Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharmaceutical Applications
Introduction to Next-Generation Biheterocyclic Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient and safer synthetic routes to complex molecular scaffolds. Patent CN115353511A introduces a groundbreaking multicomponent method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole cores. These structural motifs are ubiquitous in bioactive molecules, offering broad-spectrum pharmacological activities that are highly sought after in modern drug discovery programs. The integration of a trifluoromethyl group further enhances the metabolic stability and lipophilicity of these candidates, making them prime targets for medicinal chemistry optimization. This technology represents a significant leap forward, moving away from hazardous traditional methods toward a streamlined, one-pot cascade reaction that maximizes atom economy while minimizing environmental impact.
For research and development teams, the ability to access these complex architectures through a single operational step is transformative. Conventional approaches often require multiple isolation and purification stages, leading to significant yield losses and increased waste generation. By contrast, this novel palladium-catalyzed protocol merges carbonylation, Heck coupling, and cyclization into a unified sequence. The result is a robust platform capable of generating diverse libraries of trifluoromethyl-substituted biheterocycles with high precision. As a reliable pharmaceutical intermediate supplier, understanding and leveraging such advanced methodologies is crucial for maintaining a competitive edge in the global supply chain, ensuring that clients receive high-purity materials derived from state-of-the-art processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has been fraught with synthetic challenges that hinder both academic research and industrial production. Traditional strategies typically rely on the direct coupling of two pre-formed heterocyclic substrates, a process that often suffers from poor regioselectivity and low conversion rates due to steric hindrance. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles require harsh oxidizing agents and elevated temperatures, which can degrade sensitive functional groups and complicate downstream purification. Furthermore, classical carbonylation reactions necessitate the use of carbon monoxide gas, a highly toxic and flammable reagent that demands specialized high-pressure reactors and rigorous safety protocols, significantly inflating capital expenditure and operational risks for manufacturing facilities.
These legacy methods also struggle with substrate scope limitations. Introducing diverse substituents, particularly electron-withdrawing groups like trifluoromethyl moieties, often requires multi-step protection and deprotection sequences that drastically reduce overall efficiency. The inability to tolerate a wide range of functional groups restricts the chemical space available for exploration during lead optimization phases. Consequently, procurement managers and supply chain heads face difficulties in sourcing these intermediates reliably, as few manufacturers possess the technical capability to execute these dangerous and inefficient processes at scale. The industry urgently requires a paradigm shift towards safer, more versatile synthetic technologies that can deliver complex molecular architectures without compromising on safety or cost-effectiveness.
The Novel Approach
The methodology disclosed in patent CN115353511A offers a sophisticated solution to these longstanding problems by employing a transition metal palladium-catalyzed carbonylation cascade reaction. This innovative approach utilizes cheap and readily available starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, to construct the target biheterocyclic framework in a single pot. A critical advantage of this system is the replacement of toxic carbon monoxide gas with a safe mixture of formic acid and acetic anhydride, which generates the necessary carbonyl species in situ under mild conditions. This modification not only eliminates the need for high-pressure equipment but also significantly simplifies the reactor setup, making the process accessible for standard laboratory and pilot plant operations.
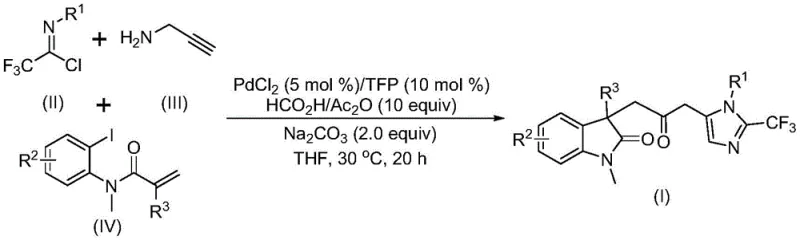
Furthermore, this novel route exhibits exceptional substrate compatibility, accommodating a wide array of substituents on the aromatic rings, including alkyl, alkoxy, halogen, and nitro groups. The reaction proceeds efficiently at a mild temperature of 30°C, preserving the integrity of sensitive functional groups that would otherwise decompose under harsher conditions. For a reliable agrochemical intermediate supplier or pharma partner, this translates to a versatile platform capable of producing a diverse portfolio of analogues without the need for extensive process re-optimization. The high reaction efficiency and simplified post-treatment procedures, involving simple filtration and column chromatography, ensure that the final products meet stringent purity specifications required for clinical applications, thereby reducing the overall cost of goods sold.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is essential for R&D directors aiming to optimize the process for specific client needs. The reaction initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck reaction, where the palladium center inserts into the alkene moiety, forming a divalent alkyl-palladium species. Crucially, the carbonylation step occurs when this intermediate reacts with carbon monoxide released from the decomposition of the formic acid and acetic anhydride mixture. This generates an acyl-palladium intermediate, which serves as the electrophilic hub for the subsequent ring-closing events that define the biheterocyclic core.
