Advanced Palladium-Catalyzed Bis-Carbonylation for High-Purity 4-Acyl-2(5H)-Furanone Intermediates
Advanced Palladium-Catalyzed Bis-Carbonylation for High-Purity 4-Acyl-2(5H)-Furanone Intermediates
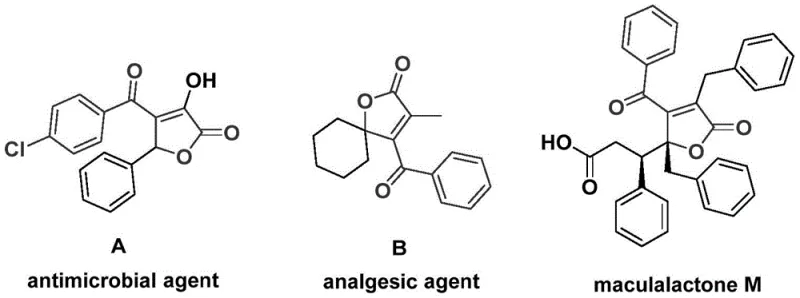
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical backbones for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a highly efficient preparation method for 4-acyl-2(5H)-furanone compounds. This structural motif is not merely an academic curiosity but a privileged scaffold found in numerous natural products and therapeutic agents, ranging from antimicrobial agents to potent analgesics. The disclosed technology leverages a sophisticated palladium-catalyzed bis-carbonylation strategy, utilizing readily available propiolic alcohols and aryl trifluoromethanesulfonates as starting materials. By operating under relatively mild thermal conditions between 100 °C and 120 °C, this process achieves the construction of the furanone ring and the installation of two carbonyl functionalities in a single operational step. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more streamlined, cost-effective, and scalable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanone derivatives has been fraught with synthetic challenges that hinder large-scale production and commercial viability. Traditional routes often rely on multi-step sequences involving harsh reagents, unstable intermediates, or the direct use of hazardous carbon monoxide gas under high pressure. These conventional approaches frequently suffer from poor atom economy, where significant portions of the starting materials end up as waste rather than incorporated into the final product. Furthermore, the requirement for specialized high-pressure equipment to handle gaseous CO introduces substantial capital expenditure and safety risks, creating bottlenecks in the supply chain for contract development and manufacturing organizations (CDMOs). The lack of functional group tolerance in older methodologies often necessitates tedious protection and deprotection strategies, further elongating the production timeline and driving up the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API).
The Novel Approach
In stark contrast, the methodology outlined in patent CN112239436A offers a transformative solution by employing a palladium-catalyzed domino reaction that elegantly assembles the target skeleton in one pot. This novel approach utilizes chromium hexacarbonyl [Cr(CO)₆] as a solid, safe, and convenient surrogate for carbon monoxide gas, thereby eliminating the need for dangerous high-pressure gas cylinders and specialized infrastructure. The reaction proceeds through a tandem sequence where the palladium catalyst mediates the insertion of two distinct carbonyl units derived from the chromium complex, followed by cyclization. This design allows for exceptional substrate flexibility, accommodating a wide array of substituents on both the alkyne and the aryl triflate components. The operational simplicity is further enhanced by the use of common organic solvents like N,N-dimethylformamide (DMF) and standard heating conditions, making this process immediately transferable to existing pilot and commercial plants without major retrofitting.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogues. The catalytic cycle initiates with the oxidative addition of the aryl trifluoromethanesulfonate to the active palladium(0) species, generating an aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is liberated in situ from the thermal decomposition of chromium hexacarbonyl, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack or coordination with the propiolic alcohol substrate, leading to the formation of a five-membered ring palladium intermediate. The uniqueness of this bis-carbonylation lies in the second insertion event, where another molecule of carbon monoxide inserts into the organopalladium bond, expanding the system to a six-membered ring palladium intermediate. Finally, reductive elimination occurs, releasing the desired 4-acyl-2(5H)-furanone product and regenerating the active palladium catalyst to continue the cycle. This intricate dance of coordination and insertion ensures high regioselectivity and minimizes the formation of side products.
