Advanced Asymmetric Hydrogenation Technology for High-Purity Chiral Lactam Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly lactam derivatives which serve as critical building blocks for numerous bioactive molecules. Patent CN111793017A introduces a groundbreaking preparation method for lactam compounds that addresses longstanding challenges in stereoselectivity and operational safety. This technology leverages a sophisticated asymmetric hydrogenation reaction mediated by specialized ruthenium catalysts and hydrogen donor reagents, bypassing the need for hazardous high-pressure hydrogen gas. The resulting compounds, characterized by the general Formula III, possess two chiral centers and are obtained with exceptional optical purity, often exceeding 98% ee. For R&D directors and process chemists, this represents a paradigm shift from traditional resolution techniques or harsh catalytic hydrogenation, offering a streamlined pathway to complex intermediates used in the synthesis of drugs like Tofacitinib. The ability to access these high-value intermediates with such precision underscores the importance of advanced catalytic systems in modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydroxy-lactams has been fraught with inefficiencies that impact both cost and timeline in drug development. Conventional approaches often rely on direct hydrogenation using molecular hydrogen under high pressure, which necessitates specialized, expensive reactor infrastructure and poses significant safety risks regarding flammability and explosion. Furthermore, many traditional routes yield racemic mixtures or poor diastereomeric ratios, forcing manufacturers to employ cumbersome chiral resolution steps that inherently discard at least 50% of the material, drastically increasing the cost of goods sold. Even when chiral auxiliaries are used, the additional synthetic steps required for their attachment and removal add unnecessary complexity and waste generation. These limitations create bottlenecks in the supply chain, making it difficult to secure reliable quantities of high-purity intermediates needed for clinical trials and commercial launch.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by employing asymmetric transfer hydrogenation, a technique that utilizes liquid hydrogen donors instead of gaseous hydrogen. As illustrated in the reaction scheme below, the precursor compound II is converted directly into the target chiral lactam III under mild conditions.
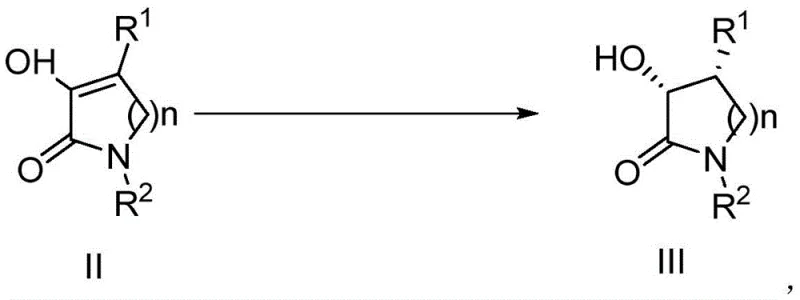
This approach eliminates the need for high-pressure equipment, allowing the reaction to proceed safely in standard glass-lined or stainless steel reactors at atmospheric pressure or slight reflux. The use of specific ruthenium complexes ensures that the hydrogen is delivered selectively to one face of the double bond, establishing the desired stereochemistry with remarkable fidelity. By avoiding the pitfalls of racemic synthesis and resolution, this novel route significantly improves atom economy and reduces the overall environmental footprint of the manufacturing process, aligning perfectly with green chemistry principles demanded by modern regulatory bodies.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological breakthrough lies in the precise design of the chiral ruthenium catalysts, which dictate the stereochemical outcome of the reaction. The patent highlights a family of catalysts, including structures like (R,R)-Ts-DENEB and various CAT series complexes, which feature chiral diamine ligands coordinated to the ruthenium center. These ligands create a rigid chiral environment around the metal, effectively shielding one face of the substrate while directing the hydride transfer from the formic acid donor to the other face. This steric control is crucial for achieving the high diastereomeric ratios observed, often favoring the cis-configured product (RR or SS) over the trans-isomer. The mechanism likely involves the formation of a ruthenium-hydride species in situ, which then coordinates with the enamide or unsaturated lactam substrate to facilitate the concerted addition of hydrogen.
