Advanced Manufacturing of 6,7-Substituted-4-Anilinoquinazoline Intermediates for Oncology Drugs
Advanced Manufacturing of 6,7-Substituted-4-Anilinoquinazoline Intermediates for Oncology Drugs
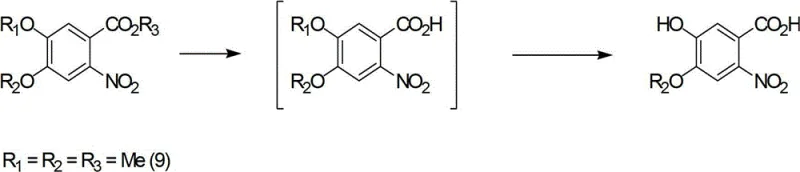
The landscape of oncology therapeutics relies heavily on the precise and efficient synthesis of tyrosine kinase inhibitors, particularly the class of 6,7-substituted-4-anilinoquinazolines which serve as the core scaffold for blockbuster drugs like Gefitinib and Erlotinib. Patent CN102746242A discloses a groundbreaking synthetic methodology that addresses the longstanding economic and technical bottlenecks associated with producing these critical pharmaceutical intermediates. By utilizing 3-methoxy-4-hydroxybenzoic acid as a cost-effective initiator, this novel pathway navigates through a sophisticated sequence of esterification, selective oxygen-alkylation, and a unique hydrolysis-demethylation protocol to achieve superior regiocontrol. This technical advancement is not merely an academic exercise but represents a tangible shift towards more sustainable and economically viable manufacturing processes for high-value anticancer agents. As a leading entity in the fine chemical sector, understanding the nuances of this patent is essential for securing a reliable API intermediate supplier partnership that can deliver consistent quality at scale.
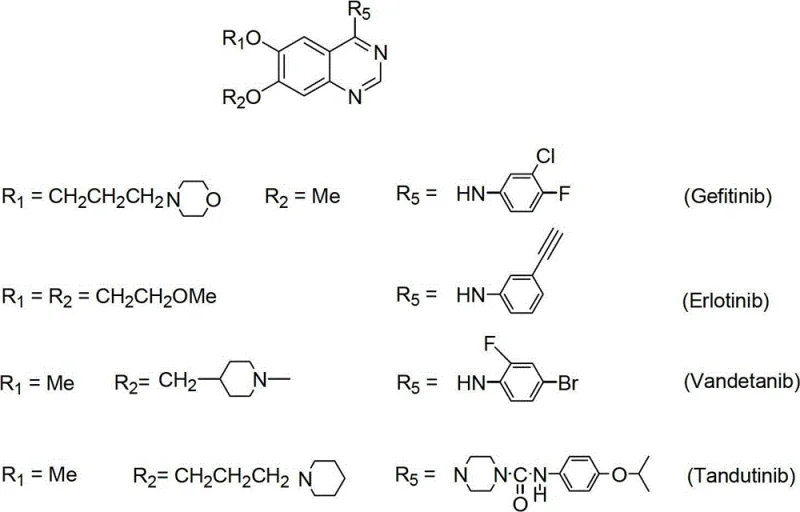
The strategic importance of this synthesis route cannot be overstated, as it directly impacts the supply chain stability for non-small-cell lung cancer (NSCLC) treatments. The patent outlines a versatile approach capable of generating various derivatives, including Vandetanib and Tandutinib, by simply modulating the alkylation and aniline substitution steps. This flexibility is crucial for contract development and manufacturing organizations (CDMOs) aiming to diversify their portfolio without overhauling their entire production infrastructure. The ability to start from readily available benzoic acid derivatives rather than expensive or unstable aldehydes significantly lowers the barrier to entry for production, thereby enhancing the competitiveness of the final drug product in the global market. Furthermore, the disclosed method emphasizes high overall yields and purity specifications that meet the rigorous demands of regulatory bodies, ensuring that the resulting intermediates are suitable for direct progression into final API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6,7-substituted-4-anilinoquinazolines has been plagued by reliance on costly starting materials such as Isovanillin, which often necessitates complex functional group transformations to install the requisite nitrile or amide moieties. Prior art methods, such as those described in WO2004024703 and WO9633980, frequently involve multi-step protection and deprotection strategies that not only extend the production timeline but also introduce significant opportunities for yield loss and impurity generation. For instance, converting an aldehyde to a nitrile and subsequently to an amide involves harsh reagents and conditions that can compromise the integrity of sensitive substituents on the aromatic ring. Additionally, traditional routes often suffer from poor regioselectivity during the nitration or alkylation phases, leading to difficult-to-separate isomers that require extensive chromatographic purification, a process that is notoriously difficult to scale and economically inefficient for cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN102746242A introduces a streamlined trajectory that bypasses many of these inefficiencies by leveraging a robust benzoic acid scaffold. The novel approach utilizes a strategic sequence where esterification and oxygen-alkylation are performed early to establish the substitution pattern, followed by a highly controlled nitration step. A key innovation is the incorporation of a hydrolysis-demethylation-esterification sequence that allows for the precise manipulation of methoxy and hydroxyl groups, enabling the synthesis of diverse analogues without the need for orthogonal protecting groups. This route culminates in a高效的 one-pot chlorination and aniline substitution reaction, which drastically simplifies the final stages of synthesis. By consolidating multiple transformation steps and utilizing cheaper raw materials, this method offers a compelling solution for commercial scale-up of complex polymer additives and pharmaceutical intermediates alike, ensuring a more resilient supply chain.
