysis technology described in CN102010376A for the production of high-value quinoxaline intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of quinoxaline derivative meets the exacting standards required for pharmaceutical and electronic material applications. We are committed to leveraging our technical expertise to optimize this aqueous phase process, delivering a reliable quinoxaline supplier partnership that prioritizes both quality and sustainability.
We invite forward-thinking R&D and procurement leaders to collaborate with us to evaluate the feasibility of this advanced synthesis route for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this water-based protocol. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is equipped with the most efficient and environmentally responsible manufacturing solutions available in the market.