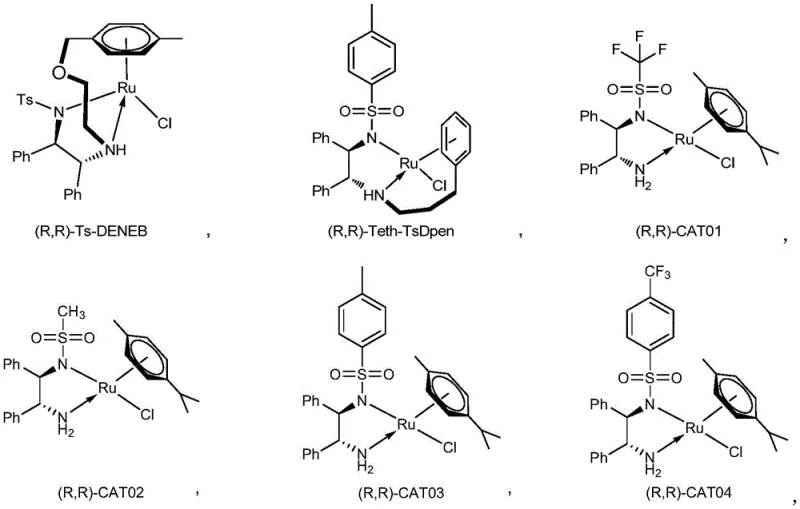
Beyond stereocontrol, the choice of catalyst also influences the reaction kinetics and tolerance to functional groups. The patent data indicates that catalysts such as (R,R)-Ts-DENEB can operate effectively at loadings as low as 0.5 mol%, demonstrating high turnover numbers that are economically viable for large-scale production. The presence of electron-withdrawing or donating groups on the ligand framework allows for fine-tuning the electronic properties of the catalyst, optimizing it for different substrate classes ranging from simple alkyl-substituted lactams to those bearing complex aryl or heteroaryl moieties. This mechanistic understanding enables process chemists to predict outcomes and troubleshoot potential impurities, ensuring that the final API intermediate meets stringent purity specifications required for regulatory filing.
How to Synthesize Chiral Hydroxy-Lactams Efficiently
The synthesis of these valuable intermediates follows a logical progression from readily available starting materials, ensuring a robust and reproducible workflow. The process begins with the preparation of the unsaturated precursor, followed by the critical asymmetric reduction step that installs the chiral centers. Detailed operational parameters regarding temperature, solvent choice, and workup procedures are essential for maximizing yield and optical purity. While the general principles are outlined here, the specific stoichiometry and purification techniques are vital for success.
- Prepare the precursor compound II by cyclization of the corresponding amine ester.
- Dissolve compound II in dichloromethane with a chiral ruthenium catalyst such as (R,R)-Ts-DENEB.
- Add triethylamine and formic acid slowly, then heat to reflux to complete the asymmetric hydrogenation.
- Perform aqueous workup and purification via column chromatography or recrystallization to isolate the target compound III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transfer hydrogenation technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of high-pressure hydrogen gas removes a major safety hazard from the manufacturing site, potentially lowering insurance premiums and simplifying site compliance audits. Moreover, the ability to run reactions at ambient pressure in common solvents like dichloromethane or toluene means that existing manufacturing assets can be utilized without costly retrofitting, accelerating the time to market for new drug candidates. The high selectivity of the process minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification units and increasing overall throughput.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the high efficiency of the catalyst system and the avoidance of wasteful resolution steps. By achieving high enantiomeric excess directly, the process saves the cost of discarding unwanted enantiomers, effectively doubling the yield compared to racemic routes. Additionally, the low catalyst loading required reduces the consumption of expensive precious metals, while the use of inexpensive hydrogen donors like formic acid replaces costly high-pressure hydrogen infrastructure. These factors combine to significantly lower the cost of goods, providing a competitive edge in pricing negotiations for bulk active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesizing chiral intermediates, but this streamlined approach mitigates those risks. The starting materials are commercially available or easily synthesized from commodity chemicals, reducing dependency on niche suppliers. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification results. This reliability allows pharmaceutical companies to maintain leaner inventory levels while confident in the steady flow of critical intermediates needed for continuous manufacturing campaigns.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant is notoriously difficult, yet this technology is inherently scalable due to its mild thermal profile and lack of exothermic hazards associated with high-pressure hydrogenation. The reduced solvent usage and higher atom economy contribute to a smaller environmental footprint, aiding companies in meeting increasingly strict sustainability goals and regulatory emissions standards. The simplified waste stream, devoid of heavy metal contaminants from excessive catalyst use or resolution byproducts, further eases the burden on waste treatment facilities, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is essential for stakeholders evaluating its integration into their supply chains. The following questions address common concerns regarding performance, safety, and applicability, drawing directly from the technical disclosures and experimental data provided in the patent literature. These insights are intended to clarify the operational realities of implementing this advanced catalytic system.
Q: What are the safety advantages of this transfer hydrogenation method?
A: Unlike traditional methods requiring high-pressure hydrogen gas, this process utilizes formic acid/triethylamine as a hydrogen donor, eliminating explosion risks and reducing equipment costs.
Q: What level of stereoselectivity can be achieved?
A: The patented ruthenium catalyst system consistently achieves enantiomeric excess (ee) values exceeding 98% and diastereomeric ratios (dr) up to 99:1.
Q: Is this method scalable for commercial production?
A: Yes, the mild reaction conditions and low catalyst loading (down to 0.5 mol%) make the process highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals depends on the availability of high-quality chiral intermediates produced via cutting-edge technology. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab scale to industrial manufacture is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of specific lactam derivatives or optimization of existing routes, our facility is equipped to handle the most demanding projects with precision and care.
We invite you to collaborate with us to leverage these advanced asymmetric hydrogenation capabilities for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance the efficiency and profitability of your pharmaceutical manufacturing operations.