Simultaneously, a parallel pathway involves the base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and propargylamine. This reaction yields a trifluoroacetamidine compound, which subsequently undergoes isomerization to a more reactive tautomer. The acyl-palladium intermediate then activates this trifluoroacetamidine species, catalyzing an intramolecular cyclization that最终 constructs the imidazole ring fused to the indolinone scaffold. This intricate dance of catalytic cycles ensures high regioselectivity and minimizes the formation of byproducts. The use of trifurylphosphine (TFP) as a ligand is particularly noteworthy, as it stabilizes the palladium center and facilitates the turnover of the catalytic cycle, contributing to the high yields observed across various substrate combinations.
How to Synthesize Carbonyl-Bridged Biheterocycles Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent quality and reaction parameters to ensure reproducibility and high yield. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques that are common in modern organic synthesis laboratories. The choice of solvent plays a pivotal role, with tetrahydrofuran (THF) identified as the optimal medium due to its ability to dissolve all reactants effectively while promoting the catalytic cycle. The molar ratios are carefully balanced, typically employing a slight excess of propargylamine and acrylamide relative to the trifluoroethylimidoyl chloride to drive the reaction to completion and suppress side reactions. Detailed standardized synthesis steps are provided below to guide process chemists in replicating this high-efficiency transformation.
- Prepare the catalytic system by mixing palladium chloride, TFP ligand, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the substituted acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction temperature at 30°C for 12 to 20 hours to allow the cascade carbonylation and cyclization to proceed to completion.
- Upon completion, filter the mixture, concentrate, and purify the crude product via silica gel column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly address the pain points of procurement managers and supply chain heads in the fine chemical sector. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike traditional methods that may require custom-synthesized, expensive, or hazardous reagents, this process relies on commodity chemicals such as propargylamine and acrylamide, which are abundantly available from global chemical suppliers. This abundance ensures supply continuity and mitigates the risk of production delays caused by raw material shortages. Additionally, the elimination of high-pressure carbon monoxide removes the need for specialized infrastructure, allowing manufacturing to take place in standard multipurpose reactors, thereby maximizing asset utilization and reducing capital investment requirements.
- Cost Reduction in Manufacturing: The economic implications of this method are profound, primarily driven by the reduction in unit operations and safety overheads. By consolidating multiple synthetic steps into a single one-pot reaction, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. The avoidance of toxic gas handling eliminates the need for expensive scrubbing systems and specialized safety training, further driving down operational expenditures. Moreover, the high atom economy of the multicomponent reaction means less waste is generated per kilogram of product, reducing waste disposal costs and aligning with green chemistry principles that are increasingly mandated by regulatory bodies.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply chain reliability for downstream customers. The mild reaction conditions (30°C) and tolerance for various functional groups mean that the process is less prone to batch failures due to minor fluctuations in temperature or reagent quality. This consistency is vital for pharmaceutical clients who require strict adherence to quality standards and consistent delivery schedules. The ability to scale the reaction from gram to kilogram levels without significant re-engineering ensures that supply can be ramped up quickly to meet market demand, providing a strategic advantage in fast-moving therapeutic areas.
- Scalability and Environmental Compliance: As the industry moves towards more sustainable manufacturing practices, this method stands out for its environmental profile. The use of in situ generated carbon monoxide avoids the transportation and storage hazards associated with gas cylinders. Furthermore, the simplified workup procedure, involving filtration and chromatography, reduces the volume of aqueous and organic waste streams. This ease of waste management facilitates compliance with increasingly stringent environmental regulations, protecting the manufacturer from potential fines and reputational damage. The scalability demonstrated in the patent data suggests that this process is ready for commercial scale-up of complex pharmaceutical intermediates, offering a future-proof solution for long-term production needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, based on the detailed experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for your specific project requirements. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing pipelines.
Q: Why is this method safer than traditional carbonylation reactions?
A: Traditional carbonylation often requires high-pressure carbon monoxide gas, which is highly toxic and poses significant safety risks. This patented method utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ under mild conditions, eliminating the need for specialized high-pressure equipment and enhancing operational safety.
Q: What is the substrate compatibility of this synthesis route?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on the aromatic rings, including alkyl, alkoxy, halogen, trifluoromethyl, and nitro groups. This versatility allows for the rapid generation of diverse chemical libraries for drug discovery without needing to protect sensitive functional groups.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly validates the scalability of this method. The reaction operates at a mild temperature of 30°C and uses commercially available, cost-effective raw materials. The successful expansion to gram-scale reactions in the patent data indicates strong potential for kilogram-to-ton scale production in an industrial setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development timelines. Our team of expert process chemists has thoroughly analyzed the potential of this palladium-catalyzed multicomponent reaction and is fully equipped to translate these laboratory findings into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from discovery to market. Our state-of-the-art facilities are designed to handle complex chemistries with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next-generation therapeutic programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data for related analogues, and obtain comprehensive route feasibility assessments tailored to your unique molecular targets. Let us be your trusted partner in navigating the complexities of modern chemical manufacturing.
