Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
The pharmaceutical industry constantly seeks efficient routes to complex heterocyclic scaffolds that serve as the backbone for novel therapeutic agents. Patent CN115353511A discloses a groundbreaking multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in organic synthesis, addressing the longstanding challenges associated with constructing carbonyl bridges between two distinct heterocyclic systems. By leveraging a transition metal palladium-catalyzed cascade reaction, this method avoids the use of toxic carbon monoxide gas, instead utilizing a safe formic acid and acetic anhydride mixture as a carbon monoxide substitute. For R&D directors and process chemists, this offers a robust platform for generating diverse libraries of bioactive molecules with high atom economy and operational simplicity, positioning it as a critical tool for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic compounds containing a carbonyl bridge has been fraught with synthetic difficulties. Conventional literature reports typically rely on three main strategies: direct coupling of two pre-formed heterocyclic substrates, oxidative cyclization of substrates bearing dual nucleophiles with activated methyl-substituted heterocycles, or traditional transition metal-catalyzed tandem cyclizations. The direct coupling approach often suffers from low regioselectivity and poor yields due to the steric hindrance inherent in linking two bulky heterocyclic rings. Furthermore, oxidative cyclization methods frequently require harsh reaction conditions, strong oxidants, and elevated temperatures, which can lead to the decomposition of sensitive functional groups often required in pharmaceutical intermediates. Most critically, traditional carbonylation reactions necessitate the use of carbon monoxide gas, which poses severe safety hazards regarding toxicity and flammability, requiring expensive high-pressure equipment and rigorous safety protocols that increase both capital expenditure and operational complexity in a manufacturing setting.
The Novel Approach
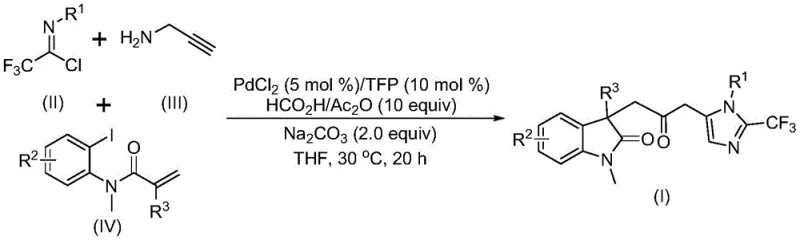
The methodology described in patent CN115353511A fundamentally disrupts these limitations by introducing a one-pot, three-component coupling strategy. This novel approach utilizes readily available starting materials—trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides—to construct the complex biheterocyclic core in a single operation. The brilliance of this system lies in its use of a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, thereby eliminating the need for external CO gas cylinders and high-pressure reactors. This not only enhances laboratory safety but also drastically simplifies the engineering requirements for scale-up. The reaction proceeds under remarkably mild conditions, typically at 30°C, which preserves the integrity of sensitive functional groups and minimizes energy consumption. As illustrated in the general reaction scheme below, this cascade process efficiently builds multiple chemical bonds simultaneously, offering a streamlined pathway to valuable carbonyl-bridged scaffolds.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is vital for process optimization and impurity control. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck-type reaction, which generates a divalent alkyl-palladium intermediate and establishes the indolinone core. Subsequently, the carbon monoxide released from the formic acid and acetic anhydride mixture inserts into the palladium-carbon bond, forming a reactive acyl-palladium intermediate. In a parallel pathway, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine forms a trifluoroacetamidine compound, which undergoes isomerization. The final step involves the activation of this trifluoroacetamidine by the acyl-palladium intermediate, catalyzing an intramolecular cyclization that closes the imidazole ring and delivers the final carbonyl-bridged biheterocyclic product. This intricate dance of catalytic cycles ensures high efficiency and selectivity.
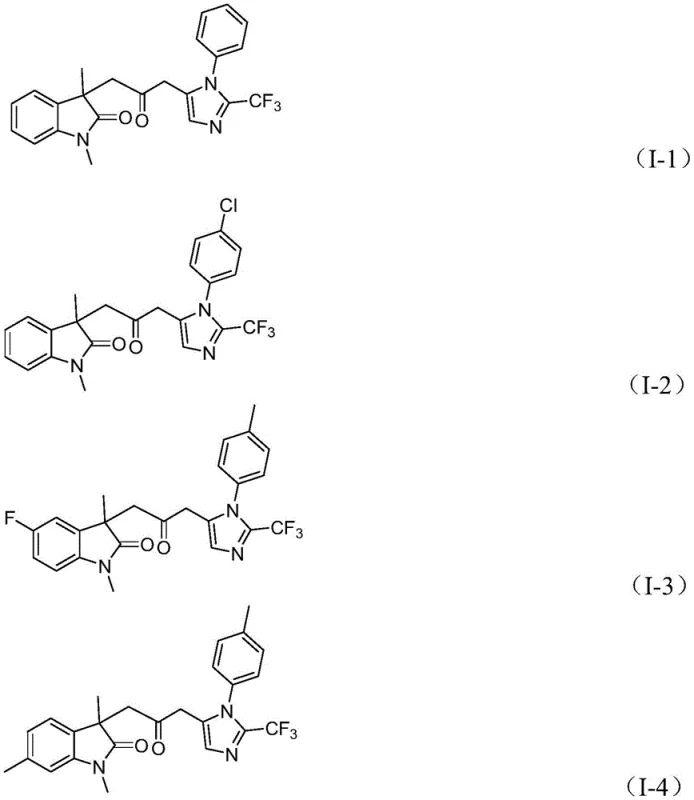
The presence of the trifluoromethyl group adds another layer of complexity and value to these molecules, enhancing their metabolic stability and lipophilicity, which are desirable traits in drug candidates. The mechanism tolerates a wide range of substituents on the aromatic rings, as evidenced by the successful synthesis of various derivatives shown in the structural examples below. Whether the substrate bears electron-donating groups like methyl or methoxy, or electron-withdrawing groups like chloro, fluoro, or nitro, the catalytic system maintains robust performance. This broad substrate compatibility is crucial for medicinal chemists who need to explore structure-activity relationships (SAR) extensively. The ability to introduce diverse R1, R2, and R3 groups without compromising yield demonstrates the versatility of this palladium-catalyzed protocol for generating high-purity pharmaceutical intermediates.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a standardized procedure where palladium chloride and a phosphine ligand (specifically trifurylphosphine or TFP) are combined with sodium carbonate as the base. The carbon monoxide source is prepared by mixing formic acid and acetic anhydride, which is then added to the reaction vessel along with the organic solvent, preferably tetrahydrofuran (THF) for optimal solubility and conversion. The three key building blocks—trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative—are introduced in precise molar ratios, typically with a slight excess of propargylamine and acrylamide to drive the reaction to completion. The mixture is stirred at a controlled temperature of 30°C for a duration of 12 to 20 hours. Post-reaction workup involves simple filtration to remove inorganic salts, followed by silica gel column chromatography to isolate the pure product. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized guide below.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the substituted acrylamide derivative to the reaction vessel.
