Advanced Palladium-Catalyzed Synthesis of High-Purity Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Manufacturing
The innovative methodology detailed in Chinese patent CN115353511A presents a significant advancement in the synthesis of carbonyl-bridged biheterocyclic compounds, offering pharmaceutical manufacturers a safer, more efficient route to critical intermediates. This multi-component approach utilizes palladium catalysis with carbon monoxide substitutes instead of toxic CO gas, operating under mild conditions at 30°C for 12-20 hours. The process demonstrates exceptional substrate compatibility across diverse functional groups while maintaining high reaction efficiency, making it particularly valuable for producing complex intermediates required in modern drug development pipelines.
Advanced Reaction Mechanism and Purity Control
The reaction mechanism involves a sophisticated cascade process beginning with zero-valent palladium insertion into the carbon-iodine bond followed by intramolecular Heck reaction to generate a divalent alkyl palladium intermediate. This intermediate then undergoes carbonylation using carbon monoxide released from formic acid/acetic anhydride mixture, forming an acyl palladium species that drives the subsequent transformation steps. The base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine creates a trifluoroacetamidine compound that undergoes isomerization before being activated by the acyl palladium intermediate to complete the cyclization process.
Purity control is achieved through the elimination of transition metal contamination pathways that commonly plague traditional carbonylation methods. The use of palladium chloride with trifuryl phosphine ligand creates a highly selective catalytic system that minimizes side reactions while maintaining excellent functional group tolerance across various substituents (R1, R2, R3). The mild reaction conditions at 30°C prevent thermal degradation of sensitive functional groups, while the one-pot nature of the process reduces opportunities for impurity formation during intermediate handling. The resulting carbonyl-bridged biheterocyclic compounds feature precise structural control with trifluoromethyl and carbonyl functional groups that are essential for pharmaceutical activity, as confirmed by comprehensive NMR and HRMS analysis in the patent examples.
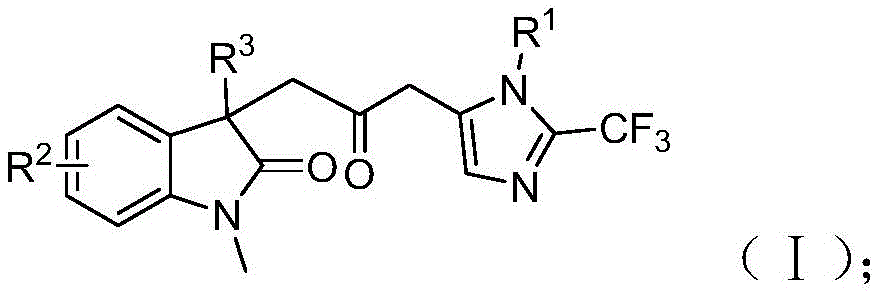
Commercial Advantages and Supply Chain Benefits
This novel synthetic approach addresses critical pain points in pharmaceutical intermediate manufacturing by eliminating hazardous materials while improving process economics and scalability. The methodology provides pharmaceutical companies with a more sustainable pathway to complex heterocyclic structures that are increasingly important in modern drug discovery programs, particularly for targets requiring trifluoromethyl-containing motifs with specific spatial arrangements.
- Elimination of toxic carbon monoxide handling: The substitution of hazardous CO gas with formic acid/acetic anhydride mixture removes significant safety risks associated with high-pressure CO systems, reducing facility requirements and operational complexity. This change eliminates the need for specialized pressure equipment and extensive safety protocols, resulting in lower capital expenditure for manufacturing facilities while improving workplace safety metrics. The removal of CO-related infrastructure also simplifies regulatory compliance and reduces insurance costs, creating substantial operational savings without compromising reaction efficiency or product quality.
- Reduced raw material costs: The use of commercially available starting materials including inexpensive palladium chloride catalyst and readily accessible propargylamine creates immediate cost advantages over conventional methods requiring specialized reagents. The patent demonstrates that trifluoroethylimidoyl chloride can be synthesized from common precursors like triphenylphosphine and carbon tetrachloride, while acrylamide derivatives are easily prepared from acryloyl chloride and corresponding anilines. This supply chain flexibility ensures stable pricing and availability of key inputs, protecting manufacturers from volatile markets for specialized chemical reagents while maintaining consistent product quality across production batches.