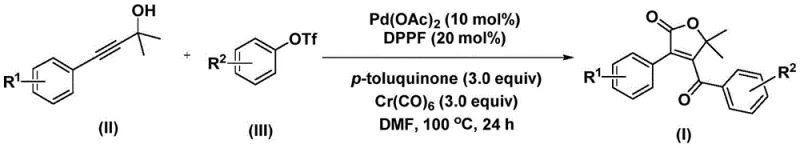
From an impurity control perspective, the choice of additives plays a pivotal role in maintaining the efficiency of the catalytic cycle. The patent specifies the use of p-toluquinone as an additive, which likely functions as an oxidant or a ligand modifier to prevent the aggregation of palladium black, a common deactivation pathway in carbonylation reactions. By stabilizing the active catalytic species, the system maintains high turnover numbers, which directly correlates to lower residual metal content in the crude product. This is a critical quality attribute for pharmaceutical intermediates, as stringent limits on heavy metals like palladium must be met before a compound can advance to clinical stages. The mechanism inherently favors the formation of the thermodynamically stable furanone ring, reducing the likelihood of linear polymerization or alternative cyclization modes that often plague alkyne carbonylation reactions.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of scale-up, addressing the common pain points of laboratory-to-plant technology transfer. The protocol involves charging a reaction vessel with precise molar ratios of palladium acetate (10 mol%), the bidentate ligand DPPF (20 mol%), chromium hexacarbonyl (3.0 equivalents), and p-toluquinone (3.0 equivalents). To this mixture, the propiolic alcohol and aryl triflate substrates are added along with the solvent, typically DMF, at a concentration that ensures complete dissolution of the solids. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are provided in the guide below to ensure consistent batch-to-batch quality.
- Combine palladium acetate, DPPF ligand, chromium hexacarbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to facilitate bis-carbonylation and cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical elegance. The shift from gaseous CO to solid Cr(CO)₆ fundamentally alters the risk profile of the manufacturing process, removing the need for specialized high-pressure reactors and the associated regulatory hurdles for handling toxic gases. This simplification translates directly into reduced capital expenditure and lower operational overheads, allowing for more competitive pricing structures in the global market. Furthermore, the use of commercially available starting materials, such as substituted phenols (precursors to triflates) and terminal alkynols, ensures a robust and diversified supply base that is less susceptible to single-source disruptions.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the consolidation of multiple synthetic steps into a single pot operation. By eliminating intermediate isolation and purification stages, manufacturers can significantly reduce solvent consumption, labor hours, and energy usage associated with heating and cooling cycles. The high reaction efficiency reported in the patent data, with yields reaching up to 95% for certain substrates, implies a drastic reduction in raw material waste. Additionally, the avoidance of expensive and hazardous gaseous reagents lowers the overall cost of goods, making the production of these complex furanone intermediates financially viable even at smaller scales.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like chromium hexacarbonyl and aryl triflates enhances the reliability of the supply chain by mitigating the risks associated with the storage and transport of compressed gases. These materials have long shelf lives and can be stocked in bulk without degradation, ensuring continuity of supply even during periods of logistical volatility. The broad substrate scope demonstrated in the patent examples means that a single manufacturing platform can be adapted to produce a wide library of analogues by simply swapping the starting aryl triflate or alkyne. This flexibility allows suppliers to respond rapidly to changing customer demands for specific derivatives without retooling the entire production line.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic gas feed controls. The post-treatment process described involves simple filtration and standard column chromatography, techniques that are easily adaptable to industrial crystallization or preparative HPLC if necessary. From an environmental standpoint, while chromium waste must be managed responsibly, the overall atom economy is superior to traditional multi-step routes. The ability to run the reaction in polar aprotic solvents like DMF, which are common in the industry, simplifies solvent recovery and recycling protocols, aligning with modern green chemistry initiatives and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed bis-carbonylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction parameters and scope limitations.
Q: What is the primary advantage of using Chromium Hexacarbonyl in this synthesis?
A: Chromium hexacarbonyl serves as a solid, safe, and controllable substitute for toxic carbon monoxide gas, releasing CO in situ under heating conditions to drive the carbonylation steps efficiently without requiring high-pressure gas equipment.
Q: Does this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl groups.
Q: What are the typical reaction yields for this transformation?
A: The patent data indicates high reaction efficiency, with isolated yields ranging significantly, often reaching up to 95% for optimized substrates like 4-methoxy derivatives, ensuring minimal material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
As the demand for complex heterocyclic intermediates continues to grow in the pharmaceutical sector, partnering with an experienced CDMO is essential for navigating the complexities of process development and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the benchtop to the marketplace. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for API intermediates.
We invite you to leverage our technical expertise to optimize your supply chain for 4-acyl-2(5H)-furanone derivatives. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential partners to contact us directly to obtain specific COA data for our reference standards and to discuss route feasibility assessments for your proprietary analogues, ensuring a collaborative path toward successful product launch.