- Stir the mixture at 30°C for 12 to 20 hours, then filter and purify the crude product via silica gel column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers compelling advantages that directly impact the bottom line and operational resilience. The shift away from hazardous gaseous reagents to liquid alternatives significantly reduces the regulatory burden and safety infrastructure costs associated with manufacturing. Furthermore, the reliance on commodity chemicals ensures a stable and continuous supply of raw materials, mitigating the risk of production delays caused by specialty reagent shortages. The mild reaction conditions also translate to lower energy costs and reduced wear on reactor equipment, contributing to overall process sustainability.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure represents a substantial capital saving. Traditional carbonylation requires specialized autoclaves and rigorous safety monitoring systems, which are expensive to install and maintain. By generating CO in situ from inexpensive formic acid and acetic anhydride, this method allows manufacturers to utilize standard glass-lined or stainless steel reactors. Additionally, the use of cheap and readily available starting materials like propargylamine and acrylamide, which are produced on a massive industrial scale, ensures that raw material costs remain low and predictable. The high reaction efficiency and yield reported in the patent further minimize waste generation, reducing the costs associated with raw material loss and waste disposal.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against varying substrate electronics means that supply chains are less vulnerable to specific raw material bottlenecks. Since the method tolerates a wide array of substituents, procurement teams have the flexibility to source alternative grades of starting materials or switch suppliers without needing to re-validate the entire process. The simplified post-treatment process, involving basic filtration and standard chromatography, streamlines the production timeline. This efficiency allows for faster turnaround times from order to delivery, ensuring that downstream pharmaceutical clients receive their critical intermediates without delay, thereby strengthening the reliability of the entire supply network.
- Scalability and Environmental Compliance: The patent explicitly notes the successful expansion of this reaction to the gram scale, indicating strong potential for kilogram and ton-scale production. The mild temperature of 30°C reduces the thermal load on cooling systems, making it easier to manage heat dissipation in large reactors compared to exothermic high-temperature processes. Moreover, avoiding toxic CO gas aligns with increasingly stringent environmental, health, and safety (EHS) regulations globally. This compliance reduces the risk of regulatory fines and shutdowns, ensuring long-term operational continuity. The reduced hazard profile also simplifies logistics and transportation of reagents, as liquid acids and anhydrides are generally easier to handle and ship than compressed toxic gases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and clients evaluating this technology for their own production needs.
Q: What are the safety advantages of this carbonylation method compared to traditional methods?
A: Unlike traditional carbonylation which requires toxic and high-pressure carbon monoxide gas, this patented method utilizes a formic acid and acetic anhydride mixture to generate CO in situ. This significantly reduces safety risks and eliminates the need for specialized high-pressure autoclaves, making it safer for laboratory and pilot plant operations.
Q: What is the substrate scope for the R1, R2, and R3 groups in this synthesis?
A: The method demonstrates excellent functional group tolerance. R1 can be alkyl or various substituted aryl groups (including methyl, tert-butyl, methoxy, halogens, nitro, and trifluoromethyl). R2 accommodates hydrogen, alkyl, halogens, or trifluoromethyl groups on the indolinone ring, while R3 allows for alkyl, phenyl, or benzyl substituents, enabling the creation of diverse chemical libraries for drug discovery.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly mentions that the method has been expanded to gram-scale reactions with high efficiency. The use of cheap, commercially available starting materials like propargylamine and acrylamide, combined with mild reaction conditions (30°C), suggests strong potential for cost-effective scale-up to industrial production levels without complex engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multicomponent reaction for the pharmaceutical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand that consistency is key in the supply of active pharmaceutical ingredients and intermediates, and our dedicated technical team is committed to maintaining the highest standards of excellence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data for our existing library of biheterocyclic compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you accelerate your drug development timeline with reliable, high-quality chemical solutions.