- Enhanced process scalability: The mild reaction conditions at ambient temperature (30°C) and atmospheric pressure enable straightforward scale-up from laboratory to commercial production without requiring major engineering modifications. The one-pot nature of the reaction minimizes intermediate isolation steps, reducing processing time and material losses during scale-up transitions. The demonstrated gram-scale feasibility in the patent provides confidence for further industrial implementation, while the simple workup procedure involving filtration and column chromatography can be readily adapted to continuous manufacturing platforms for even greater efficiency gains in large-scale production environments.
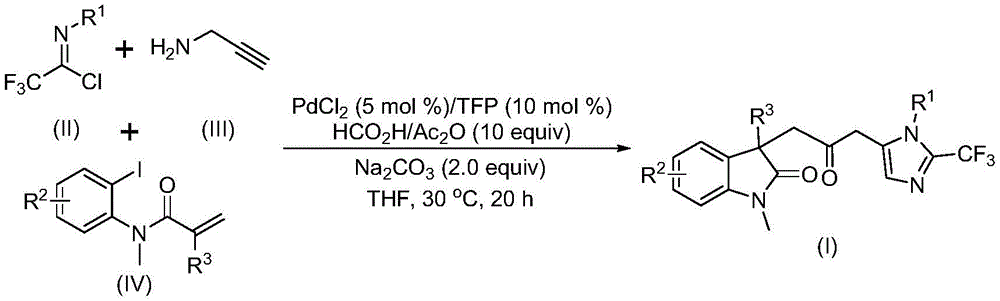
Traditional vs. Innovative Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds typically rely on three main strategies: direct coupling of two heterocycle substrates, oxidative cyclization using bis-nucleophilic substrates with activated methyl groups, or transition metal-catalyzed tandem cyclization reactions. These conventional methods often require harsh reaction conditions including high temperatures, elevated pressures, or specialized equipment for handling toxic gases like carbon monoxide. The need for precise stoichiometric control and multiple isolation steps frequently leads to reduced yields, increased impurity profiles, and higher production costs that become particularly problematic when scaling to commercial manufacturing volumes.
The Novel Approach
The patented methodology overcomes these limitations through an elegant multi-component cascade reaction that operates under remarkably mild conditions while maintaining excellent substrate scope and functional group tolerance. By replacing toxic carbon monoxide with a formic acid/acetic anhydride mixture as the CO source, the process eliminates significant safety hazards while maintaining high reaction efficiency. The use of inexpensive palladium chloride catalyst with trifuryl phosphine ligand creates a cost-effective system that delivers consistent results across diverse substrate combinations, as demonstrated by the successful synthesis of multiple compound variants (I-1 through I-5) with varying substituents.


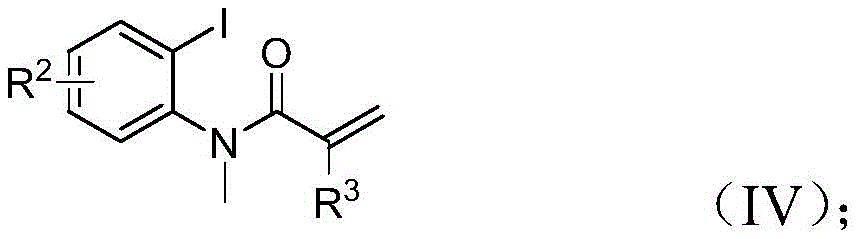
The process demonstrates exceptional versatility through its ability to accommodate various substituents on all three starting materials, enabling pharmaceutical companies to rapidly generate diverse compound libraries for structure-activity relationship studies. The simple workup procedure involving filtration followed by column chromatography purification ensures high product purity without requiring complex separation techniques, while the demonstrated scalability to gram quantities provides confidence for commercial implementation. This methodology represents a significant step forward in sustainable pharmaceutical intermediate manufacturing by combining safety improvements with economic benefits and enhanced synthetic flexibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN115353511A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
