Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial API Production
Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial API Production
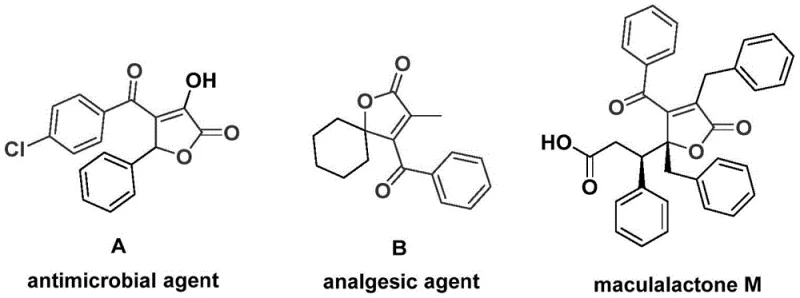
The structural motif of 4-acyl-2(5H)-furanone represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and synthetic pharmaceuticals. As illustrated in the foundational research, compounds possessing this skeleton exhibit potent biological activities, ranging from significant antibacterial properties to analgesic effects and antifouling capabilities against marine bacterial pollution. The efficient construction of this heterocyclic system has long been a challenge for process chemists due to the complexity of traditional ring-forming reactions. Addressing this critical gap, the invention disclosed in patent CN112239436A introduces a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation. This novel approach transforms simple, commercially available precursors—specifically propiolic alcohols and aryl trifluoromethanesulfonates—into high-value 4-acyl-2(5H)-furanone derivatives in a single operational step. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology offers a robust pathway to access complex molecular architectures with exceptional efficiency and purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanone derivatives has relied on multi-step sequences that often involve harsh reaction conditions, sensitive reagents, and tedious purification protocols. Traditional routes frequently necessitate the use of gaseous carbon monoxide under high pressure, which poses significant safety hazards and requires specialized high-pressure autoclaves that are not universally available in standard laboratory or pilot plant settings. Furthermore, conventional methodologies often suffer from poor atom economy and limited substrate scope, where the introduction of electron-withdrawing or bulky substituents can drastically reduce reaction yields or lead to complete failure. The reliance on unstable intermediates and the generation of substantial chemical waste during lengthy synthetic pathways also contribute to elevated production costs and environmental burdens. These factors collectively hinder the rapid scale-up and commercial viability of furanone-based drug candidates, creating a bottleneck for cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the patented process described in CN112239436A utilizes a sophisticated yet operationally simple palladium-catalyzed cascade reaction. By employing chromium hexacarbonyl as a solid carbon monoxide surrogate, the method completely circumvents the need for handling toxic CO gas, thereby enhancing workplace safety and simplifying reactor design. The reaction proceeds through a tandem insertion mechanism where two carbonyl units are sequentially incorporated into the organic framework, followed by spontaneous cyclization to form the furanone ring. This one-pot strategy not only reduces the number of unit operations but also significantly improves the overall mass balance of the process. The use of aryl triflates as electrophiles allows for the modular assembly of diverse aryl groups, enabling the rapid generation of analog libraries for structure-activity relationship (SAR) studies. This technological leap provides a scalable solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The core of this innovation lies in the elegant catalytic cycle driven by a palladium(0) species generated in situ from palladium acetate and the bidentate phosphine ligand DPPF (1,1'-bis(diphenylphosphino)ferrocene). The reaction initiates with the oxidative addition of the aryl trifluoromethanesulfonate to the active Pd(0) catalyst, forming a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is slowly liberated from the decomposition of chromium hexacarbonyl under the reaction conditions, inserts into the palladium-carbon bond to generate an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack or coordination with the propiolic alcohol substrate, leading to the formation of a five-membered ring palladium complex. A second insertion of carbon monoxide occurs, expanding the metallacycle to a six-membered intermediate, which finally undergoes reductive elimination to release the desired 4-acyl-2(5H)-furanone product and regenerate the Pd(0) catalyst. This intricate dance of organometallic steps ensures high regioselectivity and minimizes the formation of side products.