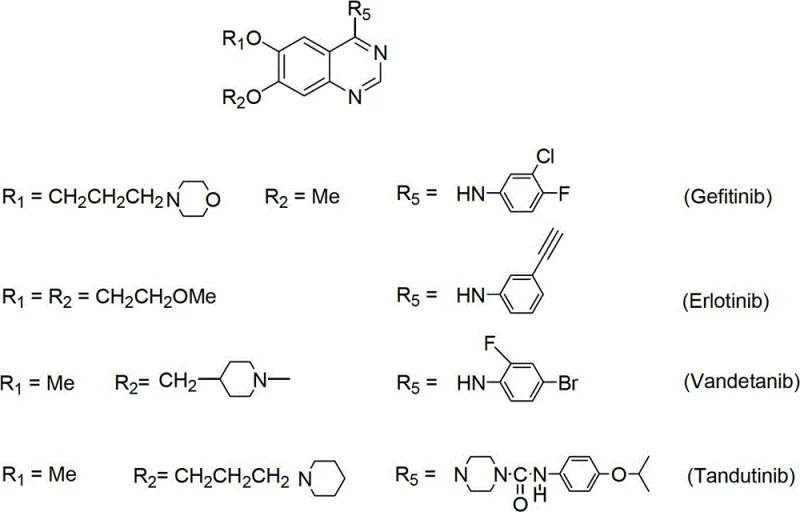
Mechanistic Insights into Selective Demethylation and Cyclization
The core mechanistic brilliance of this patent lies in its handling of the phenolic ether functionalities, specifically the selective demethylation strategy that enables the installation of diverse side chains at the 6 and 7 positions. The process involves a carefully orchestrated hydrolysis-demethylation reaction where strong alkaline conditions or Lewis acids like aluminum chloride are employed to cleave specific methyl ethers in the presence of nitro groups. This step is critical because it generates a free phenolic hydroxyl group at a precise location, which can then be re-alkylated with different halides to introduce morpholine or methoxyethoxy chains as seen in Gefitinib and Erlotinib respectively. The ability to toggle between methoxy and hydroxyl states without affecting the nitro group or the ester functionality demonstrates a high level of chemoselectivity, which is paramount for maintaining the structural integrity of the intermediate throughout the synthesis. This mechanistic control ensures that the final cyclization precursor possesses the exact substitution pattern required for biological activity.
Following the establishment of the correct substitution pattern, the cyclization mechanism proceeds through the condensation of the amino-benzoate intermediate with formamide or ammonium acetate sources. This ring-closing step forms the quinazolin-4-one core, a rigid heterocyclic structure that is essential for binding to the ATP pocket of the EGFR receptor. The patent details the use of formamide at elevated temperatures (100-200°C) to drive this dehydration cyclization to completion, ensuring high conversion rates. Subsequent chlorination of the 4-position activates the ring for nucleophilic aromatic substitution with various anilines. The final displacement reaction is executed in a one-pot fashion, where the chloro-intermediate reacts directly with the substituted aniline in the presence of a base, minimizing the isolation of unstable chloro-species. This integrated approach not only enhances safety by avoiding the accumulation of reactive intermediates but also significantly improves the overall mass balance of the process, leading to the reported high purity levels of >99.7%.
How to Synthesize 6,7-Substituted-4-Anilinoquinazoline Efficiently
Implementing this synthesis route requires a disciplined approach to reaction engineering, particularly regarding temperature control during the exothermic nitration and demethylation steps. The process begins with the esterification of the benzoic acid starting material, typically using thionyl chloride or Fischer conditions, followed by alkylation with specific halo-alkanes to set the R1 and R2 groups. Operators must pay close attention to the stoichiometry during the nitration phase to prevent over-nitration, which can lead to dinitro byproducts that are difficult to remove. The subsequent reduction of the nitro group to an amine can be achieved via catalytic hydrogenation or chemical reduction with sodium dithionite, depending on the available infrastructure and safety protocols. Detailed standardized synthesis steps see the guide below for specific parameters regarding solvent selection, reaction times, and workup procedures that are critical for reproducing the high yields described in the patent literature.
- Initiate the process with esterification of 3-methoxy-4-hydroxybenzoic acid followed by selective oxygen-alkylation to introduce specific side chains.
- Perform nitration under controlled acidic conditions, followed by a critical hydrolysis-demethylation sequence to position the hydroxyl group accurately.
- Execute nitro-reduction, cyclization with formamide derivatives, and a final one-pot chlorination-aniline substitution to yield the high-purity quinazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis methodology offers profound strategic advantages that extend beyond simple unit price reductions. By shifting the starting material basis to abundant benzoic acid derivatives, the process decouples production from the volatile pricing and availability issues often associated with specialized aldehyde precursors like Isovanillin. This fundamental change in raw material sourcing enhances supply chain resilience, ensuring that production schedules are not disrupted by upstream shortages. Furthermore, the reduction in the total number of isolation steps and the elimination of complex protection-deprotection sequences translate directly into lower operational expenditures (OPEX) related to solvent usage, waste disposal, and labor hours. These efficiencies compound to create a significantly more competitive cost structure for the final intermediate, allowing downstream API manufacturers to optimize their own margins while maintaining high quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive reagents and the consolidation of multiple reaction steps into fewer operational units drastically reduce the consumption of solvents and energy. By avoiding the use of precious metal catalysts in certain reduction steps or replacing them with more economical chemical reductants, the process achieves substantial cost savings without compromising reaction efficiency. Additionally, the high yield of the final one-pot substitution reaction minimizes the loss of valuable aniline starting materials, further contributing to the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as 3-methoxy-4-hydroxybenzoic acid ensures a stable and diversified supply base, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, allows for greater flexibility in manufacturing site selection and batch scheduling. This reliability is critical for meeting the stringent delivery timelines required by global pharmaceutical clients, ensuring that high-purity pharmaceutical intermediates are available exactly when needed to support clinical and commercial drug production.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from pilot plant to multi-ton commercial reactors. The simplified workup procedures, often involving straightforward crystallization or extraction, reduce the generation of hazardous waste streams and facilitate easier compliance with environmental regulations. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing organization, a factor that is increasingly important to stakeholders and investors in the modern chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows. The answers highlight the specific advantages in terms of purity, yield, and operational simplicity that distinguish this method from legacy technologies.
Q: What is the primary advantage of this synthesis route over conventional Isovanillin methods?
A: The primary advantage lies in the use of lower-cost starting materials like 3-methoxy-4-hydroxybenzoic acid and the implementation of a streamlined hydrolysis-demethylation step that avoids complex protection-deprotection sequences found in older literature, resulting in higher overall yields and simplified purification.
Q: How does the process ensure high purity for the final API intermediate?
A: High purity exceeding 99.7% is achieved through precise control of the nitration regioselectivity and the utilization of a one-pot chlorination-aniline substitution reaction in the final stage, which minimizes the formation of side products and facilitates efficient crystallization of the target quinazoline derivative.
Q: Is this method scalable for commercial production of EGFR inhibitors?
A: Yes, the method is designed for scalability, utilizing robust reaction conditions such as standard Fischer esterification and catalytic hydrogenation or chemical reduction, which are well-established in industrial settings, allowing for reliable commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Substituted-4-Anilinoquinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of 6,7-substituted-4-anilinoquinazoline meets the exacting standards required for oncology drug development. Our commitment to quality assurance means that you can rely on us to deliver intermediates that facilitate smooth downstream processing and regulatory approval.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient route. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain. Let us collaborate to bring life-saving medications to patients faster and more affordably through superior chemical manufacturing.