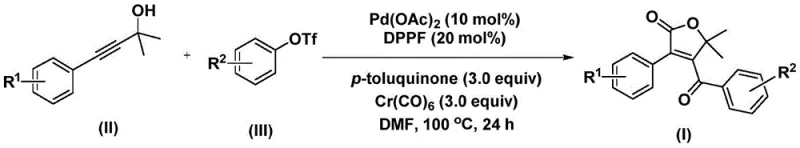
From an impurity control perspective, the choice of additives plays a pivotal role in maintaining catalyst turnover and preventing deactivation. The inclusion of p-toluquinone acts as an oxidant or stabilizer that helps maintain the palladium in the active oxidation state and suppresses the formation of palladium black, a common cause of catalyst precipitation and reaction stalling. Furthermore, the use of polar aprotic solvents like N,N-dimethylformamide (DMF) facilitates the solubility of the ionic intermediates and promotes the smooth progression of the carbonylation steps. The reaction conditions are optimized to run at temperatures between 100°C and 120°C, which provides sufficient thermal energy to drive the CO insertion steps without causing thermal degradation of the sensitive furanone ring. This precise control over the mechanistic parameters results in clean reaction profiles, as evidenced by the high isolated yields reported across various substrates, making it an ideal candidate for high-purity pharmaceutical intermediate production.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
The experimental protocol outlined in the patent provides a straightforward recipe for executing this transformation with high reproducibility. The process involves charging a reaction vessel with the palladium catalyst system, the solid CO source, and the organic substrates in a defined molar ratio, followed by heating under inert atmosphere. The simplicity of the work-up procedure, involving filtration and standard silica gel chromatography, further underscores the practicality of this method for both laboratory discovery and pilot-scale manufacturing. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Combine palladium acetate, DPPF ligand, chromium hexacarbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to facilitate bis-carbonylation and cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials; both propiolic alcohols and aryl triflates are commodity chemicals that can be sourced from multiple global vendors, reducing the risk of single-source dependency. The elimination of high-pressure gas infrastructure lowers the capital expenditure (CAPEX) required for setting up production lines, while the use of a solid CO surrogate minimizes regulatory hurdles associated with toxic gas storage and transport. This translates directly into a more resilient and agile supply chain capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its high atom efficiency and reduced processing time. By consolidating what would traditionally be a multi-step synthesis into a single pot, the method significantly lowers labor costs, solvent consumption, and energy usage per kilogram of product. The high yields achieved (often exceeding 80-90% for optimized substrates) mean that less raw material is wasted, directly improving the cost of goods sold (COGS). Additionally, the avoidance of expensive high-pressure equipment maintenance and safety monitoring further contributes to substantial operational cost savings, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The broad substrate tolerance means that the same production line can be easily adapted to synthesize a variety of furanone derivatives simply by swapping the aryl triflate or propiolic alcohol starting materials. This flexibility allows manufacturers to pivot quickly between different products based on customer demand without requiring extensive re-validation or equipment changes. Furthermore, the stability of the reagents allows for longer shelf-life and easier logistics, reducing lead time for high-purity pharmaceutical intermediates and ensuring just-in-time delivery capabilities.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of hazardous gas feeds. The use of DMF as a solvent, while requiring proper recovery systems, is well-understood in the industry, and the solid waste generated (primarily chromium salts and silica) can be managed through standard waste treatment protocols. The high selectivity of the reaction minimizes the formation of difficult-to-remove byproducts, simplifying the purification train and reducing the volume of organic waste streams. This aligns with modern green chemistry principles and helps companies meet increasingly stringent environmental regulations, securing their social license to operate and avoiding potential fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this process for adoption.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method utilizes a one-pot bis-carbonylation strategy that eliminates complex multi-step sequences. It employs cheap and readily available starting materials like propiolic alcohol and aryl triflates, achieving high conversion rates under relatively mild conditions (100°C) without requiring high-pressure carbon monoxide gas cylinders.
Q: What is the substrate compatibility for R1 and R2 groups?
A: The process demonstrates excellent functional group tolerance. R1 and R2 can independently be hydrogen, C1-C6 alkyl groups (methyl, ethyl, n-propyl), C1-C6 alkoxy groups (methoxy), halogens (F, Cl, Br), or trifluoromethyl groups, allowing for the synthesis of diverse derivatives for SAR studies.
Q: How is the carbon monoxide source managed safely in this protocol?
A: Instead of using toxic and hazardous gaseous carbon monoxide directly, the protocol utilizes chromium hexacarbonyl (Cr(CO)6) as a solid CO surrogate. This significantly enhances operational safety and simplifies the equipment requirements for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112239436A for accelerating drug development pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies, guaranteeing that every batch of 4-acyl-2(5H)-furanone intermediate meets the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and profitability.
